

Chapter 15 new
Presentation
•
Science
•
10th Grade
•
Easy
+5
Standards-aligned
Patricia Malcolm
Used 7+ times
FREE Resource
136 Slides • 65 Questions
1

2

Have you ever seen something that looked so real that you had to touch it to see if it was really real?
Each material has its own properties.
The properties of materials can be used to classify them into general categories.
3

Matter is something that has mass and takes up space.
For example, you, candy, plants and planets are all matter.
Even some of the things that you can’t see, like air and other gasses are matter.
What is Matter
4

5

6

7

8

9

10

11

12

13

14

15

16

•Table salt is a chemical compound that fits this description. Even though it looks like white crystals and adds flavor to food, its components—sodium and chlorine—are neither white nor salty.
17

18

19

20

•Are pizza and a soft drink one of your favorite meals?
•If so, you enjoy two foods that are classified as MIXTURES.
•BUT two different kinds of mixtures
21

22

23

24

25

26

27

Most of the substances you come in contact with every day are heterogeneous mixtures. Some components are easy to see, like the ingredients in pizza, but others are not.
28

29

30

Remember that soft drink you had with your pizza?
Soft drinks contain water, sugar, flavoring, coloring and carbon dioxide gas.
Soft drinks in sealed bottles are examples of homogeneous mixtures
31

Another name for homogeneous mixtures like a cold soft drink is SOLUTION.
A SOLUTION is a homogeneous mixture of particles so small that they cannot be seen with a microscope and will never settle to the bottom of their container.
32

33


Paint is an example of a liquid with suspended colloid particles.
Fogconsists of particles of water suspended in air.
Smokecontains solids suspended in air.

34

35
This scattering of light by colloidal particles is called the Tyndall effect.
light beam is invisible as it passes through a solution, but can be seen readily as it passes through a colloid. This occurs because the particles in the colloid are large enough to scatter light, but those in the solution are not.

36

37

38

39
Multiple Choice

40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice

46
Multiple Choice
Which of the following is a colloid
smoke
oxygen
water
salt
47
Multiple Choice

How many elements are found on Earth?
5
10
30
90
48
Multiple Choice
A substance is ___________
always an element
always a compound
either an element or a compound
Mixture of compounds
49
Multiple Choice

What is water
Element
Compound
Heterogeneous
Homogeneous
50
Multiple Choice

What is pepperoni pizza
heterogeneous
homogeneous
51
Multiple Choice

what is air
element
compound
heterogeneous
homogeneous
52
Multiple Choice

Sand in Water
Solution
Suspension
Colloid
53
Multiple Choice

Kool-Aid
Solution
Suspension
Colloid
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
Compounds have properties different from the elements that make it up.
Agree
Disagree
57
Multiple Choice
Cannot be broken down into a simpler substance
Elements
Compounds
Both
58
Multiple Choice

This picture represents which of the following?
element
compound
59
Multiple Choice

This picture represents which of the following?
element
compound
60

•You can stretch a rubber band, but you can’t stretch a piece of string very much.
•You can bend a piece of wire, but you can’t easily bend a matchstick.
•In each case, the materials change shape, but the identity of the substances –rubber, string, wire and wood –does not change.
61

62

63

•How would you describe a tennis ball? You could begin by describing its shape, color, and state of matter.
•You can measure some physical properties, too. For instance, you could measure the diameter of the ball.
64

•How would you describe a soft drink in a cup?
•You could start with color.
•You could measure its volume and temperature.
•These are physical properties of the drink
65

66

67


68

69

70


71

•When a substance freezes, boils, evaporates, or condenses, it undergoes physical changes.
72

73

74

75

76

77

78


79
•Burning produces new substances during a chemical change.
•CHEMICAL PROPERTY - is a characteristic of a substance that indicates whether it can undergo a certain chemical change.

80

81

82

83

•A less dramatic chemical change can affect some medicines.
•Many medicine bottles are stored in dark bottles because they contain compounds that can change chemically if they are exposed to light.
84

85

86

87

88

89

90

91


92

93

94

95

96
Multiple Choice

When something burns?
Chemical change
Physical change
No change
97
Multiple Choice

When something melts?
No change
Physical change
Chemical change
98
Multiple Choice
When substances react with each other and new substances are formed?
Physical change
No change
Chemical change
99
Multiple Choice
When food spoils?
Physical change
Chemical change
No change
100
Multiple Choice

When something sparks or explodes?
Chemical change
Physical change
No change
101
Multiple Choice

When something is cooked or baked?
No change
Physical change
Chemical change
102
Multiple Choice
When metals rust due to oxidation?
Chemical change
Physical change
No change
103
Multiple Choice
When something bubbles or fizzes? (not the same as boiling)
Physical change
Chemical change
No change
104
Multiple Choice

When something is broken, cut, torn, or squeezed?
No change
Chemical change
Physical change
105
Multiple Choice

What is a physical property of these strawberries?
Red
Rotten
Flammable
106
Multiple Choice

What is a chemical property of paper?
Flammable
White
Light
107
Multiple Choice

Is toasting bread a physical or chemical change?
Physical
Chemical
108
Multiple Choice
109
Multiple Choice

110
Multiple Choice
When something is dissolved into a liquid?
Physical change
Chemical change
No change
111
Multiple Choice
112
Multiple Choice

Food Digestion
Physical change
Chemical change
113


114


115

116


117


118

119

120
Particles of solids are tightly packed, vibrating about a fixed position.
Solids have a definite shapeand a definite volume.
Solid

121


122


123

124


125

126


127


128


129


130


131


132


133


134


135


136

137


138


139

140

141

142

143

144

145

146

147

148

149

150

A solution is a mixture that has the same composition, color, density, and even taste throughout.
151

•To describe a solution, you may say that one substance is dissolved in another.
Solutes and Solvents
152
•When a solid dissolves in a liquid, the solid is the solute and the liquid is the solvent.
•The substance being dissolved is the solute, and the substance doing the dissolving is the solvent.
153

•In salt water, salt is the solute and water is the solvent.
•In carbonated soft drinks, carbon dioxide gas is one of the solutes and water is the solvent.
•When a liquid dissolves in another liquid the substance present in the larger amount is usually called the solvent.
154

155

156

157

158

159

160

161

162

163

164

165

166

167

168

169

170

171

172

173

174

175
Multiple Choice
The amount of heat needed to change 1 gram of a substance from the liquid phase to the gas phase is ___
Heat of Vaporization
Boiling point
Heat Energy
Phase change
176
Multiple Choice
A solid
has particles that are far apart
has a size and shape of its own
takes the shape of its container
177
Multiple Choice
All _______ can move and flow.
liquids
solids
gases
178
Multiple Choice
In general, the particles of a liquid are __________ tightly packed than those in a solid.
more
less
179
Multiple Choice
The particles in a gas are ___________ apart than those of solids or liquids.
farther
never
180
Multiple Choice

The picture shows...
a solid
a liquid
a gas
181
Multiple Choice

The picture shows...
a solid
a liquid
a gas
182
Multiple Choice

The picture shows...
a solid
a liquid
a gas
183
Multiple Choice
The state of matter depends on its
volume
temperature
weight
mass
184
Multiple Choice
What must be done to change water from liquid to solid?
Raise the temperature.
Lower the temperature.
185
Multiple Choice
186
Multiple Choice
187
Multiple Choice
The Temperature when a liquid turns to a solid
freezing point
heat of fusion
thermal expansion
melting point
188
Multiple Choice
CHOOSE THE WRONG ANSWER
189
Multiple Choice
CHOOSE THE WRONG ANSWER
190
Multiple Choice
In a _______ the molecules are close together.
solid
liquid
gas
191
Multiple Choice

What phase of matter exists in part D of the graph?
Gas
Liquid
Solid
Plasma
192
Multiple Choice
a solution in which the maximum amount of solute has been dissolved in a solvent
saturated
unsaturated
supersaturated
193
Multiple Choice
The liquid in which the material is dissolved
solute
solution
solvent
solubility
194
Multiple Choice
The solid that is being dissolved
solute
solvent
solution
solubility
195
Multiple Choice
Anything that takes up space and has mass is
Matter
Volume
Liquid
Atom
196
Multiple Choice
the ability of a substance to dissolve in a solvent
solubility
conductivity
197
Multiple Choice
Temperature when a substance changes from a solid to a liquid
freezing point
heat of fusion
thermal expansion
melting point
198
Multiple Choice
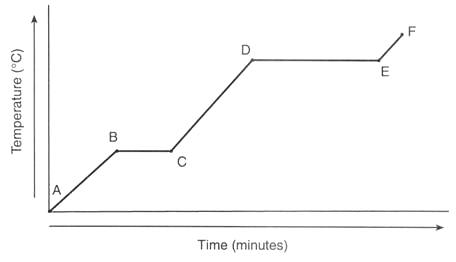
What is happening to the particles in the substance between points D and E?
Melting
Freezing
Vaporizing
Sublimating
199
Multiple Choice

The melting point of the sample is
-60 ºC
-100 ºC
60 ºC
100 ºC
200
Multiple Choice
Which factor affects solubility?
Temperature
State of Matter
201
Multiple Choice
To increase the solubility of a solid, what should you do to the temperature of the solvent ?
Increase the temperature of the solvent.
Decrease the temperature of the solvent.

Show answer
Auto Play
Slide 1 / 201
SLIDE
Similar Resources on Wayground

193 questions
LESSON 8: PRODUCING CLEANED-UP AND IN-BETWEENED DRAWINGS (CI)
Presentation
•
12th Grade

187 questions
OnRamps Whole Review
Presentation
•
9th - 12th Grade

202 questions
Bài giảng PLC S7-1200/S7-1500
Presentation
•
University

186 questions
untitled
Presentation
•
KG - University

199 questions
877891b73a
Presentation
•
KG - University

189 questions
3-1_多項式的基本概念與四則運算
Presentation
•
10th Grade

199 questions
SLAR Kindergarten EOY mClass/Lectura Daily Warm Up Day 1
Presentation
•
KG

201 questions
pemodelan bumi
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade