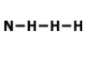Lewis structures
Authored by Sara Turner
Science, Chemistry
10th Grade
NGSS covered
Used 491+ times

AI Actions
Add similar questions
Adjust reading levels
Convert to real-world scenario
Translate activity
More...
Content View
Student View
12 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
According to the octet rule most elements need _______ valence electrons.
Tags
NGSS.HS-PS1-1
NGSS.HS-PS1-2
2.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
Hydrogen being an exception to the octet rule, needs _____ electrons in its outer energy level to be stable.
4
6
8
2
Tags
NGSS.HS-PS1-1
3.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
Three pairs of electrons are shared in a
Tags
NGSS.HS-PS1-4
4.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
A covalent bond is
a bond that shares electrons metallicaly
A bond that shares electrons between non metals
Metalloids bonding
metals and nonmetals bonding
Tags
NGSS.HS-PS1-2
5.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt

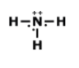
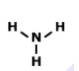
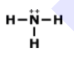
Draw the lewis dot structure for F2. Which of the following is the correct Lewis dot structure for the molecule F2?
A
B
C
D
Tags
NGSS.HS-PS1-1
6.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
How many electrons are shared in each bonding line?
1
2
3
4
Tags
NGSS.HS-PS1-2
7.
MULTIPLE CHOICE QUESTION
15 mins • 1 pt
How many valence electrons does phosphorus have?
15
4
5
31
Tags
NGSS.HS-PS1-1
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?