

Gases, Liquids, and Solids - Section 3
Presentation
•
Chemistry, Science
•
10th - 12th Grade
•
Medium
+1
Standards-aligned
Andrew Baldwin
Used 15+ times
FREE Resource
11 Slides • 24 Questions
1
Gases, Liquids, and Solids - Section 3

2

3
Poll

Where are you on the scale?
1
2
3
4
4

5
6

7

8

9

10
Multiple Choice
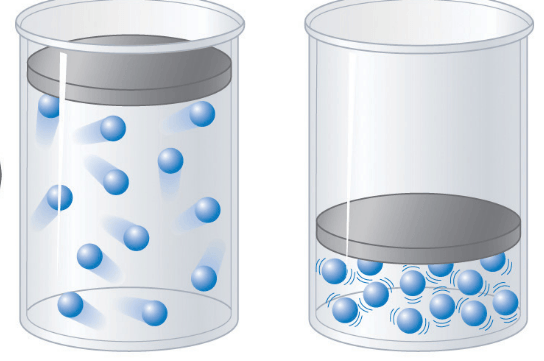
Which container will have a lower pressure?
left
right
they both have the same pressure
I don't know
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
If a hairspray can is heated, what can be expected of the pressure of the gas inside the can?
The pressure will increase
The pressure will decrease
The pressure will remain constant
The pressure will equalize
15
Multiple Choice
A student inflates a balloon with helium then places it in the freezer. The student should expect
the balloon's volume to increase
the balloon's volume to decrease
the balloon's moles to increase
the balloon's moles to decrease
16
Multiple Choice

17
Multiple Choice
If the temperature of a gas is doubled from 100K to 200K, what happens to the volume of the gas?
Cuts in half
Doubles
Triples
Stays the Same
18
Multiple Choice
According to Charles Law, if Temperature of a gas increases Volume will....
Increase
Decrease
Stay the Same
19
Multiple Choice
According to Charles Law, the relationship between Temperature and Volume can best be described as....
Direct Relationship
Inverse Relationship
20
Multiple Choice
If the Kelvin temperature of a gas is doubled, the volume of the gas will increase by ____.
A factor of 2
A factor of 1
A factor of 3
A factor of 0.5
21
Multiple Choice
When the temperature of a gas decreases (with pressure and amount held constant), does the volume increase or decrease?
Unaltered
Remains constant
Increase
Decrease
22
Multiple Choice
When the temperature of a gas decreases (with pressure and amount held constant), does the volume increase or decrease?
Unaltered
Remains constant
Increase
Decrease
23
Open Ended
Describe what is happening inside of the bag as the car goes further into the mountains? Describe in terms of what is happening to the gases particles trapped inside of the bag.
24

25
Multiple Choice
26
Multiple Choice
Gases have...
A definite shape and volume
A definite shape but no definite volume
No definite shape but a definite volume
No definite shape or volume
27
Multiple Choice
What is the variable for this number 32oC
P
T
n
V
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice
31

Determine the Kelvin temperature required for 0.0470 mol of gas to fill a balloon to 1.20 L under .998 atm pressure.
32
Multiple Choice
33

Calculate the volume that a 0.323-mol sample of a gas will occupy at 265 K and a pressure of 0.900 atm.
34
Multiple Choice
PV=nRT
35
Poll
Where do you think you are on the scale after the lesson?
1
2
3
4
Gases, Liquids, and Solids - Section 3

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

27 questions
Reaction Rates
Presentation
•
9th - 12th Grade

30 questions
History of atomic theory
Presentation
•
10th - 12th Grade

28 questions
9.1 Respiratory System
Presentation
•
11th - 12th Grade

31 questions
Quizizz Lesson: Unit 5 Lesson 2: Transition Metals
Presentation
•
10th - 12th Grade

26 questions
Simple Harmonic Motion
Presentation
•
11th - 12th Grade

26 questions
Ionic Compounds Naming and Formula Writing
Presentation
•
10th Grade - University

25 questions
Unit 4 Lesson 3 Quantum Numbers
Presentation
•
10th - 12th Grade

29 questions
Families of the Periodic Table
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade