
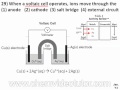
Reactivity and Function of Voltaic Cells
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard
Patricia Brown
FREE Resource
Read more
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is a voltaic cell commonly compared to?
A light bulb
A homemade battery
A wind turbine
A solar panel
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which two metals are mentioned as having different reactivity levels in a voltaic cell?
Aluminum and tin
Iron and zinc
Copper and silver
Gold and platinum
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
According to the chemistry reference table, which metal is more reactive?
Silver
Copper
Platinum
Gold
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the chemical reaction of a voltaic cell, what does solid copper (Cu) become?
Cu negative ions
Liquid copper
Cu positive ions
Solid silver
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What happens to silver ions (Ag+) in the voltaic cell reaction?
They gain electrons
They dissolve in water
They form a gas
They lose electrons
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Where do electrons travel from and to in a voltaic cell?
From gold to platinum
From silver to copper
From copper to silver
From zinc to iron
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the result of silver ions gaining electrons in the voltaic cell?
Formation of gaseous silver
Decrease in mass of the copper electrode
Increase in mass of the silver electrode
Formation of liquid silver
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
