

asam basa
Presentation
•
Chemistry
•
11th Grade
•
Hard
Anggun Putri Thalia
FREE Resource
9 Slides • 8 Questions
1

2
Multiple Choice
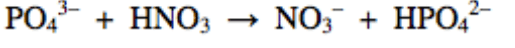
What is the Bronsted-Lowry base in the following equation?
PO43-
HNO3
NO3-
HPO42-
3

4
Multiple Choice
According to Arrhenius acid-base theory, an acid ___.
is an H+ acceptor
is a H+ donor
is a substance that produces H+ in water
turns litmus paper blue
5

6
Multiple Choice
What is the role of an acid and a base according to Lewis theory?
Acid accepts electron pairs, Base donates electron pairs
Acid donates electron pairs, Base accepts electron pairs
Acid and Base both accept electron pairs
Acid and Base both donate electron pairs
7

8
Multiple Choice
What is the equilibrium constant (K) for the ionization of water as shown in the image?
Kw = [H+][OH-]
Kw = [H2O]
Kw = [H+]/[OH-]
Kw = [H+][OH-]/[H2O]
9

10
Multiple Choice
What is the formula to calculate the concentration of strong acids?
[H+] = Ma × a
[H+] = √Ka × Ma
[H+] = α × Ma
[OH-] = Mb × b
11

12
Open Ended
Explain the relationship between pH and pOH based on the information provided.
13

14

15

16
Multiple Choice

Examine the results (color change) of the pH paper shown in the picture. What type of substance was tested?
an acid
a base
a neutral substance
none of the above
17
Poll
How confident do you feel about this topic now?

Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

11 questions
HIDROKARBON part 2
Presentation
•
11th Grade

12 questions
Intro Into Chemistry
Presentation
•
11th Grade

11 questions
Taller sumativo - Retos QuímicoMatemáticos
Presentation
•
11th Grade

11 questions
Compuestos orgánicos
Presentation
•
11th Grade

14 questions
PROFUNDIZACIÓN PRUEBA SABER GRADO 10 - 17-09-21
Presentation
•
11th Grade

13 questions
TRANSCRIPTION
Presentation
•
11th Grade

11 questions
SEnyawa Hidrokarbon
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade