
Nitrous Oxide Lewis Structure Concepts
Interactive Video
•
Chemistry, Science, Biology
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a key reason for exceptions to the octet rule in elements from the third or fourth period?
They are less reactive.
They are more electronegative.
They have fewer than eight valence electrons.
They have more than eight valence electrons.
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
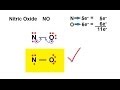
Why is nitrous oxide unable to satisfy the octet rule?
It has an odd number of valence electrons.
It has an even number of valence electrons.
It is too stable.
It is too reactive.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of valence electrons in nitrous oxide?
Ten
Thirteen
Twelve
Eleven
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the initial Lewis structure attempt for nitrous oxide, how many valence electrons are around the nitrogen atom?
Four
Six
Five
Seven
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the role of electronegativity in determining the central atom in a Lewis structure?
The most electronegative atom is central.
The least electronegative atom is central.
The central atom is always oxygen.
Electronegativity does not affect the central atom.
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens when a double bond is introduced in the Lewis structure of nitrous oxide?
Both atoms follow the octet rule.
Oxygen follows the octet rule.
Neither atom follows the octet rule.
Nitrogen follows the octet rule.
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which atom in nitrous oxide is more electronegative?
Nitrogen
Oxygen
Both are equally electronegative
Neither is electronegative
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
