

10.5 The Solid State of Matter
Presentation
•
Chemistry
•
University
•
Easy
+2
Standards-aligned

Luis Bello
Used 1+ times
FREE Resource
28 Slides • 12 Questions
1
10.5 The Solid State of Matter

Presenter: Dr. Luis Bello
Textbook link:
https://openstax.org/books/chemistry-2e/pages/10-5-the-solid-state-of-matter
2
Previous Class
Phase Transitions and Diagrams (10.3 and 10.4)
Define phase transitions and phase transition temperatures
Explain the relation between phase transition temperatures and intermolecular attractive forces
Describe the processes represented by typical heating and cooling curves, and compute heat flows and enthalpy changes accompanying these processes
3
Multiple Choice

What line segment represents only the solid state? (Diagram F)
A-B
B-C
C-D
D-E
4
Multiple Choice
5
Multiple Choice

Is the substance gaining or losing energy? (Diagram D)
Gaining
Losing
6
Multiple Choice
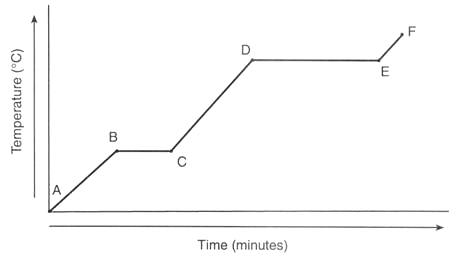
Between which points is the temperature of the substance remaining constant? (Diagram F)
A-B only.
A-B, C-D, E-F
B-C only.
B-C, D-E
7
Multiple Choice

What will occur when the substance transitions from B to A? (Diagram A)
condensation
evaporation
melting
freezing
8
Multiple Choice

What will occur when the substance changes from 0o C to 100oC at a constant pressure of 30 atm? (Diagram A)
condensation
evaporation
melting
freezing
9
10.5 The Solid State of Matter. Learning Objectives
Define and describe the bonding and properties of ionic, molecular, metallic, and covalent network crystalline solids
Describe the main types of crystalline solids: ionic solids, metallic solids, covalent network solids, and molecular solids
Explain the ways in which crystal defects can occur in a solid
10
11
The solid-state refers to a state of matter in which the constituent particles (atoms, molecules, or ions) are held tightly together by intermolecular forces, resulting in a rigid and closely packed arrangement.
The particles in a solid are in a fixed position and vibrate around that position, but they do not have enough kinetic energy to overcome the attractive forces between them and move freely, as in a liquid or gas.

12
In solids, the intermolecular forces between particles are strong enough to hold them together in a fixed position, and these forces vary depending on the type of particles and the arrangement of the solid's structure.
For example, ionic solids are held together by strong electrostatic forces between oppositely charged ions, while covalent solids are held together by covalent bonds between atoms, and metallic solids are held together by metallic bonds between atoms.

13

The arrangement of particles in a solid can also have a profound effect on its physical properties, such as density, melting point, and hardness.
Crystalline solids have a regular and repeating arrangement of particles, resulting in characteristic geometric shapes, whereas amorphous solids lack a regular pattern of particles and have a more random arrangement.
14
The four main types of crystalline solids are ionic solids, metallic solids, covalent network solids, and molecular solids.
15

16

17
Ionic Solids: Ionic solids are composed of cations and anions held together by strong electrostatic forces.
The ions are arranged in a regular, three-dimensional lattice structure. Examples of ionic solids include sodium chloride (NaCl) and magnesium oxide (MgO).

18

19
Metallic Solids: Metallic solids consist of closely packed metal atoms held together by metallic bonds, which are a type of delocalized covalent bond.
The delocalized electrons in metallic bonds give rise to the unique properties of metals, such as high thermal and electrical conductivity, ductility, and malleability. Examples of metallic solids include copper (Cu) and iron (Fe).

Figure 10.40 Copper is a metallic solid.
20
Covalent Network Solids: Covalent network solids consist of atoms held together by covalent bonds in a network or lattice structure.
They have high melting points, are hard and brittle, and have poor electrical conductivity. Examples of covalent network solids include diamond, graphite, and silicon dioxide (SiO2).

21

22
Molecular Solids: Molecular solids consist of individual molecules held together by weak intermolecular forces, such as Van der Waals forces or hydrogen bonding.
The molecules may be polar or nonpolar, and their arrangement in the solid can be amorphous or crystalline. Examples of molecular solids include ice (H2O) and solid carbon dioxide (CO2).

23

Crystal Defects
In a crystalline solid, the atoms, ions, or molecules are arranged in a definite repeating pattern, but occasional defects may occur in the pattern.
24
25

26

27

28

29

30

31

32
Semiconductors are materials that have electrical conductivity between that of insulators and conductors. They are the foundation of modern electronics and computing. Here's a brief overview of semiconductors:
Semiconductors are crystalline solids made from materials such as silicon, germanium, and gallium arsenide. They have a distinct crystalline structure with a repeating pattern of atoms.
In their pure form, semiconductors act as insulators and do not readily conduct electricity. However, they can be "doped" with tiny amounts of impurities to make them conduct electricity.
Doping introduces defects in the crystalline structure which create an excess of electrons (n-type) or electron vacancies (p-type). This allows current to flow when voltage is applied.
The junction between n-type and p-type semiconductors forms a diode that allows current to flow in only one direction. Diodes are a fundamental building block of semiconductor devices.
33

34

35
Multiple Choice
Smallest repeating unit of crystal lattice
Lattice site
Unit cell
Crystal lattice
Lattice point
36
Multiple Choice
Crystalline solids are
Isotropic
Allotropic
Anisotropic
Isomorphic
37
Multiple Choice
An amorphous solid
CaCl2
NaCl
Glass
CsCl
38
Multiple Choice
An example for Network solid
Corundum
Diamond
Dry Ice
KCl
39
Multiple Choice
Which of the following is not a characteristic of a crystalline solid?
Definite and characteristic heat of fusion.
Long range order or perfect arrangement of constituent particles
Isotropic in nature
clean cleavage or smooth edges on cutting with a knife
40
Multiple Choice

When a substance melts, the kinetic energy _____________.
decreases
stays the same
increases
decreases then increases
10.5 The Solid State of Matter

Presenter: Dr. Luis Bello
Textbook link:
https://openstax.org/books/chemistry-2e/pages/10-5-the-solid-state-of-matter
Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

35 questions
Language I. Week 2
Presentation
•
University

30 questions
VSEPR theory
Presentation
•
University

37 questions
Preterit UV, U, I, J verbs
Presentation
•
12th Grade

33 questions
CHAPTER 9: MANAGING COMMUNICATION & INFORMATION
Presentation
•
University

35 questions
Learning Styles
Presentation
•
University

35 questions
Q2. Assessment 1 Practice: Quiz
Presentation
•
KG

36 questions
334 Intracranial Regulation
Presentation
•
University

33 questions
Perfect Tenses
Presentation
•
University
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University