

2/24 - 3rd 9 Weeks Review
Presentation
•
Science
•
8th Grade
•
Easy
Rebecca Keith
Used 5+ times
FREE Resource
12 Slides • 58 Questions
1

3rd 9 Weeks Review
Chemistry
Earth and Space
2

3
Multiple Choice
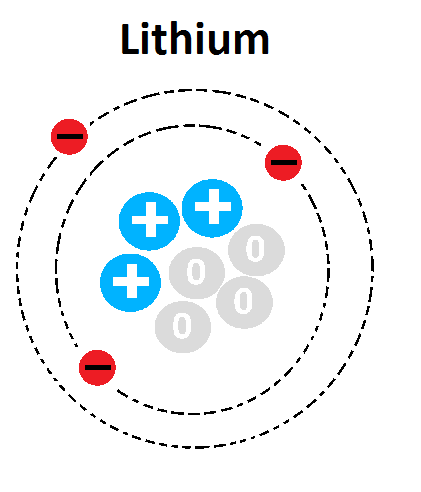
What is the atomic number of this atom?
1
3
4
7
4
Multiple Choice
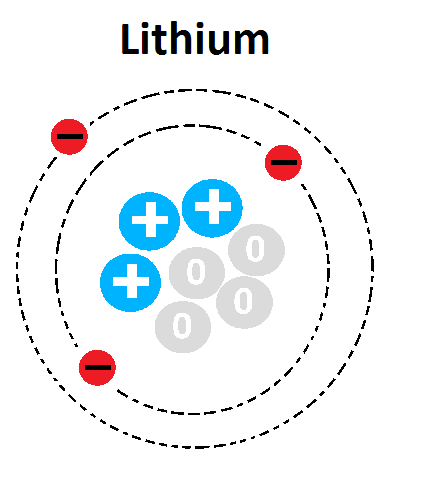
What is the mass of this atom?
1
3
4
7
5
Multiple Choice

:What is letter “A.”pointing to?
Proton
Neutron
Electron
6
Multiple Choice

What is letter “B” pointing to?
Neutron
Proton
Electron
7
Multiple Choice

What is letter “C”. pointing to?
Neutron
Proton
Electron
8
Multiple Choice

How many protons does Lithium have?
1
3
4
7
9

10
Multiple Choice

How many protons does platinum have?
78
117
195
11
Multiple Choice

How many neutrons are in this element?
26
29
30
55.845
12
Multiple Choice

How many electrons does this element have?
25
26
29
30
13

14
Multiple Select

Select the elements that have 8 valence electrons. (more than one answer)
Ne
Br
Kr
Xe
15
Multiple Choice

How many valence electrons are in this atom?
2
3
5
6
16
Multiple Choice

What is the reactivity of Argon?
High
Low
Stable / Non-reactive
17
Multiple Choice

Based on the drawings, which one is the least reactive?
Chlorine, because it has three energy levels with seven electrons in the third level
Gallium, because it has four energy levels with three electrons in the fourth level
Neon, because it has two energy levels with eight electrons in the second level
Tin, because it has five energy levels with four electrons in the fifth level
18
Multiple Choice

What is the reactivity of Group 18
High
Low
Stable / Non-reactive
19
Multiple Choice
An atom has 18 protons and 8 valence electrons. Which statement would best identify this element?
The element is "happy" or stable because its outer energy level is filled.
The element has an atomic mass of 18.
The element is highly reactive with eight electrons in its outer shell.
The element is a metal.
20
Multiple Choice
Size of its nucleus
Number of total electrons
Number of valence electrons
Number of shells
21

22
Multiple Choice
1
12
6
23
Multiple Select
What elements are in an amino acid with the formula C5H11NO2? (select all that apply)
carbon
hydrogen
nitrogen
nobelium
oxygen
24
Multiple Choice
4
12
7
24
25
Multiple Select
Which elements are in the chemical formula CH3CO2? (select all that apply)
carbon
hydrogen
cobalt
oxygen
26
Multiple Choice
Identify elements in the following compound: NaCl
Sodium, Carbon
Nitrogen, Chlorine
Sodium, Chlorine
27
Multiple Choice
The formula for baking soda is given.
NaHCO3
This formula indicates that the most basic unit of baking soda contains -
1 hydrogen atom, 1 sodium atom, 3 carbon atoms, and 3 oxygen atoms
1 sodium atom, 1 hydrogen atom, 1 carbon atom, and 3 oxygen atoms
1 potassium atom, 1 helium atom, 1 carbon atom, and 1 oxygen atom
1 carbon atom, 1 helium atom, 1 calcium atom, and 3 oxygen atoms
28

29
Multiple Choice
Bubbles form around a potato in a container of hydrogen peroxide. Bubbles indicate the formation of a new substance because bubbles -
could show that a gas is forming
always rise to the top of a liquid.
are almost always perfectly spherical.
usually burst after a few minutes.
30
Multiple Choice

In which experiment are new substances being formed?
Experiment 1
Experiment 2
Experiment 3
Experiment 4
31
Multiple Choice
Which observation is evidence that a chemical reaction has occurred?
The mass of the solution remains the same.
Phase changes are observed.
The color of the solution remains clear.
The temperature of the solution increases.
32
Multiple Choice
A sample of zinc is placed in water and it settles to the bottom
A solid block of ice is heated and it completely melts into a liquid.
Sugar that is burned gives off an odor and turns brown and then black.
A tomato is placed into a blender and chopped up into smaller pieces.
33
Multiple Choice
a new substance has formed
a solid dissolved in a liquid
interaction between 2 gases
a change in the state of matter
34
Multiple Choice
A student observes some sugar as it is heated and burns. The student concludes that a chemical reaction has occurred. Which of the following observations about the burning sugar provides evidence of a chemical reaction?
Heat is added to the sugar crystals.
The sugar melts and becomes a liquid.
Gas is produced as the sugar turns black and bubbles
35
Multiple Choice

The student inferred that a chemical reaction occurred. What evidence supports this inference?
A dark solid formed on the zinc metal.
The zinc metal remained silver-colored and shiny
The CuSO4 solution turned blue when the zinc metal was added.
None of these
36

37
Multiple Choice
Which of the following is a good conductor of heat, malleable, ductile, and shiny?
Metal
Nonmetal
Metalloid
38
Multiple Choice

Titanium
Selenium
Potassium
Polonium
39

40
Multiple Choice
Which two elements are in the same period?
Boron and Carbon
Potassium and Sodium
Lithium and Magnesium
Zinc and Nitrogen
41
Multiple Choice

Period 1, Group 18
Hydrodgen
Sodium
Flourine
Helium
42
Multiple Choice

Each diagram above represents an atom of a different element. Based on the diagrams, which two elements would be in the same period?
Elements 2 and 3
Elements 3 and 4
Elements 1 and 3
Elements 1 and 4
43
Multiple Choice
Which of the following pairs of elements is in the same period?
Li and Mg
N and P
B and F
Si and As
44
Multiple Choice

what period would this element be in?
6
8
3
16
45
Multiple Choice

Which two elements on the periodic table are in the same period?
Se and Te
Sn and Rb
K and Ba
F and Cl
46

47
Multiple Choice

seasons
moon phases
day and night
weather
48
Multiple Choice

Earth's tilt & rotation
Earth's tilt & revolution
Earth's rotation & rovolution
Earth's tilt & resolution
49
Multiple Choice

Winter
Spring
Summer
Fall
50
Multiple Choice

Which season is taking place in the Southern Hemisphere in Position 4?
Winter
Spring
Summer
Fall
51

52

53
Multiple Choice

One Day
One Week
Two Weeks
One Month
54
Multiple Choice

New moon
Full moon
55
Multiple Choice

Two friends want to go for a night hike when there is a full moon in July. If there is a third-quarter moon on July 2, what is the approximate date of the next full moon?
July 9
July 16
July 23
July 30
56
Multiple Choice

What moon phase is at position 2?
Waxing Gibbous
Waning Gibbous
Waxing Crescent
Waning Crescent
57
Multiple Choice
A student viewed the moon through binoculars one week after a new moon. Which image shows the phase of the moon that the student observed?
58
Multiple Choice
A student models moon phases. Which numbered diagram represents the student modeling a full moon?
59
Multiple Choice

A student shows a model of Earth and the moon to class. Which picture shows how the moon appears from Earth when it is in the position shown in this model?
60
Multiple Choice

Use the calendar to predict the moon phase on March 9th.
New Moon
Full Moon
Third Quarter
First Quarter
61
Multiple Choice

What moon phase is shown in the picture?
Waning Gibbous
Waning Crescent
Waxing Gibbous
Waxing Crescent
62
Multiple Choice

What moon phase is shown in the picture?
Waxing Crescent
Waxing Gibbous
Waning Gibbous
Waning Crescent
63
Multiple Choice

What moon phase is shown in the picture?
Waxing Crescent
Waxing Gibbous
Waning Gibbous
Waning Crescent
64
Multiple Choice

What moon phase is shown in the picture?
Waning Crescent
Waning Crescent
Waning Gibbous
Waxing Crescent
65

66
Multiple Choice

What type of tides would this arrangement of earth-moon-sun create?
neap tide
spring tide
new moon tide
full moon tide
67
Multiple Choice

What type of tide would this arrangement of earth-moon-sun create?
full moon tide
new moon tide
neap tide
spring tide
68
Multiple Choice
Which diagram represents the alignment of the Earth, Moon and Sun during a spring tide?
69
Multiple Choice
Which model shows the positions of the sun, moon and Earth that have the greatest effect on ocean tides?
70
Multiple Choice

The diagram models four lunar phases. During which lunar phase is the tide the highest?
1
2
3
4

3rd 9 Weeks Review
Chemistry
Earth and Space
Show answer
Auto Play
Slide 1 / 70
SLIDE
Similar Resources on Wayground

65 questions
Friday 3/15: Intro to Natural Selection
Presentation
•
8th Grade

67 questions
Physical and Chemical Change
Presentation
•
8th Grade

67 questions
EID 2.3 Inherited Traits
Presentation
•
8th Grade

66 questions
unit (1) 4 exploration 2024
Presentation
•
8th Grade

63 questions
Carew-7.7 D Newton's 1st Law, Balanced & Unbalanced Forces Notes
Presentation
•
8th Grade

61 questions
Climate Zones
Presentation
•
7th Grade

68 questions
Chemistry Review
Presentation
•
8th Grade

65 questions
Motion Graphs, practice 2024
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade