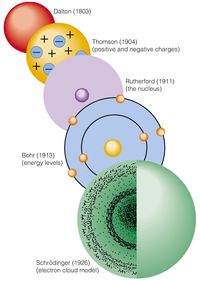
Unit 2 Atomic Theory Part 2 Test Review
Flashcard
•
Science
•
10th Grade
•
Practice Problem
•
Hard

Wayground Content
FREE Resource
Student preview

15 questions
Show all answers
1.
FLASHCARD QUESTION
Front
What model of the atom consists of protons and neutrons in a central nucleus, with electrons occupying discrete energy levels?
Back
Bohr Model
2.
FLASHCARD QUESTION
Front
What did Bohr add to the model of the atom?
Back
That atoms have orbiting electrons that can jump energy levels.
3.
FLASHCARD QUESTION
Front
Which area of a Bohr Model shows an electron occupying the lowest energy level?
Back
First shell
4.
FLASHCARD QUESTION
Front
What is the electron configuration of Phosphorus (P)?
Back
1s²2s²2p⁶3s²3p³
5.
FLASHCARD QUESTION
Front
What is the energy of a single photon of light with a wavelength of 657nm?
Back
3.03 x 10^{-19} J
6.
FLASHCARD QUESTION
Front
Define the term 'atomic theory'.
Back
A scientific theory that explains the nature of matter by stating that matter is composed of discrete units called atoms.
7.
FLASHCARD QUESTION
Front
What is the charge of a proton?
Back
Positive (+1)
Create a free account and access millions of resources
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
Similar Resources on Wayground

13 questions
Simple Tips for Healthy Eating
Flashcard
•
10th Grade

16 questions
Phrasal Verbs Related to Food & Eating
Flashcard
•
10th Grade

15 questions
Evolution Test Review
Flashcard
•
9th Grade

15 questions
Plant Structure and Function Review
Flashcard
•
9th - 12th Grade

8 questions
Understanding Heat Radiation
Flashcard
•
KG

9 questions
Parts of a Computer
Flashcard
•
9th - 12th Grade
![[AP CSP] Binary Conversion](https://cf.quizizz.com/image/image-loader.svg)
13 questions
[AP CSP] Binary Conversion
Flashcard
•
9th - 12th Grade

10 questions
Output Devices
Flashcard
•
10th Grade
Popular Resources on Wayground

5 questions
This is not a...winter edition (Drawing game)
Quiz
•
1st - 5th Grade

15 questions
4:3 Model Multiplication of Decimals by Whole Numbers
Quiz
•
5th Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

10 questions
The Best Christmas Pageant Ever Chapters 1 & 2
Quiz
•
4th Grade

12 questions
Unit 4 Review Day
Quiz
•
3rd Grade

10 questions
Identify Iconic Christmas Movie Scenes
Interactive video
•
6th - 10th Grade

20 questions
Christmas Trivia
Quiz
•
6th - 8th Grade

18 questions
Kids Christmas Trivia
Quiz
•
KG - 5th Grade
Discover more resources for Science

10 questions
Exploring the Energy Cycle: Photosynthesis and Cellular Respiration
Interactive video
•
6th - 10th Grade

10 questions
Exploring Plate Tectonics and Their Boundaries
Interactive video
•
6th - 10th Grade

10 questions
Exploring Light and Waves Concepts
Interactive video
•
6th - 10th Grade

10 questions
EOC Domain 3: Taxonomy
Lesson
•
9th - 10th Grade

9 questions
EOC Domain 4: Evolution
Lesson
•
9th - 10th Grade

10 questions
EOC Domain 5: Ecology
Lesson
•
9th - 10th Grade

11 questions
EOC Domain 2: Genetics
Lesson
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade