
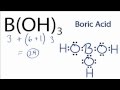
B(OH)3 Lewis Structure Concepts
Interactive Video
•
Chemistry
•
9th - 10th Grade
•
Practice Problem
•
Hard
Emma Peterson
FREE Resource
Read more
5 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons are there in total for the B(OH)3 Lewis structure?
18
21
24
27
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
In the B(OH)3 Lewis structure, which atom is placed at the center?
Boron
Oxygen
Hydrogen
Carbon
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How many valence electrons are used to form chemical bonds in the B(OH)3 structure?
12
24
6
18
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the total number of valence electrons used to fill the octets for the Oxygens in B(OH)3?
6
12
24
18
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is Boron an exception in the B(OH)3 Lewis structure?
It does not form any bonds.
It forms double bonds with Oxygen.
It is stable with only six valence electrons.
It has more than eight valence electrons.
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
