

atom theories
Presentation
•
Science
•
8th - 9th Grade
•
Medium
Zafer Yildiz
Used 13+ times
FREE Resource
21 Slides • 54 Questions
1
atom theories

2

3

4

5

6

7

8

9
Multiple Choice
Which subatomic particles are found in the nucleus of an atom?
Protons and Electrons
Protons and Neutrons
Neutrons and Electrons
Protons, Neutrons and Electrons
10

11

12
Multiple Choice

which of the below choices correctly labels the atom
A: electron
B: proton
C: neutron
D: nucleus
A: proton
B: electron
C: nucleus
D: neutron
A: electron
B: neutron
C: proton
D: nucleus
A: nucleus
B: electron
C: neutron
D: proton
13
Multiple Choice
which set of values correctly gives the charges of sub-atomic particles
electron: +
proton: -
neutron: no charge
electron: +
proton: no charge
neutron: -
electron: -
proton: no charge
neutron: +
electron: -
proton: +
neutron: no charge
14
Multiple Choice
which of the below gives the right masses of the sub-atomic particles
electron: 1
proton: 1
neutron: no mass
electron: no mass
proton: 1
neutron: 1
electron: 1
proton: no mass
neutron: no mass
electron: no mass
proton: 1
neutron: no mass
15
Multiple Choice
which of the below is a definition of an atom
Make up part of an atom and
have a negative charge.
Found in the nucleus of an
atom, these do not have
a charge
The smallest part of an
element, made up of protons,
neutrons and electrons.
16
Multiple Choice
which one of the below defines neutron
Found in the nucleus of an
atom, these do not have
a charge.
This refers to the number of
protons an element has.
Found in the nucleus of an
atom, they have a
positive charge.
The smallest part of an
element, made up of nucleons and electrons
17
Multiple Choice
which of the below defines atomic number
This refers to the number of
protons an atom has.
Found in the nucleus of an
atom, they have a
positive charge.
The smallest part of an
element, made up of protons,
neutrons and electrons.
18
Multiple Choice
which of the below defines proton
Found in the nucleus of an
atom, these do not have
a charge.
Found in the nucleus of an
atom, they have a
positive charge.
revolves around nucleus and has a negative charge
19
Multiple Choice
which one of the below defines electron
revolves around nucleus and has a negative charge
refers to the number of
protons an element has.
Found in the nucleus of an
atom, they have a
positive charge.
Found in the nucleus of an
atom, these do not have
a charge.
20
Multiple Choice

which number in this table is -1
A
B
C
D
21
Multiple Choice
22
Multiple Choice
In an atom, the number of protons is equal to the number of ____________.
energy levels
neutrons
neurons
electrons
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
What mass does a neutron have?
1 amu
0 amu
-1 amu
+2 amu
27
Multiple Choice
Which subatomic particles are found in the nucleus of an atom?
Protons and Electrons
Protons and Neutrons
Neutrons and Electrons
Protons, Neutrons and Electrons
28

29
Multiple Choice
30
Multiple Choice
Atom comes from the word atomos, meaning
microscopic
elemental
indivisible
molecular
31

32
Multiple Choice

This was the first model of the atom ever proposed. It was simple and
described atoms as tiny spheres that could not be broken down into
smaller pieces.
a) Democritus's model of the atom
b) The "Plum Pudding Model" of the atom
c) The "Rutherford Model" of the atom
d) The "Quantum Mechanical Model" of the atom
33
Multiple Choice
Democritus and Dalton did NOT believe that
atoms are indivisible and indestructible
all matter is made of atoms
atoms can combine to form compounds
the atom is made up of smaller particles
34
Multiple Choice
Democritus and Dalton did NOT believe that
atoms are indivisible and indestructible
all matter is made of atoms
atoms can combine to form compounds
the atom is made up of smaller particles
35
Multiple Choice

36
Multiple Choice
Which of the following is NOT a part of Dalton's atomic theory?
All matter is composed of atoms and empty space
Atoms are always in motion.
Atoms of the same element are identical.
Atoms can't be destroyed...merely re-arranged.
37
Multiple Choice
Which of the following is NOT a part of Dalton's atomic theory?
All matter is composed of atoms and empty space
Atoms are always in motion.
Atoms of the same element are identical.
Atoms can't be destroyed...merely re-arranged.
38
Multiple Choice
John Dalton stated:
a) elements are made of atoms
b) atoms of a given element are identical
c) atoms cannot be subdivided, created, nor destroyed
d) all of the above
39
40

41
Multiple Choice
Discovered the electron within the atom
Thomson
Chadwick
Schrodinger & Heisenberg
Dalton
42
Multiple Choice
What did the Plum pudding model suggest about the
structure of atoms?
a) That neutrons and electrons were arranged in a sphere
b) that is was a sphere of positive charge throughout which the
electrons were distributed
c) That an atom was filled with neutrons, protons and a sphere
of negative charge.
d) That neutrons, protons and electrons were distributed
throughout a sphere.
43
Multiple Choice

This model was developed after J.J. Thompson discovered
electrons, a particle smaller than an atom. This shows electrons floating freely in a positive space
a) The "Plum Pudding Model" of the atom
b) The "Rutherford Model" of the atom
c) Democritus's model of the atom
d) The "Quantum Mechanical Model" of the atom
44
Multiple Choice
Discovered the electron using cathode ray tube
a) Joseph Thomson
b) John Dalton
c) Ernest Rutherford
d) Robert Millikan
45
Multiple Choice

J.J. Thomson's experimental setup can be seen in the image. What is happening to the cathode rays?
they are attracted to the negative plate
they are attracted to the positive plate
they are stopped by the paltes
they are unattracted to either plate
46

47

48
49

50
Multiple Choice
The scientist responsible for "discovering" the nucleus is:
bohr
rutherford
shrodinger
einstein
51
Multiple Choice
The nucleus of an atom can be described as:
a) spacious and negatively charged
b) dense and positively charged
c) spacious and positively charged
d) dense and negatively charged
52
Multiple Choice
How did Rutherford discover the proton?
a) Cathode tube ray experiment
b) Gold Foil Experiment
c) Planetary Model
d) Plum Pudding Model
53
Multiple Choice
What particle was used in the gold foil experiment?
beta particle
alpha particle
gamma ray
54
Multiple Choice
Most of the alpha particles went straight through the gold
foil. This suggested that....
a) There is no interference between alpha and gold.
b) Most of the atom is made up of empty space.
c) The alpha particles were very tiny.
d) None of the above
55
Multiple Choice
Some of the alpha particles fired at the gold foil were
deflected at angles. This was due to...
a) reflection of the alpha source when it it the fluorescent screen.
b) The alpha particles hitting the gold nucleus.
c) Repulsion forces between the positively charged nucleus and
the positively charged alpha particles.
d) A mis-firing of the alpha source
56
Multiple Choice
Rutherford’s gold foil scattering experiment indicated that
a) the nucleus of an atom occupies most of an atom’s volume
b) positive charges are dispersed throughout the atom.
c) positive charges are concentrated in a very small core at the
atom’s center
d) protons and neutrons are located around the nucleus.
57
Multiple Choice
His atomic model included a dense positive core in an atom,
surrounded by negatively charged particles
joseph thomson
niels bohr
ernst rutherford
58
Multiple Choice
How did Rutherford develop his atomic theory?
Testing gamma rays against dogs
Testing alpha rays of radiation against gold foil
Testing zinc against silver
Testing zinc against gold
59
Multiple Choice
Which scientist determined that the mass of electrons is negligible compared to the mass of the atom?
Thomson
Rutherford
Bohr
Dalton
60
Multiple Choice
Who discovered the nucleus of the atom?
Dalton
Thomson
Rutherford
Bohr
61
Multiple Choice
Why were Rutherford's students surprised by the results of the gold foil experiment?
they didn't expect the alpha particles to bounce back from the foil
they didn't expect the alpha particles to continue in a straight path
they expected only a few alpha particles to bound back from the foil
they expected the alpha particles to be deflected by electrons
62
Multiple Choice
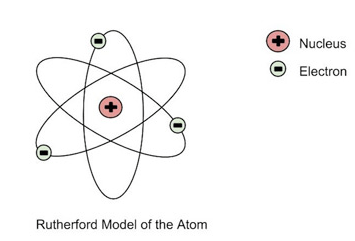
Rutherford's gold foil experiment provided evidence that...
negative and positive charges are spread evenly throughout the atom.
alpha particles have a positive charge.
gold is not a dense as previously thought.
there is a dense positively charged nucleus at the center of an atom.
63
Multiple Choice

Rutherford's gold foil experiment provided evidence that...
negative and positive charges are spread evenly throughout the atom.
alpha particles have a positive charge.
gold is not a dense as previously thought.
there is a dense positively charged nucleus at the center of an atom.
64

65

66

67
Multiple Choice

68
Multiple Choice
Which scientist proposed a model of the atom in which the electrons are orbiting at different energy levels?
Niels Bohr
James Chadwick
John Dalton
Ernest Rutherford
69
Multiple Choice
How is Bohr's atomic model different from Rutherford's model?
Bohr's model has a nucleus
Bohr's model has electrons
electrons in Bohr's model are located farther from the nucleus
electrons in Bohr's model are located in circular energy levels
70
Multiple Choice
Based on his research of the atom, James Chadwick concluded that
a cathode ray has a negative charge
the electrons are found in an electron cloud
the nucleus contains neutrons as well as protons
the mass of an atom is concentrated in its center
71
Multiple Choice
Whose atomic model is the most widely accepted model today?
Rutherford's Atomic Model
Dalton's Billiard Ball Model
J.J Thompson's Plum Pudding Model
Bohr's Model and Schrodinger's Quantum Atomic Models
72
Multiple Choice
which particle is responsible for the volume of the atom
proton
electron
neutron
notrino
73
Multiple Select
which particles are responsible for the mass of the atom
proton
electron
neutron
boson
74
Multiple Choice
Which metal element is used in the Rutherford experiment as
foil?
silver
iron
gold
copper
75
Multiple Choice
why rutherford chose gold metal as a foil
it is elegant
it is extremely heavy
it is shiny
it can be made extremely thin
atom theories

Show answer
Auto Play
Slide 1 / 75
SLIDE
Similar Resources on Wayground

69 questions
2-20-25 types of bonding slides 1-22
Presentation
•
8th Grade

70 questions
Chemical Reaction Lessons
Presentation
•
8th Grade

69 questions
Re-introduction to Geography
Presentation
•
9th Grade

68 questions
Review for US Constitution Test
Presentation
•
8th Grade

74 questions
4.2 Ionic Bonding Notes
Presentation
•
9th Grade

66 questions
Forces
Presentation
•
8th Grade

70 questions
10.3 Stem & Leaf Diagrams review lesson + Quiz
Presentation
•
8th Grade

70 questions
2/24 - 3rd 9 Weeks Review
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade