

PhySci Final Review 5: Matter
Presentation
•
Science, Chemistry
•
9th Grade
•
Medium
William Jared Lovering
Used 3+ times
FREE Resource
37 Slides • 36 Questions
1
PhySci Final Review 5: Matter
By William Lovering
2

3

4

5

6

7

8

9

10

11

12
Elements are represented on the Periodic Table by their own symbol...
Copper (Cu)
Oxygen (O2)
13
Elements
Pure substance
Made up of identical atoms
Cannot be separated into a simpler substance by physical or chemical means

14
Atoms
The basic building block of all matter.
Made up of protons (+), neutrons (0), and electrons (-).
Different atoms have different numbers of these energy particles

15

16
Compounds contain more than one element:
Copper oxide (CuO)
Methane (CH4)
17

18
Homogeneous Mixtures
blended evenly throughout
you CANNOT tell the different substances apart
more examples: creamy peanut butter, Gatorade, hand sanitizer
19

20
Heterogeneous Mixtures
you can look at the different substances and tell them apart
more examples: trail mix, crunchy peanut butter
21
Mixtures are NOT pure substances
Mixtures are not bonded together, therefore there is no chemical change. Each source of matter KEEPS its original properites!

22
23

24

25

26

27

28

29

30

31

32

33

34

35

36

37

38
Multiple Choice

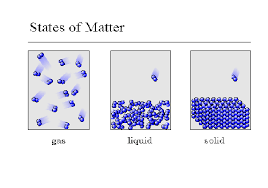
Which state of matter has both definite shape and definite volume?
Gas
Liquid
Solid
39
Multiple Choice

Which state of matter has no definite shape but does have definite volume?
Gas
Solid
Liquid
40
Multiple Choice
Matter is _______________________________.
The amount of matter in an object.
The amount of space an object occupies.
Anything that has mass and volume.
41
Multiple Choice
Mass is ________________________________.
The amount of matter in an object.
Anything that has Mass and volume.
The amount of space an object occupies.
42
Multiple Choice
Volume is _______________________.
the amount of matter an object has.
the amount of space an object occupies.
anything that mass and volume.
43
Multiple Choice
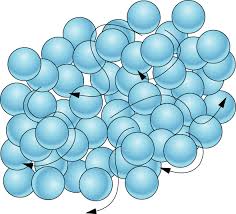
What state of matter?
Solid
Liquid
Gas
44
Multiple Choice
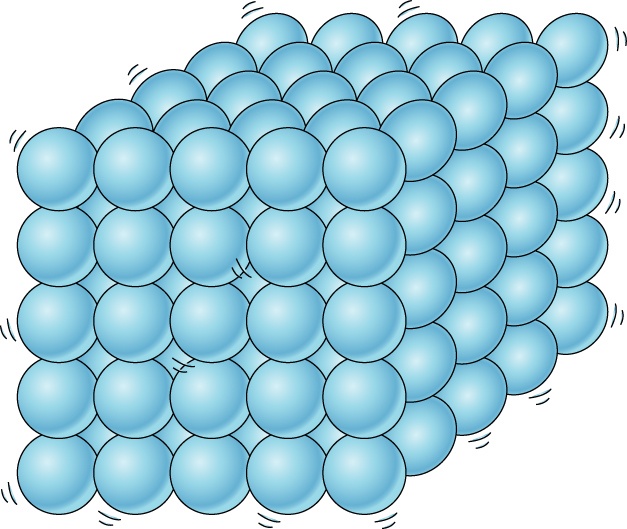
What state of matter?
Solid
Liquid
Gas
Plasma!!!
45
Multiple Choice
46
Multiple Choice
What is the phase change of a solid to a liquid?
freezing
melting
boiling
condensation
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice

50
Fill in the Blanks
Type answer...
51
Multiple Select
What are the two types of physical properties used to classify matter?
intensive
extensive
chemical
substance
gravity
52
Multiple Choice
flammability of a substance
physical change
physical property
chemical change
chemical property
53
Multiple Choice
54
Multiple Choice
sharpening a pencil
physical change
physical property
chemical change
chemical property
55
Multiple Choice
size of an object
physical change
physical property
chemical change
chemical property
56
Multiple Choice
inflating a tire
physical change
physical property
chemical change
chemical property
57
Multiple Choice
corrosion of a bicycle frame
physical change
physical property
chemical change
chemical property
58
Multiple Choice
formation of water when hydrogen burns
physical change
physical property
chemical change
chemical property
59
Multiple Choice
a burning candle
physical change
physical property
chemical change
chemical property
60
Multiple Choice
61
Multiple Choice

62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice

66
Multiple Choice
1 A substance made up of two or more chemically combined elements is —
an element
mixture
compound
solution
67
Multiple Choice
freezing point
physical change
physical property
chemical change
chemical property
68
Multiple Choice

69
Multiple Choice

This picture represents which of the following?
A single element
Mixture of molecules
mixture of atoms
mixture of compounds
70
Multiple Choice

What does this illustrate
Elements
Compunds
Mixture of Elements and Compunds
Mixture of Compounds
71
Multiple Choice
72
Multiple Choice

Which compound is best represented by this particle diagram?
C2H6
C6H12O6
CO2
NaCl
73
Multiple Choice

Which compound is best represented by this particle diagram?
NH3
H2O
CH4
HCl
PhySci Final Review 5: Matter
By William Lovering
Show answer
Auto Play
Slide 1 / 73
SLIDE
Similar Resources on Wayground

67 questions
Ecological Succession
Presentation
•
9th - 10th Grade

65 questions
Waves Properties
Presentation
•
9th Grade

68 questions
Byzantine Empire
Presentation
•
9th Grade

67 questions
Grade 10: Mirrors and Lenses
Presentation
•
10th Grade

66 questions
North America Physical Geography
Presentation
•
8th Grade

64 questions
C4 Electrochemistry
Presentation
•
9th Grade

65 questions
Physical Science 6.1 & Ionic Bonds Gizmo
Presentation
•
9th - 10th Grade

63 questions
Heat and Heat Transfer
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

10 questions
Exploring Newton's Laws of Motion
Interactive video
•
6th - 10th Grade