

Solutions and Solubility Curves
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Standards-aligned
Erica Britton
FREE Resource
17 Slides • 33 Questions
1

2

3
Multiple Choice

Which of the following refers to the substance that is being dissolved in a solution?
solvent
solute
mixture
4
Multiple Choice
In sweet tea the solvent would be
water
tea
sugar
lemon
5

6
Multiple Choice
Which of the following is a solvent?
Salt
Water
Sugar
Oxygen
7
Multiple Choice
Electrolytes contain atoms from what type of bonds that help them conduct electricity?
Ionic
Covalent
Metallic
Polar
8
Multiple Choice

What type of bonds form partial charges allowing it to dissolve or like molecules?
Non-polar covalent bonds
Ionic bonds
Polar covalent bonds
Metallic bonds
9
Multiple Choice
What type of molecules consist of ions and can conduct electricity?
Water
Solutions
Electrolytes
10
Multiple Choice
What property of water makes it an excellent solvent?
Its solid state density
Its molecular weight
Its polarity
Its boiling point
11
Multiple Choice

Which of the following is a reason why oil (a non-polar substance) does not mix with water?
Oil molecules are too large to interact with water molecules
Oil and water have the same density
Oil molecules cannot bonds with water molecules due to intermolecular forces
Oil is denser than water
12

13
Multiple Choice
What type of substance dissolves in water but does not form ions or conduct electricity?
electrolyte
nonelectrolyte
saturated
insoluble
14
Multiple Choice
15
Multiple Choice
16
Multiple Choice
Electrolytes have a greater effect on colligative properties than nonelectrolytes do because electrolytes
are volatile
have higher boiling points
produce fewer moles of solute particles per mole of solvent
produce more moles of solute particles per mole of solvent
17

18
Match
Match the following descriptions to the correct type of electrolyte:
Weak electrolytes
Non-electrolytes
Strong electrolytes
Insulators
Compounds whose aqueous solutions conduct electricity poorly due to partial dissociation of ions
Compounds whose aqueous solutions do not conduct electricity because they do not dissociate into ions
Compounds whose aqueous solutions conduct electricity effectively due to complete dissociation of ions
Materials that do not conduct electricity
Compounds whose aqueous solutions conduct electricity poorly due to partial dissociation of ions
Compounds whose aqueous solutions do not conduct electricity because they do not dissociate into ions
Compounds whose aqueous solutions conduct electricity effectively due to complete dissociation of ions
Materials that do not conduct electricity
19
Explanation Slide...
Compounds that can conduct electricity effectively due to complete dissociation of ions are called strong electrolytes.
20
Multiple Choice
What happens to NaCl when it is dissolved in water?
A) It forms a precipitate
B) It decomposes into gases
C) It dissociates into Na+ and Cl- ions
D) It reacts to form a new compound
21
Explanation Slide...
When NaCl is dissolved in water, it dissociates into Na+ and Cl- ions, which are surrounded by water molecules due to their charges.
22
Match
Match the following numbers of moles produced when dissolved in solution to their corresponding compounds.
2 moles
3 moles
4 moles
5 moles
NaCl dissolved in solution produces:
MgCl2 dissolved in solution produces:
Al2(SO4)3 dissolved in solution produces:
K2SO4 dissolved in solution produces:
NaCl dissolved in solution produces:
MgCl2 dissolved in solution produces:
Al2(SO4)3 dissolved in solution produces:
K2SO4 dissolved in solution produces:
23
Explanation Slide...
When MgCl2 is dissolved, it produces 3 moles of ions: 1 mole of Mg2+ ions and 2 moles of Cl- ions, totaling 3 moles of ions.
24

25
Multiple Choice
The exposed area of a solid; expressed as squared units of length
Volume
Surface Area
Molarity
Solubility
26
Multiple Choice
The stirring or mixing of a solution as to increase the particle movement of the solute particles in solution
Agitation
Settling
Falling Out
Crystallization
27

28

29

30

31

32

33

34

35
Multiple Choice

Most substances increase in solubility as temperature increases, why don't HCl, NH3, and SO2?
They are insoluble in water
They are all non-polar
They are all polar
They are all gases
36
Multiple Choice

If you halve the amount of water you dissolve a solute in (so 50 g instead of 100 g of water), what do you do to the amount of solute to find the saturation point?
Halve it
Leave it alone
Double it
Quadruple it
37
Multiple Choice

If you have 250 g of water, how many grams of NH3 will dissolve in 40 oC water?
40 g
60 g
80 g
100 g
38
Multiple Choice

How many grams of KNO3 will fall out of solution if 145 g are dissolved in 60oC water?
40 g
35g
30 g
25 g
39
Multiple Choice

How many more grams of NaNO3 do you need to 85 g of the solute in 70 oC of water?
135 g
50 g
85 g
25 g
40
Multiple Choice
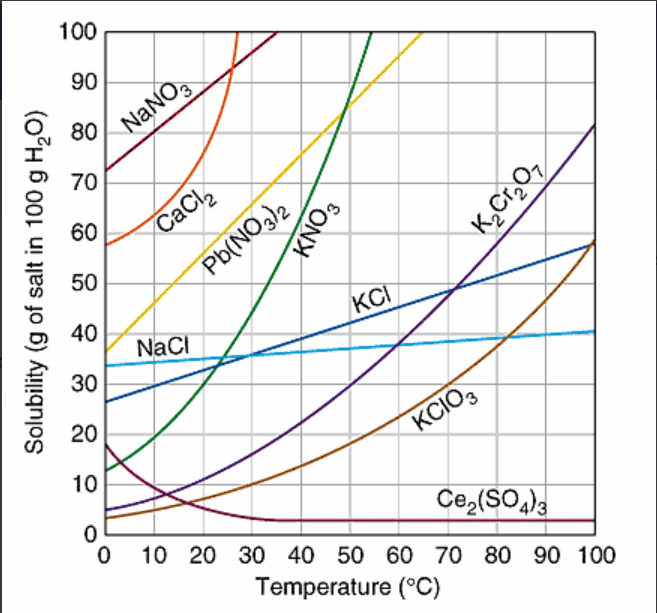
Which substance is MOST soluble at 0oC?
KI
NaNO3
NaCl
Ce2(SO4)3
41
Multiple Choice
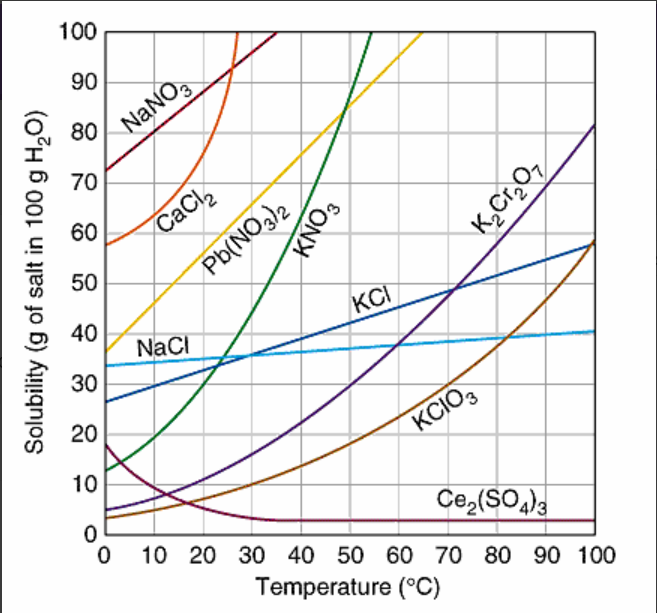
How many grams of K2Cr2O7, are soluble in 100 g of water at 95oC?
83 grams
75 grams
40 grams
12 grams
42
Multiple Choice

At approximately what temperature does the solubility of sodium chloride, NaCl, match the solubility of potassium dichromate, K2Cr2O7?
60oC
30oC
50oC
83oC
43
Multiple Choice
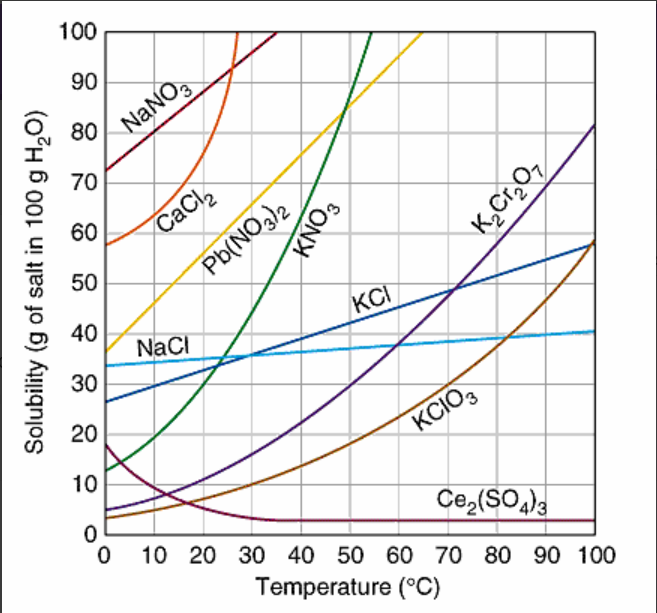
When 42 grams of potassium chloride, KCl, is dissolved in 100 grams of water at 50oC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
44
Multiple Choice
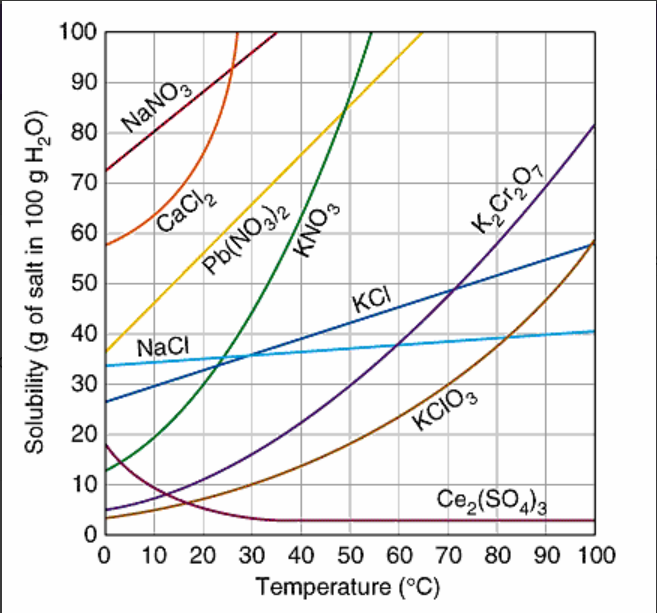
When 20 grams of potassium chlorate, KClO3, is dissolved in 100 g of water at 80oC, the solution can be correctly describe as:
supersaturated
saturated
unsaturated
45
Multiple Choice
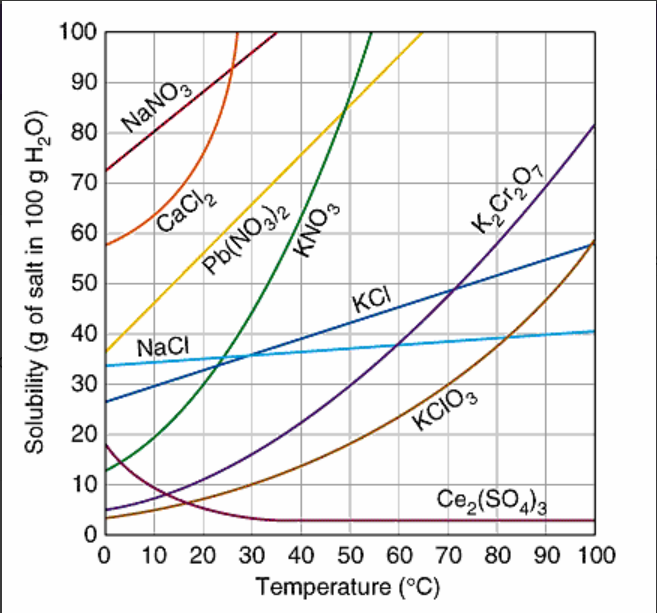
What temperature does the solubility of NaCl match KNO3?
50oC
40oC
35oC
25oC
46
Multiple Choice
47
Multiple Choice
Which of the following would result in being able to dissolve a greater amount of gas in a solution?
Lower the temperature of the solution.
Decrease the pressure of the solution.
Stir the solution.
Heat the solution.
48
Multiple Choice
You have some leftover soda after a party. Eventually, the soda will go flat. Which of the following options will slow the rate at which the CO2 gas escapes?
Stir the soda.
Store the soda bottle in the fridge.
Store the soda in your hot car.
Store the soda bottle in the room temperature cabinet.
49
Multiple Choice

Which solute is most likely a gas?
NH3
NaNO3
KCl
NaCl
50
Multiple Choice

Show answer
Auto Play
Slide 1 / 50
SLIDE
Similar Resources on Wayground

41 questions
Covalent Bonding
Presentation
•
8th - 12th Grade

47 questions
Unit 8- Lesson 4: Trade and Globalization
Presentation
•
9th - 12th Grade

42 questions
Chapter 13 Vocab Review - Thermochemistry
Presentation
•
10th - 12th Grade

46 questions
Orbital Notation
Presentation
•
10th - 12th Grade

47 questions
Review Chemical Reactions
Presentation
•
9th - 12th Grade

41 questions
Lewis Dot Structures
Presentation
•
9th - 12th Grade

44 questions
Introduction to Moles
Presentation
•
9th - 12th Grade

45 questions
(Full Lesson) Biology EOC Reporting Category 5 Review
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

19 questions
Review Set 3 - Moles & Chemical Formulas
Presentation
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Stoichiometry Practice
Quiz
•
10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade