

Errors and Significant Figures
Presentation
•
Chemistry
•
11th - 12th Grade
•
Medium
Standards-aligned
Angela Toh
Used 7+ times
FREE Resource
28 Slides • 56 Questions
1
By Angela Toh
Errors and Uncertainties
2

3

4
Multiple Choice
A measurement result is considered ________ if it is judged to be close to the true value.
Precise
Accurate
An error
Resolution
5

6
Multiple Choice
Which of the following are characteristics of random errors?
Doing several trials and finding the average will minimize them
The observed results will usually be consistently too high, or too low
Proper design of the experiment can eliminate them
There is no way to know what they are
7

8

9
Multiple Choice
Which of the following is a correct statement about systematic errors?
They arise due to incorrect judgment of a measurement.
Their magnitude depends on the experimenter’s skill.
They are consistent in magnitude.
They occur as a result of a person’s inability to perform the experiment correctly.
All of the above
10

11
Multiple Choice
A student is measuring the length of a set of pipes. Unfortunately his ruler has been damaged and starts at 0.2cm, which he fails to notice. This damage causes:
Random and systematic errors
Random error
Systematic error
Neither random or systematic error
12
Multiple Choice
True or False?
Zero error are random errors.
True
False
13
Multiple Select
Which of the following errors as are Random errors?
Student A uses same spatula for all chemicals and does not clean it
between use.
Student B takes only one titration reading.
Student C only notices that the burette has an air bubble after the
experiment.
Student D notices that her thermometer is not properly calibrated.
Student E measures the mass of sodium carbonate on the electronic
balance while the fan is switched on.
14
Multiple Select
Which of the following errors as are Systematic errors?
Student A uses same spatula for all chemicals and does not clean it
between use.
Student B takes only one titration reading.
Student C only notices that the burette has an air bubble after the
experiment.
Student D notices that her thermometer is not properly calibrated.
Student E measures the mass of sodium carbonate on the electronic
balance while the fan is switched on.
15

16

17
Multiple Choice
True or False?
Repeating a reading never removes the systematic error.
True
False
18
Multiple Choice
What could reduce systematic errors?
averaging a large number of measurements
careful calibration of measuring instruments
reducing the sample size
repeating measurements
19

20

21

22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26

27

28

29

30

31

32
Multiple Choice

33
Multiple Choice

34
Multiple Choice

Which Student is the most Precise?
Alex
Shandra
Luis
35
Multiple Choice

36
Multiple Choice
37
Multiple Choice

38
Multiple Choice

39
Multiple Choice

40
Multiple Choice

41
Multiple Choice

42
Multiple Choice

This bullseye demonstrates...
High Accuracy & High Precision
High Accuracy & Low Precision
Low Accuracy & High Precision
Low Accuracy & Low Precision
43
Multiple Choice
Which is the more precise measurement?
4 mL
4.3 mL
4.30 mL
4.300 mL
44

45
Fill in the Blanks
Type answer...
46
Multiple Choice
A student's measured length during a science experiment was 12.00 cm. The actual size was 14.25 cm.
What was the percent error?
*Hint: You now know how to calculate error, and you should know how to calculate a percent, so put the two together to find this answer.
15.79%
18.75%
2.25%
47
Multiple Choice
Jessie estimates the weight of her cat to be 8 pounds. The actual weight of the cat was 10 pounds.
What was the percent error?
*Hint: You now know how to calculate error, and you should know how to calculate a percent, so put the two together to find this answer.
15%
20%
25%
30%
48
Multiple Choice
Measurements: 24.9, 25.2, 25.1, 24.8
True Value: 25.0
These measurements are…
Accurate, but not precise
Precise, but not accurate
Both precise and accurate
Neither precise nor accurate
49

50
Multiple Choice
51
Multiple Select
A student’s calculation was found to have a 35.5% error, and his experimental measurement was 15.6 cm. What are the possible values for the actual measurement?
5.538 cm
21.138 cm
10.062 cm
51.1 cm
52
Multiple Choice
What kind of error is Parallax error or the viewing consistently from the wrong angle for all readings?
Systematic errors
Random errors
Both systematic and random errors
Neither systematic nor random errors
53
Multiple Choice
What errors occur in time measurements by a clock that runs too fast or slow?
Systematic errors
Random errors
Both systematic and random errors
Neither systematic nor random errors
54
Multiple Choice
True or False?
Repeating a reading never removes the systematic error.
True
False
55
Multiple Choice
Repeated measurements of a quantity can reduce the effects of
both random errors and systematic errors
neither random errors nor random errors
random errors
systematic errors
56
Multiple Choice
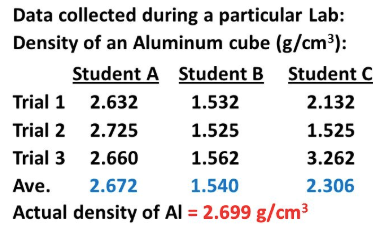
Which student is the most ACCURATE?
A
B
C
57
Multiple Choice
Systematic errors lead to a lack of:
accuracy in the measurement.
significant digits in the measurement.
precision in the measurement.
gradation of the measuring instrument
58
Multiple Choice
Random errors lead to a lack of:
accuracy in the measurement.
significant digits in the measurement.
precision in the measurement.
gradation of the measuring instrument
59

60

61

62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
0.000008
67
Multiple Choice
100000000
68
Multiple Choice

What is the mass and uncertainty indicated by this electronic balance?
198.61 ± 0.01 g
198.61 ± 0.02 g
198.61 ± 0.05 g
198.61 ± 0.005 g
69
Multiple Choice
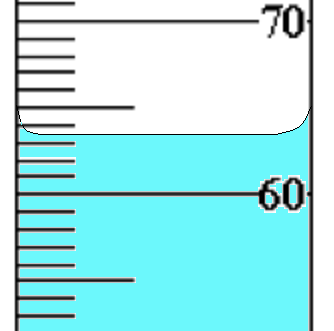
Based on the markings of this graduated cylinder, what is the estimated volume?
63.5 cm3
63 cm3
63.55 cm3
0.063 cm3
70

71

72

73
Multiple Choice
In this problem 2(9.8 cm +12.53 cm) = the answer should be.
44.0
44.7
44.6
44.66
45.00
74
Multiple Select
If the precision of your answer is specified, you should report your answer to that level of precision. However, if the required precision of the answer is not given (specified), what are the things to consider when deciding the number of digits to report (precision) when reporting a calculated value?
Common sense
The most precisely measured value used
Accepted convention
The targeted rule
The least precisely measure value used
75
Multiple Choice

76


77

78
Multiple Choice
The correct result (indicating the proper number of significant figures) of the following addition is ________.
12 + 1.2 + 0.12 + 0.012
13
13.3
13.33
13.332
79
Fill in the Blanks
Type answer...
80
Fill in the Blanks
Type answer...
81
Fill in the Blanks
Type answer...
82
Fill in the Blanks
Type answer...
83
Fill in the Blanks
Type answer...
84
Fill in the Blanks
Type answer...
By Angela Toh
Errors and Uncertainties
Show answer
Auto Play
Slide 1 / 84
SLIDE
Similar Resources on Wayground

78 questions
Cellular respiration and photosynthesis
Presentation
•
12th Grade

79 questions
Part 2 Mosby's Federal Laws
Presentation
•
12th Grade

79 questions
The Civil War
Presentation
•
11th Grade

78 questions
SOLUBILITY
Presentation
•
11th Grade

78 questions
Coulombic Attraction
Presentation
•
10th - 12th Grade

80 questions
DB014 Chapter 5: Expression of Biological Information
Presentation
•
11th Grade - University

78 questions
joints
Presentation
•
12th Grade

78 questions
Revision For August exam(5A)
Presentation
•
12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade

36 questions
Unit 4 (Part 2) Solutions
Quiz
•
10th - 12th Grade

30 questions
Final Exam Prep S22: 2.1.A.1 Isotopes
Quiz
•
9th - 12th Grade

25 questions
Stoichiometry Mass to Mass
Quiz
•
10th - 11th Grade

15 questions
Calculating pH and pOH
Quiz
•
10th - 12th Grade

15 questions
Balancing Chemical Equations
Quiz
•
10th - 12th Grade