

Chemistry Review for Final 2: Atomic Structure and Models
Presentation
•
Science, Chemistry
•
11th Grade
•
Medium
+1
Standards-aligned
William Jared Lovering
Used 10+ times
FREE Resource
50 Slides • 14 Questions
1
Chemistry Review for Final 2: Atoms, Ions, and Isotopes
By William Lovering
2
There have been many atomic models over the ages
Billiard Ball - Dalton
Tiny Real-Life - Democrats
Plumb Pudding - Thomson
Planetary - Rutherford
Energy Level - Bohr
Quantum - Schrodinger

3
Atomic Models
A visual representation of the structure of an atom, (how an atom looks and made-up of)
Nucleus
Inside Nucleus: Protons (positive charge) and Neutrons (no charge)
Outside Nucleus: Electrons (negative charge)

4

5
Dalton
Was the first modern scientist to propose the atomic theory.
He thought that atoms were made of microscopic hard balls where every atom of an element had the same properties (for example: radius and mass)

6
Problems with Dalton's Model
In experimentation with different gases scientists, most notably J.J. Thomson, discovered positive and negative charged particles in the gas.
Dalton's Model didn't account for this
7

8

9

10

11

12

13

14

15

16

17

18

19

20

21

22

23

24

25

26
Atomic Number, Average Atomic Mass, Mass Number, Isotopes

27

28

29

30

31

32

33
Isotope Symbols
As you can see to the right there are certain ways elements and isotopes are represented as symbols. There is the element symbol (the biggest part). Then the upper number will the the mass number (number of protons + number of neutrons) and the lower number is just the atomic number (number of protons).

34
Isotope Symbol Examples

35
ISOTOPES:
Same Element
Different Mass Number
WHICH MEANS DIFFERENT # of NEUTRONS

36
What does changing # of Neutrons do?
Neutrons make atoms stable
Unstable = Wants to fall apart
Fall Apart = Split into smaller atoms
Split Apart = RADIOACTIVE

37
Average Atomic Mass
DIFFERENT than Mass Number
Average of all of the isotopes of an atom found in nature.

38
How they get that number
% abundance of that isotope x actual mass of that isotope
add together
divide by 100 (if you didn't put into percentage first)

39
How to tell which isotope is more abundant?
CLOSEST TO ROUNDED AVERAGE ATOMIC MASS
So the most abundant isotope of Gold would be?
Gold - 197

40
Orbitals
a region of probability where an electron can be found.
s
d
p
f
Remember these are NOT electrons, they're just regions of space where an electron can be found.(Each can hold up to 2 electrons)

41
The 4 Quantum Numbers

42
Principle Quantum Number
n=1
n=2
n=3
n=4

43
Pauli Exclusion Principle
No two electrons in the same atom can have identical values for all four of their quantum numbers.
In other words:
(1) no more than two electrons can occupy the same orbital
(2) two electrons in the same orbital must have opposite spins

44
Aufbau Principle
states that electrons fill lower-energy atomic orbitals before filling higher-energy ones (Aufbau is German for "building-up")

45
Hunds Rule
every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.

46
Orbital Configuration
the more visual way to represent the arrangement of all the electrons in a particular atom
Each sublevel is labeled by its principal energy level and sublevel
Electrons are indicated by arrows inside the circles. An arrow pointing upwards indicates one spin direction, while a downward pointing arrow indicates the other direction.

47
Electron Configuration
Electron configuration notation eliminates the boxes and arrows of orbital filling diagrams.

48
How to Write an Electron Configuration:
1. Start with the shell/period number
2. Followed by the type of orbital
3. Finally the superscript indicates the number of electrons in the orbital
Example: B = 1s2 2s2 2p1

49

50

51
Multiple Choice

This model was developed after J.J. Thompson discovered electrons, a particle smaller than an atom. It shows electrons floating freely in a positive region.
The "Plum Pudding Model" of the atom
The "Rutherford Model" of the atom
Democritus's model of the atom
The "Quantum Mechanical Model" of the atom
52
Multiple Choice

Which has more electrons?
neither
carbon - 12
carbon - 13
53
Multiple Choice

Choose the correct electron configuration for Nickel (Ni).
1s2 2s2 2p6 3s2 3p6 4s2 3d8
1s2 2s2 2p6 3s2 3p6 4s2 3d2
1s2 2s2 2p6 3s2 3p6 4s10
54
Multiple Choice

Choose the correct electron configuration for Krypton (Kr).
1s2 2s2 2p6 3s2 3p6 4s8 3d8
1s2 2s2 2p6 3s2 3p6 4s2 3d8 4p3
1s2 2s2 2p6 3s2 3p6 4s2 3d8 4p6
55
Fill in the Blanks

Type answer...
56
Multiple Choice

How many neutrons does this bromine atom have?
35
45
80
57
Multiple Choice
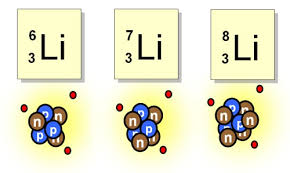
58
Multiple Choice

Which has more neutrons?
neither
carbon - 12
carbon - 13
59
Multiple Choice
In the gold foil experiment, most of the positively charged alpha particles passed through the gold foil, but some were deflected or bounced back. What did we conclude because of this?
Atoms are small indivisible spheres
Atoms are mostly empty space with a small, dense, positive center
Atoms have negatively charged particles which orbit the nucleus
Light is a wave, not a particle
60
Fill in the Blanks

Type answer...
61
Multiple Choice

Who is credited with developing model D?
Thomson
Dalton
Bohr
Rutherford
62
Multiple Choice
Which scientist is responsible for first stating that all the atoms of a particular element are identical but are different from the atoms of all other elements?
Dalton
Thomson
Aristotle
Democritus
Proust
63
Multiple Choice

Choose the correct electron configuration for Chlorine (Cl).
1s2 2s2 2p1
1s2 2s2 2p6 3s2
1s2 2s2 2p6 3s2 3p5
64
Multiple Choice
Chadwick is credited with discovering which part of the atom?
nucleus
protons
electrons
neutrons
Chemistry Review for Final 2: Atoms, Ions, and Isotopes
By William Lovering
Show answer
Auto Play
Slide 1 / 64
SLIDE
Similar Resources on Wayground

59 questions
Oliver Twist
Presentation
•
11th Grade

58 questions
Stomach and Intestines Semester Review
Presentation
•
11th - 12th Grade

61 questions
Federal Budget
Presentation
•
11th - 12th Grade

56 questions
Science Form 4 KSSM (Revision c9-12)
Presentation
•
12th Grade

59 questions
Hazards
Presentation
•
11th Grade

60 questions
Atomic Theory Notes
Presentation
•
11th Grade

57 questions
Untitled Lesson
Presentation
•
KG

58 questions
Unit 3: Chemical Bonds
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade