

Ch. 6 L. 1 Atoms, Elements & Compounds
Presentation
•
Science, Biology, Chemistry
•
7th - 12th Grade
•
Medium
D Olguin
Used 1+ times
FREE Resource
37 Slides • 82 Questions
1
Ch. 6 L. 1 Atoms, Elements & Compounds
By D Olg

2

3

4

5

6

7

8

9
Multiple Choice
The smallest particle that makes up matter.
Atoms
Elements
Molecules
Compounds
10
Multiple Choice

11
Multiple Choice
Atoms are made up of __________________________.
germs, red bloods cells, and liver
elements
molecules and mixtures
protons, neutrons, & electrons
12
Multiple Choice
A neutron has a _________ charge.
negative
positive
neutral
13
Multiple Select
Which two parts of the atom are in the nucleus?
protons and electrons
electrons and neutrons
neutrons and protons
neutrons and elements
14
Multiple Choice
The atomic number of an element represents the ___________________.
number of molecules
number of protons
number of electrons
number of neutrons
15
Multiple Choice
What is a proton?
A part of an atom with a positive charge goes in the center of the atom(nucleus).
A molecule
An atom
A compound
16
Multiple Choice
What is an electron?
A proton
A part of an atom with a negative charge, goes in the outer shells
A neutron
An atom
17
Multiple Choice
What is a neutron?
An element
Atomic mass
A mixture
A part of an atom with no charge, goes in the center of the atom(nucleus).
18
Multiple Choice
What is an atom made of?
Elements
Compounds
Electrons, Neutrons and Protons
Mixtures
19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
22

23

24

25

26

27

28

29

30

31
Multiple Choice

This picture represents which of the following?
Element
Compound
Molecule
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice
What is a compound?
Two or more different elements bonded together.
Two of the same elements bonded together.
A mixture of different substances.
Two substances stuck together with glue.
35
Multiple Choice
Lithium-6 and lithium-7 are examples of
compounds
neutrons
isotopes
protons
36
Multiple Choice
Is H2 an element or a compound?
element
compound
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
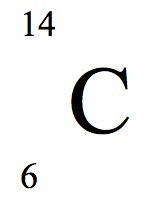
43
Multiple Choice
44
Multiple Choice

45
Multiple Choice

46
Multiple Choice

47
Multiple Choice

This picture represents which of the following?
Element
Compound
Molecule
48
Multiple Choice
A substance whose molecules contain DIFFERENT elements is call a ___________.
compound
atom
element
proton
49
Multiple Select

This picture represents which of the following?
Element
Compound
Molecule
50

51

52

53

54

55

56
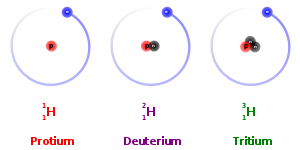
Multiple Choice
What is the correct Lewis Dot Structure for ammonia NH3
57
Multiple Choice

By replacing the element symbol, this could be a diagram of which element?
Li
Al
C
Be
58
Multiple Choice
According to the Octet Rule, atoms of elements react with each other in order to attain ____ electrons in their outermost energy level or shell.
2
4
6
8
10
59
Multiple Choice
Which of the following elements GAINS 1 electron in order to attain an octet?
sodium
calcium
helium
boron
chlorine
60
Multiple Choice
Which of the following elements GAINS 2 electrons in order to attain an octet?
cesium
magnesium
phosphorus
sulfur
iodine
61
Multiple Choice
What is the best definition of valence electrons?
The outermost electron NOT involved in chemical reactions
The innermost electrons involved in chemical reactions
The outermost electrons involved in chemical reactions.
The innermost electrons NOT involved in chemical reactions
62
Multiple Choice
How do can atoms complete the octet rule?
They lose or gain protons
They lose or gain neutrons
They lose or gain electrons in the outer most level
They lose of gain electrons from the inner most level
63
Multiple Choice
Metals tend to _______________ electrons and form __________ ions. These ions are called ______________.
gain, negative, anions
lose, positive, cations
gain, negative, negatrons
lose, positive, positrons
64
Multiple Choice
Anions have a ________ charge.
positve
negative
neutral
65
Multiple Choice
Cations have a __________ charge.
positive
negative
neutral
66

67

68

69

70

71
Multiple Choice
Valence electrons are shared
Ionic bonds
Covalent bonds
72
Multiple Choice

What type of bonding does this picture show?
Ionic
Covalent
73
Multiple Choice

What type of bonding does this picture show?
Ionic
Covalent
74
Multiple Choice
75
Multiple Choice
The chemical bond between a metal and a non-metal will be a _____ bond.
metal
ionic
covalent
polar
76
Multiple Choice
The chemical bond between a non-metal and another non-metal will be a ________ bond.
metal
ionic
covalent
polar
77
Multiple Choice

In an ionic bond the metal wants to ______ electrons.
lose
gain
share
78
Multiple Choice

When a metal loses an electron(s) it becomes a ...
positive ion
negative ion
metalloid
nonmetal
79
Multiple Choice

In an ionic bond the non-metal wants to ______ electron(s).
lose
gain
share
80
Multiple Choice

When a non-metal gains an electron it becomes a...
positive ion
negative ion
metalloid
metal
81

82

83

84
Multiple Choice
Which is the correct description about van der Waals' forces?
They are the weakest of the intermolecular forces arising from instantaneous dipole - induced dipole interactions.
They are the strongest of the intermolecular forces arising from dipole - dipole interactions.
85
Multiple Choice
Water is made up of hydrogen and oxygen, two elements that were chemically combined. This makes water a(n)
atom
compound
element
mixture
86
Multiple Choice

Which of the diagrams below represent a mixture?
A
B
C
D
87
Multiple Choice
How are elements and compounds different?
A compound is represented by a chemical formula, and elements are represented by a chemical symbol.
Compounds are represented by chemical symbols of one letter, and elements are represented by symbols of two letters.
Elements are pure substances, and compounds are not.
Elements are always larger units of matter, and compounds are smaller.
88
Multiple Choice
How many elements are in the compound Fe2O3?
0
1
2
3
89
Multiple Choice
Hydrogen is considered an element, while hydrogen peroxide is considered a compound. This is because hydrogen peroxide is...
is used for cleaning wounds.
made of two different elements.
is a liquid.
useful to human beings.
90
Multiple Choice
Element or Compound? O6
Element
Compound
91
Multiple Choice
Element or Compound? H6O5
Element
Compound
92
Multiple Choice
What is matter?
anything that has mass and takes up space
anything that has volume and is solid
anything that you can see
93
Multiple Choice

Is this a compound or element?
compound
element
94
Multiple Choice
95
Explanation Slide...
This is the compound Carbon Dioxide (what we breathe out). It is made of the elements Carbon and Oxygen.

96
Multiple Choice
97
Explanation Slide...
Matter is EVERYTHING

98
Multiple Choice
99

100
Multiple Choice
101

102
Multiple Choice
103

104
Multiple Choice
105

106
Multiple Choice
107
Multiple Choice
108
Multiple Choice
109
Multiple Choice

This picture represents which of the following?
Element
Compound
Molecule
110
Multiple Choice

This picture represents which of the following?
Element
Compound
Molecule
111
Multiple Choice
112
Multiple Choice
Every compound is a molecule but not every molecule is a compound.
113
Multiple Choice

114
Multiple Choice

115
Multiple Choice

116
Multiple Choice
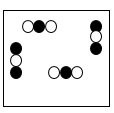
117
Multiple Choice
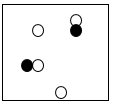
118
Multiple Choice
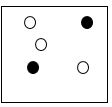
119
Multiple Choice
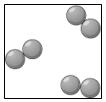
Ch. 6 L. 1 Atoms, Elements & Compounds
By D Olg

Show answer
Auto Play
Slide 1 / 119
SLIDE
Similar Resources on Wayground

108 questions
Heat Energy
Presentation
•
9th - 12th Grade

115 questions
Who - Which - That
Presentation
•
KG - University

119 questions
REview MK Logika Matematika
Presentation
•
9th Grade - University

109 questions
Analisis Data
Presentation
•
KG - University

119 questions
Biology Summary EOC Review
Presentation
•
9th - 12th Grade

113 questions
TNReady EOC Released Items
Presentation
•
9th - 12th Grade

115 questions
Semester 1 Final Exam Review
Presentation
•
8th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade