

Ions and Isotopes
Presentation
•
Science
•
8th - 12th Grade
•
Medium
Standards-aligned
B Taylor
Used 99+ times
FREE Resource
7 Slides • 18 Questions
1
Ions and Isotopes
Ms. Taylor
2

An atom is a neutral particle.
Atoms have the equal number of positive proton to negative electrons.
Atoms are identified based on the number of protons they contain.
Atoms of the same element have the same number of protons
Atoms
3
Multiple Choice
4
Multiple Choice

What is the mass number of this Neon atom?
10
11
21
20
5
Multiple Choice
The atomic number is equal to _.
the number of protons
the number of neutrons only
the total number of protons and neutrons
the number of protons, neutrons, and electrons
6

Isotopes are atoms of the same element that have the same number of protons but different number of neutrons.
They therefore have the same atomic number but different mass number.
Most elements exist as isotopes. Usually written as symbol-mass # Ex. C-12
Look for similar proton number to identfy isotopes
ISOTOPES
7


All have atomic number of 1.
Each have a different mass number
Hydrogen Isotopes
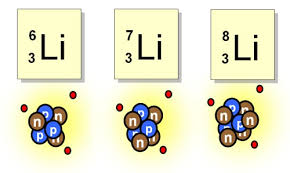
All have atomic number of 3.
Each have a different mass number
Lithium Isotopes
The Isotopes are determined by the number of Neutrons they contain.
One of the isotopes will be more common than the others.
The atomic masses in the Periodic table is an average mass based on the Percent of each in nature.
Ex. H-1 is the most common. The mass of H is periodic table is closer to 1.
With isotopes:
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice

12

All atoms want to have a 'full' valence (outer) shell to be stable.
To get this, they either lose or gain electrons and so are no longer neutral. They are charged.
An ion is a charged atom that has an unequal number of protons and electrons.
The charge depends on if they gained or lost electrons
IONS
13

When atoms lose electrons they now have more positively charged Protons in their nucleus than negatively charged electrons.
They therefore have excess positive charge and so become positive. Charge based on how many more protons than electrons they have.
Cations are positively charged ions.
Cations
When atoms gain electrons they now have more than negatively charged electrons than positively charged Protons in their nucleus .
They therefore have excess negative charge and so become negative. Charge based on how many more electrons than protons they have.
Anions are negatively charged ions.
Anions
14
Examples:
Fluorine (F) has 7 valence electrons. It gains 1 electron to be form F- ion.
Oxygen (O) has 6 valence electons and so gains 2 electrons to form O2- ion.
ANION
Sodium (Na) has 1 valence electron. It loses 1 electron and so forms ion Na+
Calcium (Ca) has 2 valence elctrons. It loses 2 electrons t form ion Ca2+
CATION
15
Multiple Choice

16
Multiple Choice

17
Multiple Choice
An atom that has gained or lost electrons in an attempt to become stable/bond with another atom.
Normal/Neutral Atom
Ion
Isotope
18
Multiple Choice
If an atom has 9 protons and 10 electrons it has a charge of ____.
0
-1
+1
-2
19
Multiple Choice
If an atom has 4 protons and 2 electrons it has a charge of ____.
0
-2
+2
+1
20
Multiple Choice
What is a representation of an ion?
Li
Na-22
O-2
21
Multiple Choice

What type of atom is this?
Neutral atom
Cation
Anion
Isotope
22
Multiple Choice

How many electrons does this magnesium ion have?
24
12
10
14
23
Multiple Choice
Which particle changes in an ion?
The number of electrons
The number of protons
The number of neutrons
24
Multiple Choice

What does it happen to this atom?
It LOSES electrons
It GAINS electrons
It GAINS protons
It LOSES protons
25
Multiple Choice

What type of atom is element C?
Normal atom
Ion
Isotope
Ions and Isotopes
Ms. Taylor
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

15 questions
Work, Energy, & Power Review
Presentation
•
8th - 12th Grade

23 questions
Energy in an Ecosystem
Presentation
•
8th - 12th Grade

21 questions
Mean, Median, Mode
Presentation
•
8th - 12th Grade

20 questions
Solving equations
Presentation
•
9th - 12th Grade

20 questions
Properties of Water
Presentation
•
9th - 12th Grade

19 questions
Target Heart Rate
Presentation
•
8th - 12th Grade

20 questions
Mr. Bradley's Tools of Science Exam
Presentation
•
9th - 12th Grade

20 questions
Big Bang Review
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade