

Integrated Science Final Reveiw
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+19
Standards-aligned
David Blankenship
Used 21+ times
FREE Resource
1 Slide • 166 Questions
1
Integrated Science Final Reveiw
by David Blankenship
2
Multiple Choice
3
Multiple Choice
Samuel adds a teaspoon of salt to a glass of water. He notices that the salt disappears. Samuel takes a sip to discover that the water tastes salty. What kind of change has occurred?
It’s a chemical reaction because a new substance has formed.
It’s a physical change because the water and the salt kept their original properties.
It’s an example of synthesis because two substances reacted to give one substance.
It’s a chemical reaction because the water now tastes salty but originally did not.
4
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of sulfur atoms is ___?
1
2
4
8
5
Multiple Choice
Which chemical reaction shows photosynthesis?
water + carbon dioxide + sunlight → oxygen + glucose
carbon + oxygen → carbon dioxide
hydrogen + oxygen → water
nitrogen + hydrogen → ammonia
6
Multiple Choice
Jack tries to place magnets on the door of his refrigerator. He observes that the magnets don’t stick. He guesses that the door of the refrigerator is made of a nonmagnetic substance. What stage of scientific investigation does his guess represent?
A. ask questions
B. construct a hypothesis
C. explain the results
D. make observations
7
Multiple Choice
Which of these reactions is a combustion reaction?
burning wood
decomposition of water
production of food by plants with the help of the Sun's energy
rotting of fruits and vegetables
8
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of oxygen atoms is _____?
1
2
4
8
9
Multiple Select
Which sentences describe disadvantages of burning fossil fuels instead of using hydrogen fuel cells?
Burning fossil fuels pollutes the environment.
Burning fossil fuels takes place inside expensive fuel cells.
Fossil fuels aren’t widely available, but pure hydrogen is.
Fossil fuels are nonrenewable.
10
Multiple Choice
Carbon burns in the presence of oxygen to give carbon dioxide. Which chemical equation describes this reaction?
carbon + oxygen + carbon dioxide
carbon + oxygen → carbon dioxide
carbon dioxide → carbon + oxygen
carbon dioxide + carbon → oxygen
11
Multiple Choice
In a given chemical reaction, the energy of the products is less than the energy of the reactants. Which statement is true for this chemical reaction?
Energy is absorbed in the reaction.
Energy is released in the reaction.
There is no transfer of energy in the reaction.
Energy is lost in the reaction.
12
Multiple Choice
When sodium reacts with chlorine, sodium chloride is produced.
Andrew represented this reaction with this equation:
Na + Cl2 → 2NaCl
The equation is _____________.
balanced
not balanced
13
Multiple Choice
A hydrogen fuel cell is more efficient than a combustion reaction because very little of the released energy is released as ______?
heat
electrical energy
chemical energy
14
Multiple Choice

15
Multiple Choice
When used at home, a fuel cell eliminates the need for _________ because the cell is portable.
oxygen
hydrogen
an electrical grid
16
Multiple Choice
When sodium reacts with chlorine, sodium chloride is produced. Andrew represented this reaction with this equation:
__Na + __Cl2 →__NaCl
Which set balances the equation?
1,1,1
1,2,1
1,1,2
2,1,2
17
Multiple Choice
In a chemical reaction, activation energy increases __________ of the reactants.
the amount of chemical energy
number of particles
the temperature
18
Multiple Choice
Iron oxide reacts with aluminum to give aluminum oxide and iron. What kind of chemical reaction is this?
combustion
decomposition
synthesis
replacement
19
Multiple Choice
Increasing activation energy causes the particles to collide, which results in the _____________ of new products.
breaking up
formation reaction
20
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of carbon (C) on each side of the equation?
2
4
6
8
21
Multiple Select
Which chemical reactions are based on whether they absorb energy.
respiration in cells
photsynthesis
burning a candle
baking bread
activating a cold pack
22
Multiple Choice
Meg goes swimming on a hot afternoon. When she comes out of the pool, her foot senses that the pavement is unbearably hot. Suppose Meg wants to apply the scientific method to discover reasons for the hot pavement. What is the next step she should take?
A. analyze the data
B. ask questions
C. communicate the results
D. make observations
23
Multiple Select
Which chemical reactions are based on whether they release energy.
respiration in cells
producing electric current
burning a candle
baking bread
activating a cold pack
24
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of oxygen (O) on each side of the equation?
2
13
15
26
25
Multiple Choice

Arrange the characteristics of the reaction in order from start to finish.
1. lower energy of reactants
2. transition state
3. higher energy of products
1. lower energy of reactants
2. higher energy of products
3. transition state
1. higher energy of reactants
2. lower energy of products
3. transition state
1. higher energy of products
2. transition state
3. lower energy of reactants
26
Multiple Choice
What kind of chemical reaction does the chemical equation sodium + chlorine → sodium chloride represent?
combustion
decomposition
single replacement
synthesis
27
Multiple Choice

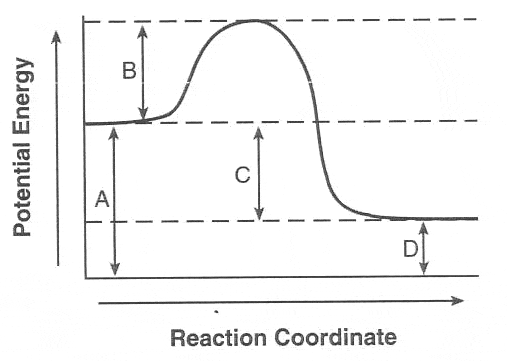
Identify the parts of the energy diagrams.
Part A.
energy released
energy absorbed
reactants
products
28
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of hydrogen (H) on each side of the equation?
10
12
20
26
29
Multiple Choice

Identify the parts of the energy diagrams.
Part B.
energy released
energy absorbed
reactants
products
30
Multiple Choice

31
Multiple Choice

Identify the parts of the energy diagrams.
PartC.
energy released
energy absorbed
reactants
products
32
Multiple Choice
Iron reacts with oxygen to produce iron oxide (rust). This reaction is represented in an equation as 4Fe + xO2 → 2Fe2O3.
Identify the value of x that will balance the equation.
1
2
3
4
33
Multiple Choice

Identify the parts of the energy diagrams.
PartC.
energy released
energy absorbed
reactants
products
34
Fill in the Blanks
Type answer...
35
Multiple Choice

Identify the parts of the energy diagrams.
Part B..
energy released
energy absorbed
reactants
products
36
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
carbon
1
3
5
7
No Answer
37
Multiple Choice
Some fuel cells are powered by hydrogen. Scientists are looking into the decomposition of water (H2O) to make hydrogen fuel with the help of the Sun’s energy.
What challenge do scientists face with this task?
Decomposing water requires a high activation energy.
The Sun isn't a renewable resource.
Decomposing water results in toxic byproducts.
Scientists are uncertain what to do with the excess oxygen.
38
Multiple Choice
When a candle burns, which forms of energy does the chemical energy in the candle change to?
A. light and sound
B. heat and sound
C. light and heat
D. electric and light
39
Multiple Choice
Scientists are looking into the decomposition of water (H2O --> H2 + H2O) to make hydrogen fuel with the help of the Sun’s energy. What challenge do scientists face with this task?
Decomposing water requires a high activation energy.
The Sun isn't a renewable resource.
Decomposing water results in toxic byproducts.
40
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
hydrogen
1
3
5
7
No Answer
41
Multiple Choice
Endothermic reactions ______.
release more energy than they absorb
absorb more energy than they release
release and absorb exactly the same amount of energy
can either release more energy than they absorb or absorb more energy than
they release, depending on the reaction
42
Multiple Choice
Water breaks down into hydrogen and oxygen.
neutralization
combustion
synthesis
decomposition
43
Multiple Choice
Energy is _________ when bonds form.
released
absorbed
destroyed
created
44
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
sulfur
1
3
5
7
No Answer
45
Multiple Choice
Energy is _________ when bonds break.
absorbed
created
destoryed
released
46
Multiple Choice

47
Multiple Choice
Energy is needed to break bonds in chemical reactions.
True
False
48
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
Oxygen
1
3
5
7
No Answer
49
Multiple Choice
A chemical reaction in which more energy is absorbed than is released is endothermic
True
False
50
Multiple Choice
Leaves make starch using chlorophyll and carbon dioxide.
neutralization
combustion
synthesis
decomposition
51
Multiple Choice

Temperature usually _____ in EXOthermic reactions.
Stays the same
Increases
Decreases
52
Multiple Select
Which chemical equations are being balanced ?
CaO + 3C → CaC2 + CO
Na + H2O → 2NaOH + H2
4Fe + O2 → 2Fe2O3
2Mg + O2 → 2MgO
53
Multiple Choice

Photosynthesis is an _____ reaction.
EXOthermic
ENDOthermic
54
Multiple Choice
Based on the law of conservation of energy, which statement is correct?
A. Energy is always being added to all parts of the Universe.
B. Energy is often destroyed in some parts of the Universe.
C. Energy in a closed system cannot change forms.
D. Energy in an isolated system remains constant.
55
Multiple Choice
56
Multiple Choice
Calcium carbonate is a compound that is used to make chalk for drawing on rocks or pavement. One molecule has one atom of calcium (Ca), one atom of carbon (C), and three atoms of oxygen (O). Which is the correct way to write the chemical formula for calcium carbonate?
3CaCO
Ca3CO
CaCO3
Ca3C3O3
57
Multiple Choice

Which letter corresponds to the activation energy?
A
B
C
D
58
Multiple Choice
Food burns in oxygen gas and releases a lot of energy.
neutralization
combustion
synthesis
decomposition
59
Multiple Choice

The picture represents sulfur trioxide.
Which statement is true for one molecule of sulfur trioxide?
There are three atoms of sulfur and one atom of oxygen.
There are three atoms of sulfur and three atoms of oxygen.
There is one atom of sulfur and one atom of oxygen.
There is one atom of sulfur and three atoms of oxygen.
60
Multiple Choice
61
Multiple Choice
If blue represents one atom of nitrogen, and red represents one atom of oxygen, which picture represents 2NO2?
62
Multiple Choice
Adding vinegar (acid) to baking soda (alkali) give sa product that is neither acidic nor alkaline.
neutralization
combustion
synthesis
decomposition
63
Multiple Choice
Sodium bicarbonate is the chemical name for baking soda. Its chemical formula is NaHCO3. What does the subscript 3 mean?
There are three molecules of sodium bicarbonate.
There are three molecules of oxygen in sodium bicarbonate.
There are three atoms of oxygen in sodium bicarbonate.
There are three atoms of sodium bicarbonate.
64
Multiple Select
Classify the items based on whether they are or are not matter. Check all the examples of matter.
toothpaste
light
gasoline
cell
sound
65
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of hydrogen atoms is_____?
1
2
4
8
66
Multiple Choice
Not all tiles will be used. Match the reactants with the products.
copper + oxygen →
aluminum chloride + copper
zinc sulfate + copper
copper oxide
copper sulfate + oxygen
copper + water
67
Multiple Choice
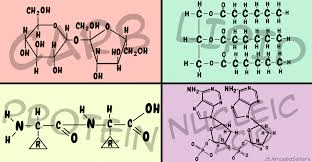
Which biomolecule is made of nucleotides and contains your DNA?
Carbohydrate
Lipids
Protein
Nucleic Acid
68
Multiple Choice
Not all tiles will be used. Match the reactants with the products.
aluminum + copper chloride →
aluminum chloride + copper
zinc sulfate + copper
copper oxide
copper sulfate + oxygen
copper + water
69
Multiple Select

Select the forms of energy in the image.
glass
heat
light
tungsten
electricity
70
Multiple Choice
Not all tiles will be used. Match the reactants with the products.
copper oxide + hydrogen →
aluminum chloride + copper
zinc sulfate + copper
copper oxide
copper sulfate + oxygen
copper + water
71
Multiple Choice
Which biomolecule helps form muscles?
Nucleic Acid
Carbohydrate
Protein
Lipids
72
Multiple Choice
Not all tiles will be used. Match the reactants with the products.
copper sulfate + zinc →
aluminum chloride + copper
zinc sulfate + copper
copper oxide
copper sulfate + oxygen
copper + water
73
Multiple Select
Select the volume units that are greater than one liter.
kiloliter
milliliter
megaliter
centiliter
nanoliter
74
Multiple Choice

Identify the following process: We light a matchstick.
Physical change
Chemical change
75
Multiple Choice
76
Multiple Choice
What type of changes the appearance change but the substance is the same.
Physical
Chemical
77
Multiple Choice

Identify the type of energy conversion represented by each picture.
wind energy to electric energy
electric energy to heat energy
solar energy to light energy
electric energy to light energy
78
Multiple Choice

Identify the following process: The food and the air react.
Physical change
Chemical change
79
Multiple Choice
80
Multiple Choice

The initial substances before occurring the chemical reaction are called...
Reactants
Products
81
Multiple Choice

Identify the type of energy conversion represented by each picture.
wind energy to electric energy
electric energy to heat energy
solar energy to light energy
electric energy to light energy
82
Multiple Choice

The new substances after occurring the chemical reaction are called...
Reactants
Products
83
Multiple Choice
84
Multiple Choice

What is the speed of this chemical reaction (rusting)
SLOW
FAST
85
Multiple Choice

Identify the type of energy conversion represented by each picture.
wind energy to electric energy
electric energy to heat energy
solar energy to light energy
electric energy to light energy
86
Multiple Choice

87
Multiple Choice

What are lipids?
Polymers made of amino acids
Monomers that store and transfer genetic information
Fatty acids not generally soluble in water
Complex carbohydrates like starch
88
Multiple Choice

89
Multiple Choice

Identify the type of energy conversion represented by each picture.
wind energy to electric energy
electric energy to heat energy
solar energy to light energy
electric energy to light energy
90
Multiple Choice

91
Multiple Choice

Which biomolecule makes up cell membranes?
Protein
Carbohydrate
Lipid
Nucleic acid
92
Multiple Choice
The mass conservation law says
During a chemical reaction, atoms are destroyed. The number of atoms don't remains constant throughout the reaction.
During a chemical reaction, atoms are created. The number of atoms remains constant throughout the reaction.
During a chemical reaction, atoms are neither created nor destroyed. The number of atoms remains constant throughout the reaction.
93
Multiple Choice
Match the sentences with the steps of the scientific method.
State a hypothesis.
An indoor plant in a dark room withers faster than the same
plant in a room with ample sunlight.
The plant in the pot with black paper withered.
The other plant was healthy.
Sunlight is probably essential for plants to grow and live.
Plants need sunlight to make food so they can live.
94
Multiple Choice

Identify the biomolecule
Carbohydrate
Lipid
Protein
Nucleic Acid
95
Multiple Choice
Match the sentences with the steps of the scientific method.
Make an observation.
An indoor plant in a dark room withers faster than the same
plant in a room with ample sunlight.
The plant in the pot with black paper withered.
The other plant was healthy.
Sunlight is probably essential for plants to grow and live.
Plants need sunlight to make food so they can live.
96
Multiple Choice
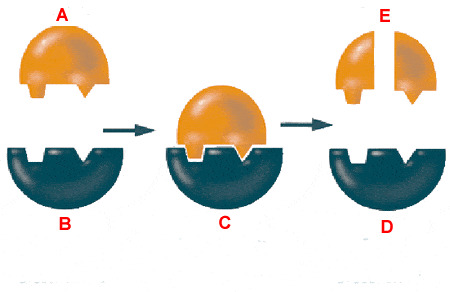
97
Multiple Choice
Match the sentences with the steps of the scientific method.
Analyze the results.
An indoor plant in a dark room withers faster than the same
plant in a room with ample sunlight.
The plant in the pot with black paper withered.
The other plant was healthy.
Sunlight is probably essential for plants to grow and live.
Plants need sunlight to make food so they can live.
98
Multiple Choice
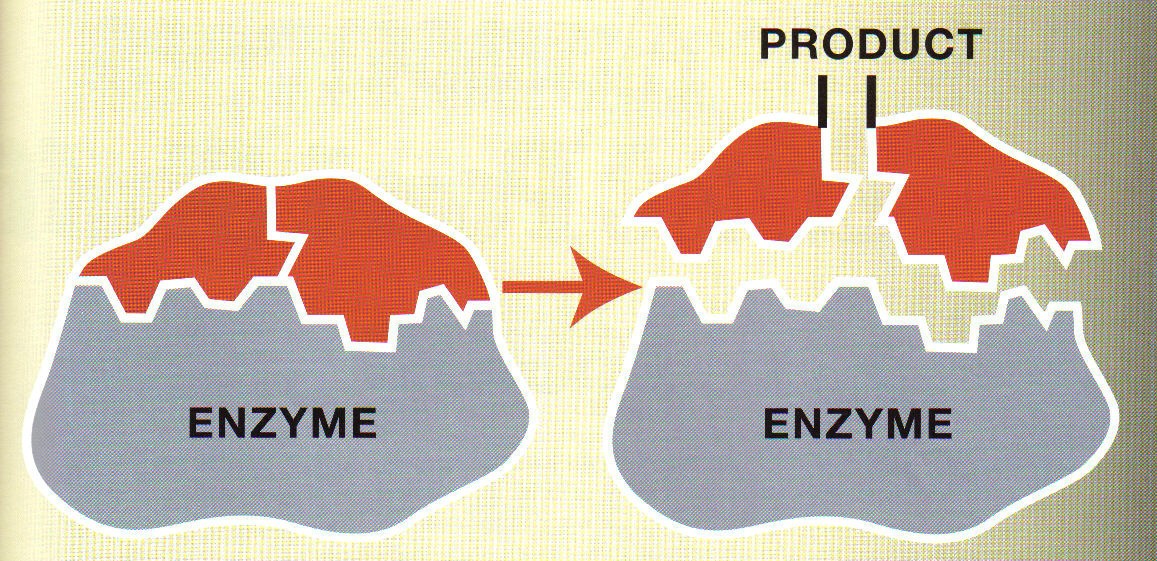
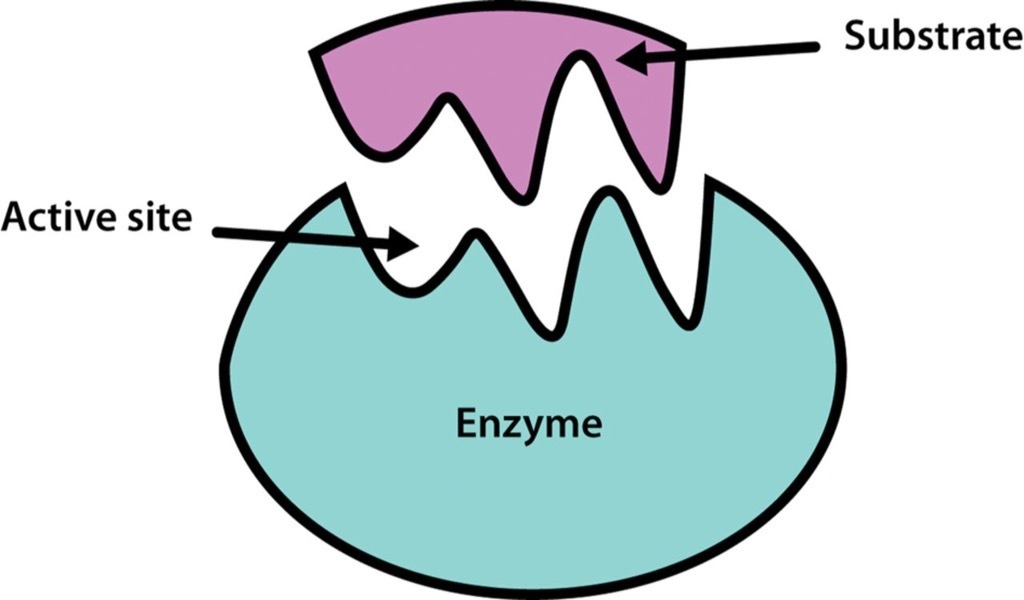
The area that the substrate fits into on the enzyme is called the:
Active Site
Allosteric Site
Construction Site
Geometric Site
99
Multiple Choice
Match the sentences with the steps of the scientific method.
Communicate the results to others.
An indoor plant in a dark room withers faster than the same
plant in a room with ample sunlight.
The plant in the pot with black paper withered.
The other plant was healthy.
Sunlight is probably essential for plants to grow and live.
Plants need sunlight to make food so they can live.
100
Multiple Choice
What two activities are basic to most of science?
Getting good grades and asking questions.
Paying attention in class and memorizing
Observing the world and asking questions
Observing science and reading answers
101
Multiple Choice
Science starts with gathering ________________________ about the natural world.
hypothesis
Answers
Experiments
Knowledge
102
Multiple Choice
The stuff that everything is made of is:
Earth
wind
Water
Matter
103
Multiple Choice
The ability to do work is called:
Power
energy
force
training
104
Multiple Choice
Moving objects have energy of:
motion
Chemical
Stored
mechanical
105
Multiple Choice
SI units to measure volume are:
Cups and quarts.
Gallons
Liters and cubic meter
Kilograms and pounds
106
Multiple Choice
How many mL are in 3.5 KL?
0.0035 mL
3,500 mL
35,000 mL
3,500,000 mL
107
Multiple Choice
Convert 70 ounces (Oz) to kilograms
4.375 Kg
31.76 Kg
1.985 Kg
1120 Kg
108
Multiple Choice
Convert 6000 mL to qt
6 qt
6.34 qt
5,676,000 qt
none
109
Fill in the Blanks
Type answer...
110
Multiple Choice
Mathew has a filtration kit, which consists of a funnel, a flask, and filter papers. Which of these mixtures can he separate using filtration?
111
Multiple Select
Check all the sentences based on whether they describe the properties of a heterogeneous mixture.
Its components are visible.
It may be mistaken for a pure substance.
It can be separated using distillation.
Concrete is an example of this kind of mixture.
112
Multiple Choice
Which unit is commonly used to measure density?
A. gcm3
B. g/cm3
C. cm3/g
D. g/cm
113
Multiple Choice
An alloy is made up of two kinds of metals. This alloy is uniform in appearance. How would this material be classified?
A. substance
B. homogeneous mixture
C. heterogeneous mixture
D. compound
114
Multiple Choice
An electric iron is used to remove wrinkles from clothing. The electric iron is made of metal. Which property of this substance is most useful for this application?
A. boiling point
B. density
C. electrical conductivity
D. thermal conductivity
115
Multiple Choice
What is a substance?
A. a uniform mixture that can’t be separated
B. a mixture that can be separated
C. a single component that can’t be separated
D. a single component that can be separated
116
Multiple Choice
Daniel has a sample of pure copper. Its mass is 89.6 grams (g), and its volume is 10 cubic centimeters (cm3). What’s the density of the sample?
A. 0.11 g/cm3
B. 8.96 g/cm3
C. 11.1 g/cm3
D. 896 g/cm3
117
Multiple Choice
Erica is working in the lab. She wants to remove the fine dust particles suspended in a sample of oil. Which method is she most likely to use?
A. reverse osmosis
B. osmosis
C. filtration
D. dilution
118
Multiple Choice
Which of the following property does not describe a compound:
it is composed of two or more elements
it is a pure substance
constituents cannot be separated by physical means
mixed in any proportion by mass
119
Multiple Choice
Suspension can be separated by filtration.
TRUE
FALSE
120
Multiple Choice
Why does a solid change to liquid when heat is added?
A. The spacing between particles decreases.
B. Particles lose energy.
C. The spacing between particles increases.
D. The temperature decreases.
121
Multiple Choice
Why does ice float on water?
A. The temperature of ice is lower than the temperature of water.
B. The temperature of water and ice are the same.
C. The density of ice is less than the density of water.
D. The density of ice is greater than the density of water.
122
Multiple Choice
When a perfume bottle is opened, some liquid changes to gas and the fragrance spreads around the room. Which sentence explains this?
A. Gases do not have a definite volume.
B. The particles of gases are closely packed.
C. The force of attraction between particles in gases is very high.
D. Gases are not matter.
123
Multiple Choice
An iron nail is made up of particles. What is true about the particles?
A. They move all over the place.
B. The force of attraction between them is small.
C. The spacing between them is large.
D. They stay in place and vibrate.
124
Multiple Select
Select the correct answer from each drop-down menu.
When a liquid is cooled, the kinetic energy of the particles __________________ , and the matter changes its state to solid.
decreases
increases
remains the same
125
Multiple Select
Select the correct answer from each drop-down menu.
When a liquid is cooled, the force of attraction between the particles ________________ , and the matter changes its state to solid.
decreases
increases
remains the same
126
Multiple Select
Select the correct answer from each drop-down menu.
When a liquid is cooled, the space between the particles ______________.
, and the matter changes its state to solid.
decreases
increases
remains the same
127
Multiple Choice

Identify the arrows that show removal of thermal energy when matter changes state.
Arrows to the left
Arrows to the right
128
Multiple Choice
129
Multiple Choice
130
Multiple Choice
131
Multiple Choice

132
Multiple Choice
133
Multiple Choice
134
Multiple Choice
135
Multiple Choice

136
Multiple Choice
What is an example of sublimation?
Ice cream melting
Dry ice being added to water
Frost forming on grass
137
Multiple Choice
A phase change from liquid to gas is called ...
condensation
evaporation
sublimation
deposition
138
Multiple Choice

139
Multiple Choice
140
Multiple Choice
141
Multiple Choice
142
Multiple Choice
143
Multiple Choice
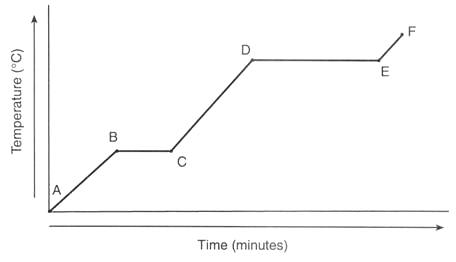
Is the substance gaining or losing energy?
Gaining
Losing
144
Multiple Choice

Are the particles moving faster or slower as time goes on?
Faster
Slower
145
Multiple Choice

What state/phase is the object likely from points A to B?
Solid
Liquid
Gas
Plasma
146
Multiple Choice

What is happening to the particles in the substance between points A and B?
Melting
Freezing
Speeding Up
Slowing Down
147
Multiple Choice

What is happening to the particles in the substance between points D and E?
Melting
Freezing
Vaporizing
Sublimating
148
Multiple Choice

Is the substance gaining or losing energy?
Gaining
Losing
149
Multiple Select

Where on the graph are phase changes taking place?
1
2
3
4
5
150
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of sulfur atoms is ___?
1
2
4
8
151
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of oxygen atoms is _____?
1
2
4
8
152
Multiple Choice
When sodium reacts with chlorine, sodium chloride is produced.
Andrew represented this reaction with this equation:
Na + Cl2 → 2NaCl
The equation is _____________.
balanced
not balanced
153
Multiple Choice
When sodium reacts with chlorine, sodium chloride is produced. Andrew represented this reaction with this equation:
__Na + __Cl2 →__NaCl
Which set balances the equation?
1,1,1
1,2,1
1,1,2
2,1,2
154
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of carbon (C) on each side of the equation?
2
4
6
8
155
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of oxygen (O) on each side of the equation?
2
13
15
26
156
Multiple Choice
When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in this equation:
2C4H10 + 13O2 → 8CO2 + 10H2O
How many atoms of hydrogen (H) on each side of the equation?
10
12
20
26
157
Multiple Choice
Iron reacts with oxygen to produce iron oxide (rust). This reaction is represented in an equation as 4Fe + xO2 → 2Fe2O3.
Identify the value of x that will balance the equation.
1
2
3
4
158
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
carbon
1
3
5
7
No Answer
159
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
hydrogen
1
3
5
7
No Answer
160
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
sulfur
1
3
5
7
No Answer
161
Multiple Choice
The chemical formula for artificial sweetener is C7H5NO3S.
Match the elements with the numbers found in one molecule of artificial sweetener.
Oxygen
1
3
5
7
No Answer
162
Multiple Select
Which chemical equations are being balanced ?
CaO + 3C → CaC2 + CO
Na + H2O → 2NaOH + H2
4Fe + O2 → 2Fe2O3
2Mg + O2 → 2MgO
163
Multiple Choice
Calcium carbonate is a compound that is used to make chalk for drawing on rocks or pavement. One molecule has one atom of calcium (Ca), one atom of carbon (C), and three atoms of oxygen (O). Which is the correct way to write the chemical formula for calcium carbonate?
3CaCO
Ca3CO
CaCO3
Ca3C3O3
164
Multiple Choice

The picture represents sulfur trioxide.
Which statement is true for one molecule of sulfur trioxide?
There are three atoms of sulfur and one atom of oxygen.
There are three atoms of sulfur and three atoms of oxygen.
There is one atom of sulfur and one atom of oxygen.
There is one atom of sulfur and three atoms of oxygen.
165
Multiple Choice
If blue represents one atom of nitrogen, and red represents one atom of oxygen, which picture represents 2NO2?
166
Multiple Choice
Sodium bicarbonate is the chemical name for baking soda. Its chemical formula is NaHCO3. What does the subscript 3 mean?
There are three molecules of sodium bicarbonate.
There are three molecules of oxygen in sodium bicarbonate.
There are three atoms of oxygen in sodium bicarbonate.
There are three atoms of sodium bicarbonate.
167
Multiple Choice
The chemical formula for sulfuric acid is H2SO4.
In 2H2SO4, the number of hydrogen atoms is_____?
1
2
4
8
Integrated Science Final Reveiw
by David Blankenship
Show answer
Auto Play
Slide 1 / 167
SLIDE
Similar Resources on Wayground

166 questions
Regents Biology Marathon Runner
Presentation
•
10th Grade

155 questions
Review#1
Presentation
•
9th - 12th Grade

171 questions
Greek Gods and Goddesses 1.0: 2025-2026
Presentation
•
9th Grade - University

153 questions
JC2 09-19-25
Presentation
•
9th - 12th Grade

153 questions
ULTIMATE STAAR REVIEW
Presentation
•
9th Grade

148 questions
APES Unit 5 Videos
Presentation
•
9th - 12th Grade

157 questions
Earth Science SSA Review
Presentation
•
6th - 8th Grade

169 questions
Nuclear Power
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade