

Atoms and Elements Lesson
Presentation
•
Science
•
9th - 12th Grade
•
Medium
Standards-aligned
David Blankenship
Used 3+ times
FREE Resource
27 Slides • 45 Questions
1
Atoms and Elements Tutorial
by David Blankenship
2

3

4

5

6

7

8
Multiple Choice
The atomic number tells you what?
number of electrons
number of protons
number of neutrons
both electrons and protons in an atom.
9
Multiple Choice
Element is a
pure substance
compound
mixture
molecule
10
Multiple Choice

What does the element AI stand for ?
Gold
Actinium
Iron
Aluminum
11
Multiple Choice
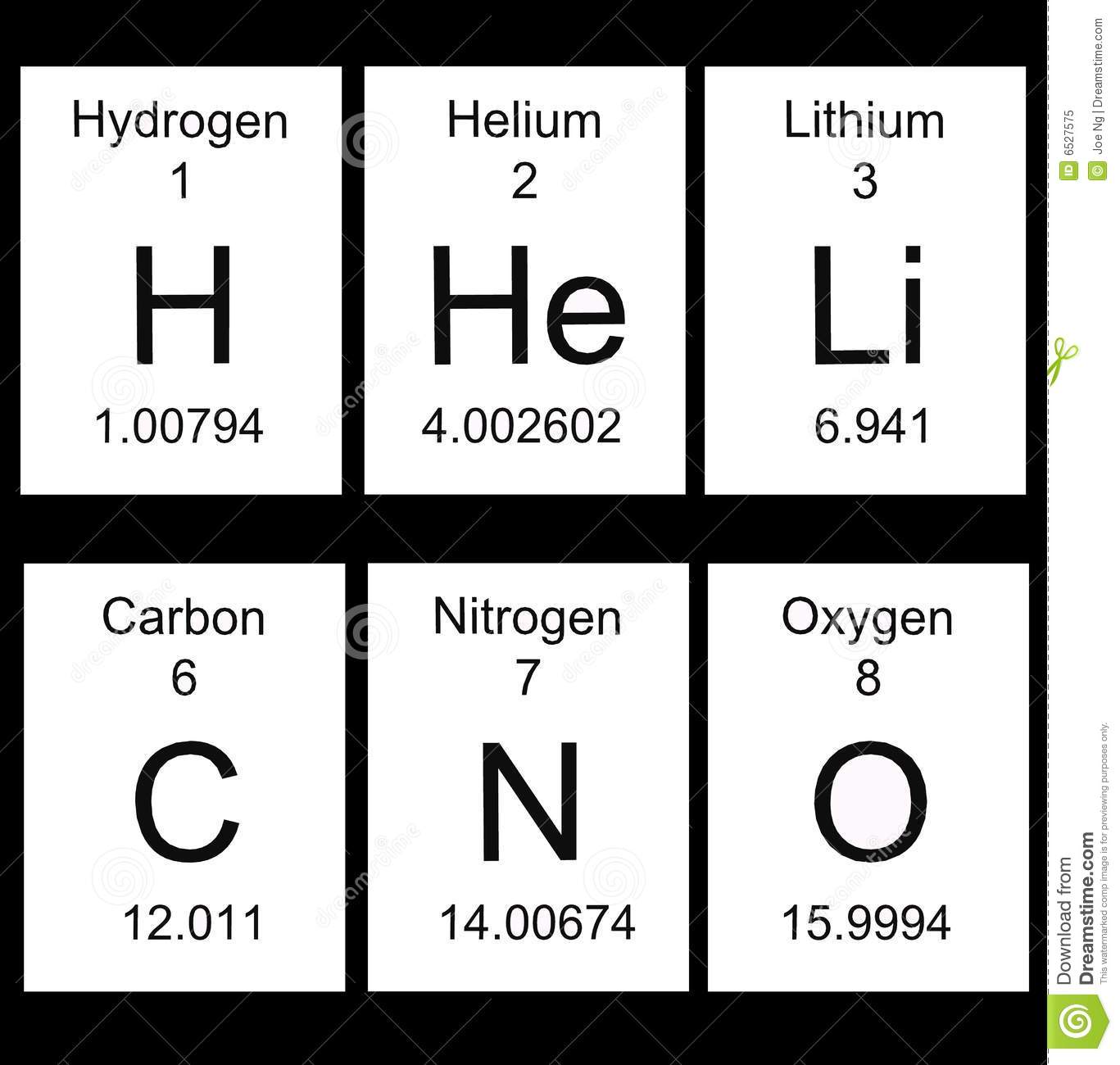
The number at the top of each square on the periodic table is the ...
atomic number
atomic mass
Chemical Symbol
Element Name
12

13

14

15

16

17
Multiple Choice
What is atomic number?
proton number
electron number
18
Multiple Choice

The fluorine atom has an atomic number of
19
9
19
Multiple Choice

What is the mass number and atomic number ?
Mass number = 7, atomic number = 14
Mass number = 14, atomic number = 7
20
Multiple Choice

How many neutrons are there in one atom of magnesium?
12
6
24
21
Multiple Choice
What is neutron number?
the number of neutrons in the nucleus of an atom, equal to the mass number plus the atomic number of the atom
the number of neutrons in the nucleus of an atom, equal to the mass number minus the atomic number of the atom
22
Multiple Choice
ALL atoms of the same element have:
same number of proton
same number of nucleon
different number of neutron
different number of electron
23
Multiple Choice
Elements that have _____________ are in the same group
physical properties
chemical properties
24
Multiple Choice
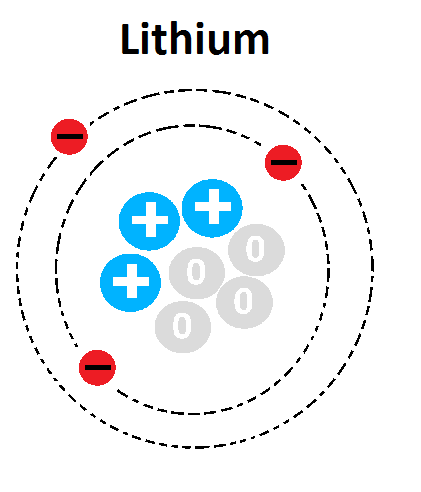
What is the atomic number of this atom?
1
3
4
7
25
Multiple Choice
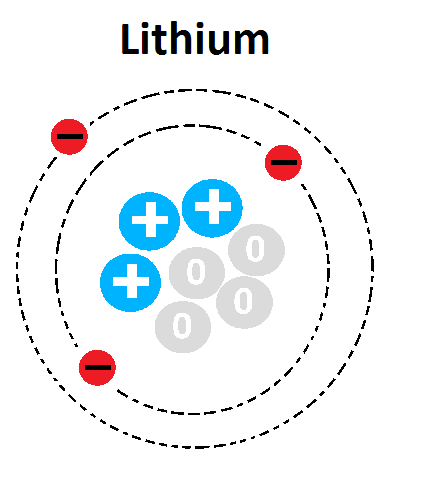
What is the mass number of this atom?
1
3
4
7
26
Multiple Choice
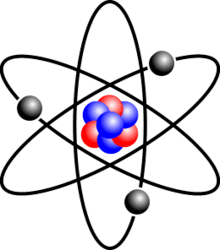
Which subatomic particle has a negative charge in the atom?
proton
neutron
electron
quark
27

28
Multiple Choice

Please choose the correct answer of the Periodic Table Trends.
atomic mass increase across the period.
atomic decrease down the group
29

30

31
Multiple Choice
Valence electrons are the...
Innermost electrons
Middle electrons
Outermost electrons
Any electrons
32
Multiple Choice

2
8
3
13
33
Multiple Choice

2
3
5
34
Multiple Choice

12
7
8
14
35
Multiple Choice

7
8
9
10
36
Multiple Choice
USE THE PERIODIC TABLE
Find the element that has 3 Valence Electrons and 2 energy levels.
Magnesium - Mg
Lithium - Li
Aluminum - Al
Boron - B
37
Multiple Choice
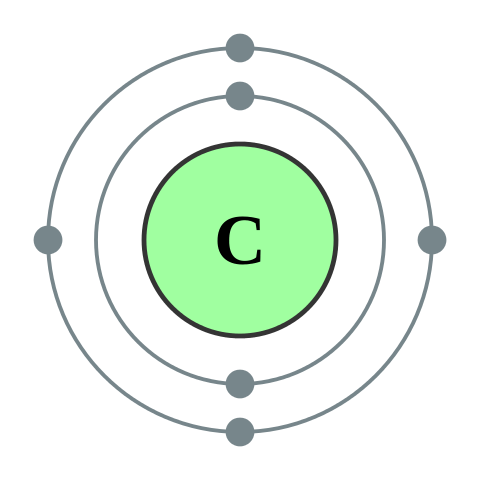
How many valence electrons does carbon have?
4
5
6
7
38
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does sodium have?
1
2
3
4
39
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does Phosphorus have?
31
5
15
4
40
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does carbon have?
4
12
6
14
41

42

43
44

45

46

47
Multiple Choice
Metals generally have the following properties EXCEPT for being:
Shiny
Non-conductors
Malleable
Reactive with HCl/CuCl2
48
Multiple Choice
Nonmetals do NOT have which of the following characteristics:
Shiny
Brittle
Non-conductor
Non-reactive with HCl/CuCl2
49
Multiple Choice
Which of the following is a physical property?
Flammability
Reactivity with H2O2
Color
Toxicity
50
Multiple Choice
Iodine (I2) crystals would be expected to be ______________ because it is a ___________________.
shiny, metal
shiny, nonmetal
malleable, metal
brittle, nonmetal
51
Multiple Choice
An atom is made up of how many different types subatomic particles?
One
Two
Three
Four
52
Multiple Choice
Where are protons found in an atom?
The nucleus
Energy level
The atomic mass
By electrons
53
Multiple Choice
What are the rows of the periodic table of elements called?
Columns
Groups
Periods
Lines
54
Multiple Choice
________ are good conductors of electricity; they are often shiny solids at room temperature.
Non-metals
Metalloids
Metals
Noble gases
55
Multiple Choice
The __________ tells you how many protons an element has.
atomic number
atomic mass
energy levels
electrons
56
Multiple Choice
Which of these is a distinguishing property for the alkali metals?
Soft - can cut with a plastic knife
Unreactive - don't give or receive electrons
Shiny - exhibits luster
Poor conductors of electricity
57
Multiple Choice
Which of these is a distinguishing property for the noble gases?
Soft - can cut with a plastic knife
Unreactive - don't give or receive electrons
Shiny - exhibits luster
Poor conductors of electricity
58
Multiple Choice
Which of these is a distinguishing property for the nonmetals?
Soft - can cut with a plastic knife
Unreactive - don't give or receive electrons
Shiny - exhibits luster
Poor conductors of electricity
59
Multiple Choice
An unknown element is observed to be shiny, malleable, ductile, and is found to conduct heat and electricity very well. What group might this element fall into?
Alkali metals
Alkali earth metals
Transition metals
Non-metals
60
Multiple Select
Select which of these are properties of non-metals
Good conductors of heat & electricity
Colorful
Form anions (negative charges)
Soft
Varying physical states at room temp
61
Multiple Choice
Nickel would be expected to be ____________________ because it is a __________________.
conductive, metal
conductive, nonmetal
nonconductive, metal
nonconductive, nonmetal
62
63

64

65

66
Multiple Choice
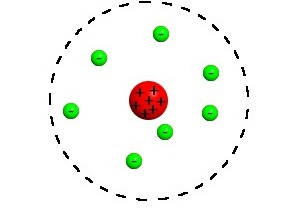
This model this model was the first to show a nucleus, consisting of protons and neutron. Electrons surround the nucleus but are not shown in distinct energy levels.
The "Rutherford Model" of the atom
The "Plum Pudding Model" of the atom
The "Quantum Mechanical Model" of the atom
67
Multiple Choice

This model was developed after J.J. Thompson discovered electrons, a particle smaller than an atom. It shows electrons floating freely in a positive region.
The "Plum Pudding Model" of the atom
The "Rutherford Model" of the atom
Democritus's model of the atom
The "Quantum Mechanical Model" of the atom
68
Multiple Choice

Ernest Rutherford discovered that atoms were mostly _________________.
negatively charged
positively charged
electrons
empty space.
69
Multiple Choice

In the atomic model nicknamed the "plum pudding" model, what do the plums represent?
the nucleus
the atom
the electrons
the positive material
70
Multiple Select
What subatomic particles make up the nucleus of an atom?
electrons
protons
neutrons
leptons
mesons
71

72

Atoms and Elements Tutorial
by David Blankenship
Show answer
Auto Play
Slide 1 / 72
SLIDE
Similar Resources on Wayground

65 questions
Organic Chemistry
Presentation
•
9th - 12th Grade

71 questions
The Water Cycle Lesson
Presentation
•
9th - 12th Grade

69 questions
Evidences of Evolution
Presentation
•
9th - 12th Grade

69 questions
Introduction to Chemical Reactions
Presentation
•
9th - 12th Grade

64 questions
Plant Nutrient Requirements
Presentation
•
9th - 12th Grade

64 questions
Biology Honors Chapter 3.2 - Climate, Weather, and Life
Presentation
•
9th - 12th Grade

66 questions
Human Body Cells LPN
Presentation
•
KG - University
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade