

AKS 5 Review
Presentation
•
Science
•
8th - 12th Grade
•
Medium
+1
Standards-aligned
Marissa Brown
Used 1+ times
FREE Resource
7 Slides • 33 Questions
1
AKS 5 Review
By Marissa Brown
2

3
Multiple Choice
Matter is _______________________________.
The amount of matter in an object.
The amount of space an object occupies.
Anything that has mass and volume.
4
Multiple Choice
Which of the following is NOT a part of the Kinetic Theory of Matter?
All matter is made up of tiny particles.
Particles are always moving in random paths
Occasionally, particles stop moving.
The type of movement of particles depends on the amount of energy.
5
Multiple Choice
Which of the following states of matter has the LOWEST level of energy?
Solid
Liquid
Gas
Plasma
6
Multiple Choice
The energy of an object as it is in motion is defined as
Potential Energy
Elastic Energy
Electric Energy
Kinetic Energy
7
Multiple Choice

What happens to the average kinetic energy of matter when it is heated?
It increases
It stays the same
It decreases
It cannot be determined
8
Multiple Choice

What state of matter is shown in the image?
Solid
Liquid
Gas
Thermal Energy
9
Multiple Choice
At what temperature does matter change phases from a solid to a liquid?
Melting Point
Boiling Point
Condensation Point
0 degrees Celsius
10
Multiple Choice
If the atoms of an object are moving fast, the temperature would be...
High
Low
11
Multiple Choice
When thermal energy is added to a substance, the substance's particles move:
More rapidly at an increased distance from each other.
More slowly with a greater distance between each other.
More rapidly with less distance between each other.
More slowly with a reduced distance between each other.
12
Multiple Choice

Which phase change occurs between points D and E if heat is added?
boiling
freezing
melting
condensing
13
Multiple Choice
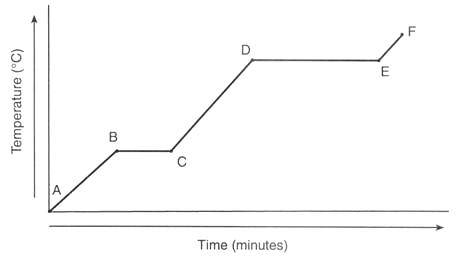
14
Multiple Choice

The melting point of the sample is
-60 ºC
-100 ºC
60 ºC
100 ºC
15
Multiple Choice

Given the heating curve of water, what is happening to potential and kinetic energy during segment CD?
the potential energy and kinetic energy both increase
the potential energy increases while the kinetic energy decreases
the potential energy is constant while the kinetic energy increases
the potential energy increases while the kinetic energy is constant
16
Multiple Choice

17
Multiple Choice

18
Multiple Choice
Volume is _______________________.
the amount of matter an object has.
the amount of space an object occupies.
anything that mass and volume.
19
Multiple Choice
Mass is ________________________________.
The amount of matter in an object.
Anything that has Mass and volume.
The amount of space an object occupies.
20
Multiple Select
A liquid has _______________ . (Pick 3)
A lot of space between particles.
Very little space between particles.
Particles that have have no definite pattern.
Particles hold that a very definite pattern.
Particles that freely flow past each other.
21
Multiple Select

A solid has _______________ . (Pick 3)
A lot of space between particles
Very little space between particles
Particles with no definite pattern.
Particles that hold a very definite pattern.
Particles that only vibrate in place.
22
Multiple Select

A gas has _______________ . (Pick 3)
A lot of space between particles
Very little space between particles
Particles with no definite pattern.
Particles that hold a very definite pattern.
Particle that bounce all over the place.
23
AKS 5B - Gas Laws

24
25
26
27
Multiple Choice
28
Multiple Choice

29
Multiple Choice
30
Multiple Choice
Which measurement measures the amount of energy in a substance?
Temperature
Volume
Pressure
Mass
31
Multiple Choice
According to Charles Law, if Temperature of a gas increases Volume will....
Increase
Decrease
Stay the Same
32
Multiple Choice
A container of gas is held at a constant temperature. What is the most reasonable prediction of the graph of pressure and volume for a gas in this situation?
33
Multiple Choice
34
35
Multiple Choice
Relate the temperature of a gas to the kinetic energy of its particles.
As the temperature extends, the kinetic energy of the particles decreases.
As the temperature increases, the kinetic energy of the particles increases.
As the temperature decreases, the kinetic energy of the particles increases.
As the temperature intensifies, the kinetic energy of the particles decreases.
36
Multiple Choice
Jayden was given a marshmallow and a syringe in class to experiment with. She placed the marshmallow in the syringe and sealed the end. When she depressed the plunger of the syringe what happened to the marshmallow and why?
The increased pressure, pushed the molecules closer together, and caused the marshmallow to shrink.
The decreased pressure, pushed the molecules closer together, and caused the marshmallow to shrink.
The increased pressure, allowed the molecules to spread apart, and caused the marshmallow to grow larger.
The decreased pressure, allowed the molecules to spread apart, and caused the marshmallow to grow larger.
37
Multiple Choice
Three balloons are inflated to the same size. The three balloons are submerged in buckets filled with ice water, tap water and water heated to 65°C. What results would you expect from this experiment?
All the balloons would shrink.
All the balloons would expand.
The balloon in the ice water would expand.
The balloon in the 65°C water would expand.
38
Multiple Choice
How is the volume of a gas related to its concentration of particles?
As the volume of a contained gas expands, its pressure concentration of particles will increase.
As the volume of a contained gas extends, its pressure concentration of particles will increase.
As the volume of a contained gas decreases, its pressure concentration of particles will increase.
As the volume of a contained gas compresses, its pressure concentration of particles will decrease.
39
Multiple Choice
How is the pressure of a gas related to its concentration of particles?
Pressure will expand a gas, enlarging its volume and reducing its density and concentration of particles.
Pressure will magnify a gas, developing its volume and multiplying its density and concentration of particles.
Pressure will compress a gas, reducing its volume and giving it a greater density and concentration of particles.
Pressure will accelerate a gas, extending its volume and allowing a smaller density and concentration of particles.
40
Multiple Choice
Tamika inflates and ties off a balloon in her classroom and then takes it outside on a cold, winter day. Immediately, she notices that the balloon has gotten a little bit smaller. Why does this occur?
because there are fewer molecules in the balloon outside
because the air pressure is significantly higher when it is colder
because the molecules in the balloon have slowed down with the temperature
because the molecules shrink in size when exposed to the colder, outside air
AKS 5 Review
By Marissa Brown
Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

37 questions
Energy and Energy Transformations
Presentation
•
9th - 12th Grade

35 questions
MS-PS4-1: Wave Energy and Properties
Presentation
•
8th Grade - University

36 questions
Marine Science Midterm Review Part 2
Presentation
•
8th - 12th Grade

34 questions
Ionic and Covalent Bonding
Presentation
•
9th - 12th Grade

32 questions
Physical and Chemical properties -Study guide
Presentation
•
7th - 12th Grade

36 questions
Levels of Organization - Cells to Biome
Presentation
•
KG

36 questions
Mitosis (part 2)
Presentation
•
7th - 12th Grade

36 questions
Lab Safety Rules
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade