

Chemical Reactions Lesson One
Presentation
•
Science
•
6th - 8th Grade
•
Practice Problem
•
Medium
+3
Standards-aligned
Kerry Grumbles
Used 6+ times
FREE Resource
2 Slides • 19 Questions
1
Multiple Choice

Why is observing a color change important when identifying chemical reactions in everyday life, such as cooking?
It shows that a new substance may have formed.
It always means the food is cooked.
It makes food look more appealing.
It indicates the food is safe to eat.
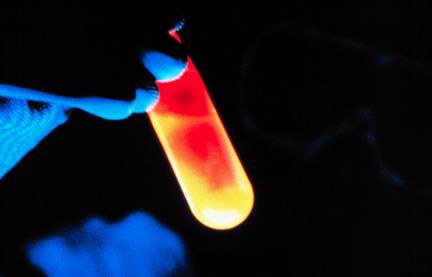
2
Multiple Choice
3
Open Ended

How does the production of gas during the making of pizza dough provide evidence for a chemical reaction?
4
Multiple Choice

5
Multiple Choice

6
Multiple Choice

Which of the following best describes the difference between exothermic and endothermic reactions as explained in the text?
Exothermic reactions absorb energy, while endothermic reactions release energy.
Exothermic reactions release energy, while endothermic reactions absorb energy.
Both exothermic and endothermic reactions absorb energy.
Both exothermic and endothermic reactions release energy.
7
Open Ended

Analyze the graphs of exothermic and endothermic reactions. What does the difference in the energy of reactants and products indicate about the direction of energy flow in each reaction?
8
Multiple Choice
Equal amounts of four different substances (A-D) were added separately to equal amounts of an acid, and a thermometer was placed in the mixture. For which substance is the reaction the least endothermic?
A. temperature falls by 3 °C
B. temperature rises by 5 °C
C. temperature falls by 5 °C
D. temperature rises by 3 °C
9
Multiple Choice
Equal amounts of four different substances (A-D) were added separately to equal amounts of an acid, and a thermometer was placed in the mixture. For which substance is the reaction the most exothermic?
A. temperature rises by 3 °C
B. temperature falls by 5 °C
C. temperature falls by 3 °C
D. temperature rises by 5 °C
10
Multiple Choice
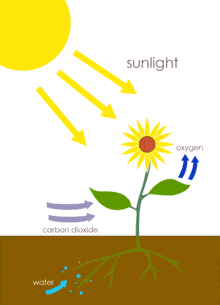
11
Multiple Select

Which of the following factors can affect the rate of a chemical reaction, as described in the text?
Surface area
Temperature
Concentration
All of the above
12
Multiple Choice

What is the effect of a catalyst on the activation energy of a chemical reaction, as shown in the graph?
It increases the activation energy needed for the reaction.
It decreases the activation energy needed for the reaction.
It has no effect on the activation energy.
It stops the reaction from occurring.
13
Fill in the Blanks

14

15
Multiple Choice

16
Multiple Choice
What is the primary reason that endothermic reactions require an input of energy?
To break bonds in the reactants
To form new bonds in the products
To increase the reaction rate
To decrease the reaction rate
17
Multiple Choice

18

19
Open Ended
How are chemical reactions used in art, and what are some examples of their application as described in the text?
20
Open Ended
Reflecting on what you learned about chemical reactions in the kitchen, how might you use this knowledge to identify chemical changes in other everyday activities?
21
Multiple Choice
What are some observable signs that a chemical reaction has occurred when making pizza?
Color change, gas production, and formation of a precipitate
Change in temperature only
Change in shape only
No observable signs

Why is observing a color change important when identifying chemical reactions in everyday life, such as cooking?
It shows that a new substance may have formed.
It always means the food is cooked.
It makes food look more appealing.
It indicates the food is safe to eat.
Show answer
Auto Play
Slide 1 / 21
MULTIPLE CHOICE
Similar Resources on Wayground

15 questions
Types of Cells
Presentation
•
6th - 8th Grade

14 questions
Kinetic Energy
Presentation
•
6th - 8th Grade

18 questions
Solar System
Presentation
•
6th - 8th Grade

16 questions
Fossils
Presentation
•
6th - 8th Grade

15 questions
Repetition vs Replication
Presentation
•
6th - 8th Grade

15 questions
Genetics
Presentation
•
6th - 8th Grade

17 questions
ECOLOGY PART 2
Presentation
•
6th - 8th Grade

17 questions
Engineering Design Process
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade