

8.5A Atomic Structure Reteach
Presentation
•
Science
•
7th - 8th Grade
•
Medium
Standards-aligned
Frankey Brewer
Used 56+ times
FREE Resource
12 Slides • 36 Questions
1
8.5A Atomic Structure Reteach
by Frances Brewer
2
3
Subatomic Particles
Atoms are made of 3 substomic particles
protons
neutrons
electrons
Protons and Neutrons are in the nucleus (center) and electrons are outside the nucleus in the electron cloud

4
Subatomic Particles
Each particle has a specific electric charge
Protons-- Positive (charge = +1)
Neutrons-- Neutral (charge = 0)
Electrons-- Negative (charge = -1)

5
Subatomic Particles
Protons and neutrons are together in the dense center region called the nucleus
Electrons are never in the nucleus. They orbit around the nucleus in a region known as the electron cloud.

6

7
Multiple Choice
What is the charge of a proton?
Negative
Positive
Neutral
8
Multiple Choice
What is the charge of a neutron?
Negative
Positive
Neutral
9
Multiple Choice
Which subatomic particles are found in the nucleus of an atom?
Protons and Electrons
Protons and Neutrons
Neutrons and Electrons
Protons, Neutrons and Electrons
10
Multiple Choice
What best describes the nucleus?
Located in the center of the atom, holds the atom's mass, and is positively charged.
Located in the center of the atom, does not hold the atom's mass and is negatively charged
Located in the center of the atom, holds the atom's mass, and has no charge
Located in the center of the atom, does not hold the atom's mass, and is positively charged.
11
Multiple Choice
What best describes a neutron?
It is located outside of the nucleus and has a negative charge
It is located inside of the nucleus and has a positive charge
It is located outside the nucleus and has no charge
It is located inside the nucleus and has no charge
12
Multiple Choice
What best describes a proton?
It is located outside the nucleus and has a positive charge.
It is located inside the nucleus and has a positive charge.
It is located inside the nucleus and has no mass
It is located inside the nucleus and has no charge.
13
Multiple Choice
What best describes an electron?
It is located outside of the nucleus and has a negative charge
It is located outside of the nucleus and has no charge
It is located inside the nucleus and has a mass
It is located inside of the nucleus and has no mass
14
Multiple Choice
Which subatomic particle has a negative charge?
proton
neutron
electron
all of the above
15
Multiple Choice
What best describes a proton?
It is located outside the nucleus and has a positive charge.
It is located inside the nucleus and has a positive charge.
It is located inside the nucleus and has no mass
It is located inside the nucleus and has no charge.
16
Multiple Choice

17
Multiple Choice
Which subatomic particle has a negative charge?
proton
neutron
electron
all of the above
18
Multiple Choice
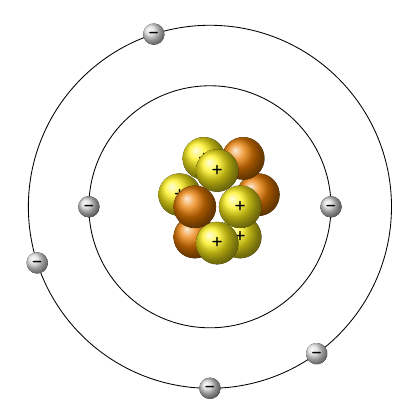
How many electrons does this atom have?
2
4
6
10
19
Elements on the Periodic Table
Every element has its own entry on the periodic table that tells the element's name, symbol, atomic number and atomic mass

20
Multiple Choice

What does the 6 represent?
Atomic mass
atomic number
chemical symbol
element name
21
Multiple Choice

What is the atomic mass of barium
56
82
137
138
22
Atomic Number
The number of Protons in an atom identify what element it is
The number of protons is known as the atomic number
Every element has a unique atomic number
For example, Carbon has 6 protons in its nucleus so it's atomic number is 6

23
Multiple Choice
What is the atomic number?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons in the energy levels
24
Multiple Choice
What particle number is equal to the atomic number ?
Protons
Electrons
Neutrons
25
Multiple Choice
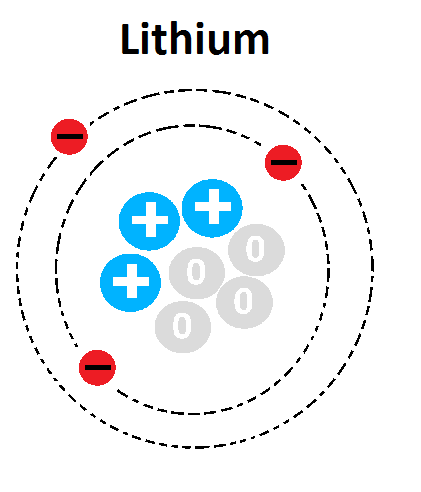
What is the atomic number of this atom?
1
3
4
7
26
Multiple Choice

Carbon has how many protons?
12
6
7
4
27
Multiple Choice

What is the atomic number of this atom?
8
16
24
none of the above
28
Multiple Choice

How many protons are in Gold
79
197
118
276
29
Multiple Choice
The atomic number of an element that has 9 protons, 9 electrons, and 10 neutrons is _____.
9
10
19
28
30
Multiple Choice

What is the atomic number of this atom?
2
4
6
none of the above
31
Multiple Choice
Which of the following determines the identity of an element?
number of protons
atomic mass
number of neutrons
number of shells
32
Multiple Choice
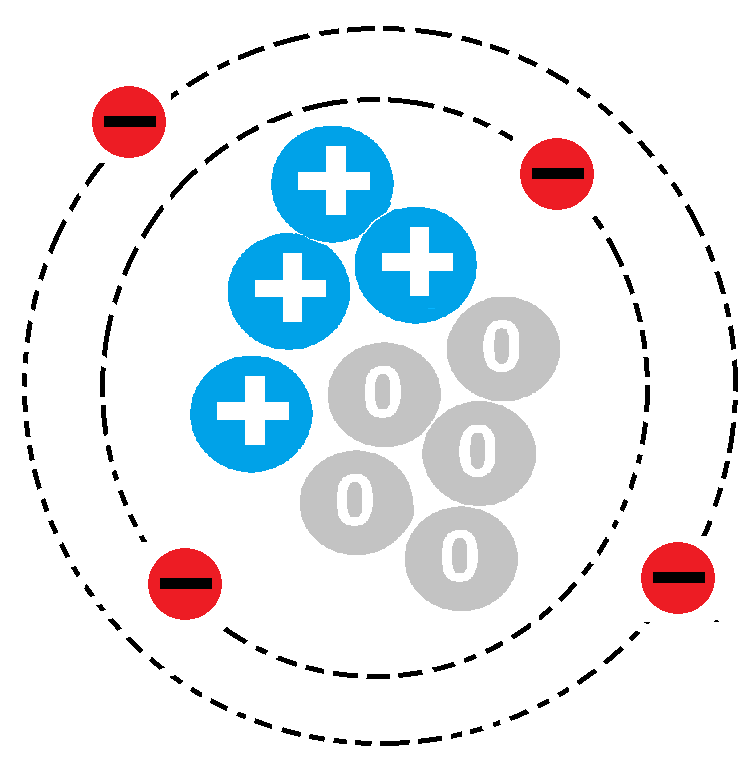
What is the atomic number of this atom?
4
5
9
none of the above
33
Atomic Mass
Protons and neutrons each have a mass of 1 amu, while electrons weigh nothing
All the mass of an atom is in its nucleus
When you add the protons and neutrons together, you get the atomic mass.
Carbon has 6 protons and 6 neutrons, so its atomic mass is 12

34
Multiple Choice
What is the atomic mass number?
the number of protons in the nucleus
the number of protons and neutrons in the nucleus
the number of neutrons in the nucleus
the number of protons and electrons in the atom
35
Multiple Choice

Oxygen has a mass of...
8
16
15
6
36
Multiple Choice

True or False - Protons and Neutrons have about the same mass.
True - They have about the same mass
False - They do not have about the same mass
37
Multiple Choice
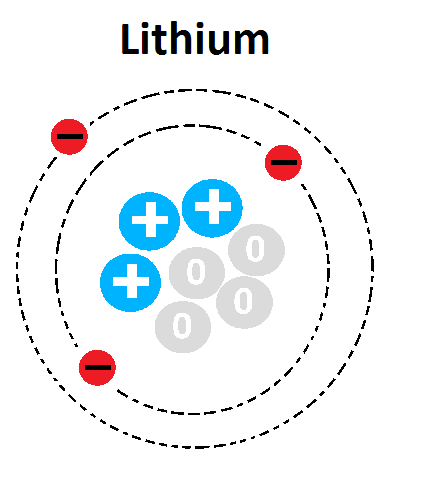
What is the mass number of this atom?
1
3
4
7
38
Number of Protons and Electrons
Atoms are most stable when their electrical charge = 0
An electrically neutral atom needs an equal number of protons (each has a +1 charge) and electrons (each has a -1 charge)

39
Multiple Choice
In order for an atom to be neutral what has to be true?
The atom has more protons than neutrons
The atom has more neutrons than protons
The atom has the same number of protons and neutrons
The atom has the same number of protons and electrons
40
Multiple Choice

How many electrons does sulfur have
16
32
48
12
41
Multiple Choice
An atom has an atomic number of 15 and a mass number of 31. How many protons are there in the atom?
15
31
16
47
42
Number of Neutrons
Neutrons have no charge and are not always the same as the number of protons or electrons
To find the number of neutrons you subtract the atomic number (number of protons) from the atomic mass (protons and neutrons)

43
44
Multiple Choice
How is the number of neutrons in the nucleus of an atom calculated?
Add the number of e- and p+ together
Subtract the number of e- from p+
Subtract the number of p+ from the mass number
Add the mass number to the number of e-
45
Multiple Choice

How many neutrons does platinum have?
78
117
195
196
46
Multiple Choice
An element with a mass number of 11 and an atomic number of 5 has how many neutrons?
11
5
6
16
47
Multiple Choice

Bromine has how many Neutrons?
35
80
45
8
48
Multiple Choice

How many neutrons are in Tungsten
110
74
183
258
8.5A Atomic Structure Reteach
by Frances Brewer
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

42 questions
AKS 9B - Electromagnetic vs. Mechanical Waves
Presentation
•
8th Grade

43 questions
Earth in Space
Presentation
•
7th - 8th Grade

44 questions
Rock Transformations Mini Review
Presentation
•
6th - 8th Grade

39 questions
The Sky from Earth
Presentation
•
7th - 8th Grade

44 questions
Unit 1 - Matter and Chemistry Review
Presentation
•
7th - 9th Grade

44 questions
Clouds
Presentation
•
6th - 8th Grade

44 questions
Earths Layers Review
Presentation
•
6th - 8th Grade

42 questions
Mitosis and Meiosis Review
Presentation
•
7th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade