

Compound and Mixture _2_Lesson
Presentation
•
Science, Physics, Chemistry
•
7th Grade
•
Medium
Kasthuri Subramaniam
Used 1+ times
FREE Resource
6 Slides • 38 Questions
1
Compound and Mixture _2_Lesson

2
Chemical Formula
a representation of a substance (compound) in which the elements are represented by their symbols and subscripts represent the number of atoms of each element
Possible examples may include: CO2: carbon dioxide CO: carbon monoxide H2O: water NaCl: sodium chloride C6H12O6: glucose CaCO3: calcium carbonate SiO2: silicon dioxide
3
Compound
A pure substance made by chemically combining two or more elements
Represented by a chemical formula
The proportion of each element in a compound is constant (if the subscript is changed, the substance is changed)
4
Elements
Made up of ONE type of Atom
Cannot be broken down chemically into simpler substances
Organized on the Periodic Table of the Elements
Represented by a Chemical symbol
Properties can be used to identify different elements
5

Sample questions
6

7
Multiple Choice

If I have a mixture of wooden blocks and metal blocks. What is a fast way to separate them?
relative density (put them in water)
filter
sieve
8
Multiple Choice

If I pour sand and water onto a filter, what will happen?
water and sand will go through the filter
sand will get caught in the filter, water will pass through
both sand and water will get caught in the filter
9
Multiple Choice

A solution of salt and water was put over heat. What will happen next?
The salt will evaporate
the salt and water evaporate
the water will evaporate
10
Multiple Choice

This type of paper allows liquid to pass through it. What tool is it?
sieve
filter
11
Multiple Choice

What tool is this?
sieve
filter
12
Multiple Choice

The best way to separate trail mix would be:
filter
by hand
sieve
evaporation
13
Multiple Choice

If I added 27 grams of salt to water and separated the solution with a hot plate, what amount of salt would be left behind?
20 grams
0 grams
27 grams
10 grams
14
Multiple Choice
To separate a mixture of salt and water what separation tool/method is used?
Filter
Sieve
Magnet
Evaporation
15
Multiple Choice

If you spilled nails in a sandbox, what separation tool/method could you use to clean out the sandbox so it would be safe for children to play in?
Filter
Screen
Magnet
Evaporation
16
Multiple Choice
You have a glass of salt and water, what is the best method to separate the salt out of the water?
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
An Element is made up of how many types of atoms?
12
36
58
1
21
Multiple Choice
Sam's mother is cooking breakfast in a cast-iron frying pan. If the pan is made only of iron, what can Sam conclude?
The pan is a pure substance with the chemical symbol Ir.
The pan is a pure substance with the chemical symbol I.
The pan is a pure substance with the chemical symbol F.
The pan is a pure substance with the chemical symbol Fe.
22
Multiple Choice

Which of the following is true of oxygen?
Oxygen is an element because its most basic unit is a single type of atom
Oxygen is a compound because its most basic unit is a single type of atom.
Oxygen is a mixture because its most basic unit contains more than one particle.
Oxygen is a compound because its most basic unit contains more than one type of particle.
23
Multiple Choice
Beatriz wants to make a mixture out of her bowl of blueberries. Which of the following would turn her bowl of blueberries into a mixture?
adding more blueberries to the bowl
adding raspberries to the bowl
cutting up the blueberries
mashing the blueberries
24
Multiple Choice
Humans absorb oxygen from the air they breathe in and exhale carbon dioxide gas, CO2. Carbon dioxide gas is
an element made of carbon and oxygen atoms.
a solution made of carbon and oxygen atoms.
a compound of carbon and oxygen atoms.
a mixture of carbon and oxygen atoms.
25
Multiple Choice
Mixtures are different than pure substances because mixtures-
have lower boiling points
can be separated physically
are less dense than pure substances
contain only one type of atom
26
Multiple Choice
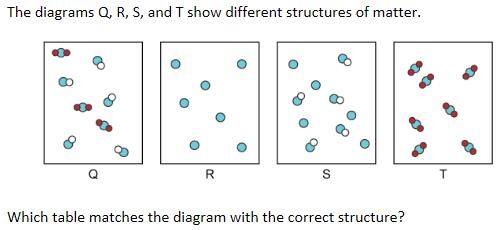
Which answer correctly matches the diagram with the correct description?
Q- Multiple compounds R-A single element S-An element and a compound T- A single compound
Q- A single element, R- An element and a compound S-A single compound T-Multiple compounds
Q- A single compound, R- A single element, S- Multiple compounds, T- An element and a compound
Q- An element and a compound, R- a single compound, S- a single element, T- multiple compounds
27
Multiple Choice
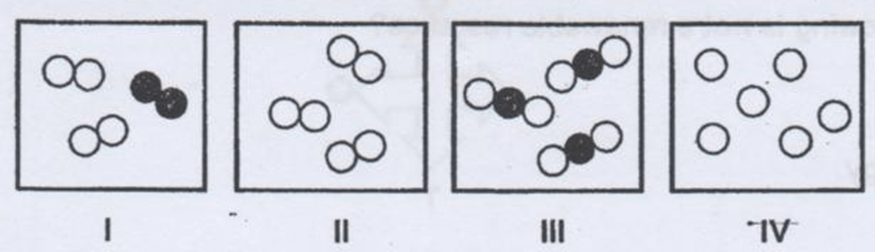
In the diagrams provided, the circles of different colors represent the atoms of different elements. Which diagram represents a mixture?
I
II
III
IV
28
Multiple Choice
What is the relationship between molecules and atoms?
Molecules are combinations of two or more atoms.
Molecules are the parts that make up atoms
Molecules are the gaseous parts of atoms
Molecules are atoms that have a positive charge
29
Multiple Choice
What makes elements and compounds different from mixtures? Elements & compounds-
have a constant composition (pure substances)
are made of gases
Will not change when heated
conduct electric current
30
Multiple Choice

Label 3
Beaker
Conical Flask
Boiling tube
rounded bottom flask
31
Multiple Choice

Label part 1
Thermometer
Fractionating column
condenser
32
Multiple Choice
Which of the following pairs of substances may best be separated through through fractional distillation
sand and soil
water and soil
salt and sand
water and alcohol
33
Multiple Choice
34
Multiple Choice

35
Multiple Choice

36
Multiple Choice

37
Multiple Choice

38
Multiple Choice

This diagram represents...
An element
A molecule
A compound
A mixture of elements and compounds
39
Multiple Choice

40
Multiple Choice
41
Multiple Choice
42
Multiple Choice

Name the method of separation
Fractional distillation
Chromatography
Condensation
Evaporation
43
Multiple Choice
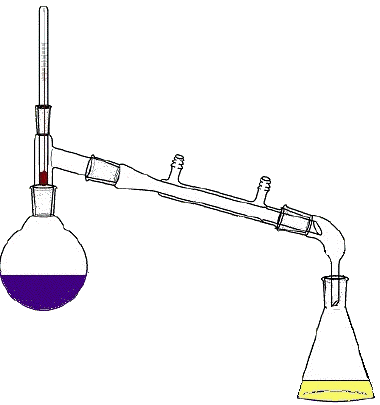
44
Multiple Choice
Which one of the following would you use to separate sand from iron filings?
a bar magnet
filter paper
chromatography paper
alum
Compound and Mixture _2_Lesson

Show answer
Auto Play
Slide 1 / 44
SLIDE
Similar Resources on Wayground

35 questions
Wave Properties
Presentation
•
7th Grade

35 questions
Structure and Function of Cells
Presentation
•
7th Grade

38 questions
Science 7 Year End Review - Unit A
Presentation
•
7th Grade

42 questions
Biomes
Presentation
•
7th Grade

37 questions
Force and Motion Review
Presentation
•
7th - 8th Grade

40 questions
Volcanoes and Earthquakes
Presentation
•
7th Grade

35 questions
Science 7 End of Year Review - Unit E
Presentation
•
7th Grade

35 questions
Genetics and Heredity
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

19 questions
Natural and Artificial Selection
Quiz
•
7th Grade