
Understanding Molar Mass and Conversions
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is Avogadro's number used for in chemistry?
To determine volume
To calculate speed
To count particles in a mole
To measure temperature
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
When solving a two-step problem, what is the first step if you are given mass?
Convert mass to volume
Use Avogadro's number directly
Calculate temperature
Convert mass to moles using molar mass
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
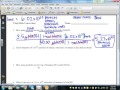
In the problem involving sodium hydroxide, what is the molar mass used?
20.00 g/mol
40.00 g/mol
80.00 g/mol
60.00 g/mol
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to use parentheses when entering scientific notation into a calculator?
To make the number look neat
To decrease the number's value
To increase the number's value
To ensure the correct order of operations
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the first step in solving the problem involving ethane?
Convert particles to moles using Avogadro's number
Convert moles to particles
Calculate the temperature
Determine the volume
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molar mass of ethane (C2H6) used in the problem?
50.08 g/mol
40.08 g/mol
30.08 g/mol
60.08 g/mol
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the final unit you are looking for in the ethane problem?
Moles
Particles
Grams
Liters
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
