

Chemistry Final Exam Review Part 1
Presentation
•
Chemistry
•
10th Grade
•
Medium
+6
Standards-aligned

llanham summitt
Used 18+ times
FREE Resource
52 Slides • 107 Questions
1
Chemistry Final Exam Review Part 1
By llanham summitt
2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
11
Multiple Choice

This symbol indicates a _______.
neutron
proton
electron
12
Multiple Choice

What is the atomic mass for this element?
6
12.0107
6.0107
18.0107
13
Multiple Choice
The center of an atom is called
a/an __________.
atomic mass
nucleus
proton center
energy shell
14
Multiple Choice

How many neutrons are in an Calcium atom?
20
40.08
60
15
Multiple Choice
The atomic number of an element is determined by the _________ of ________ in the nucleus.
protons, neutrons
number, electrons
electrons, protons
number, protons
16
Multiple Choice

Which image shows a Bohr model for Carbon.
17
Multiple Choice
If the protons and electrons do not match, the atom is ____.
neutral
ion
18
Multiple Choice
An electron is
positively charged
neutral
negatively charged
19
Multiple Choice
atomic number is
the number of protons
the number of electrons
the number of neutrons
the location of an atom on the periodic table
20
Multiple Choice
21
Multiple Choice
22
Multiple Choice
Subatomic particles with a negative charge
Electrons
Neutrons
Protons
Quarks
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice
30
Multiple Choice

What is the atomic number?
35
18
17
52
31
Multiple Choice

How many protons and electrons does this element have?
35
18
17
52
32
Multiple Choice

What is the atomic mass of Neon?
10
20
30
33
Multiple Choice
How many valence electrons are found in atoms of group 4?
4
3
14
16
34
Multiple Choice
How many valence electrons are found in atoms of group 5A?
4
5
2
3
35
Multiple Choice
How many valence electrons are found in atoms of group 5A?
4
5
2
3
36
Multiple Choice
All atoms are most stable with (or would "prefer") how many electrons in their valence shell?
1
2
8
18
37
Multiple Choice
What groups are the most reactive non metals?
1
7A
2
6A
38
Multiple Choice
Why are halogens so reactive?
They want to get rid of their only valance electron
They only need one more electron
They are non reactive, they have a full shell
39
Multiple Choice
How do electrons of an element determine that element's reactivity?
If the valence shell has missing electrons it is very reactive to attract or lose more electrons.
If the valence shell has a full ring of electrons it is very reactive because it needs more.
If the valence shell has a full shell of electrons it is very reactive because it is already full.
If the valence shell has missing electrons it is not reactive because it is missing electrons.
40
Multiple Choice
41
Multiple Choice
The atom with the largest atomic radius in Group 8A is -
Ar
He
Kr
Rn
42
Multiple Choice
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
N or C?
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
Which of the following would have the smallest ionization energy?
K
P
S
Ca
Mg
50
Multiple Choice
Which of the following would have the largest ionization energy?
Na
Al
Mn
N
Pd
51
Multiple Choice
What is ionization energy?
The amount of energy to add an electron to an element
The amount of energy added to make a bond with a valence electron
The energy required to remove an electron
The energy added to promote an electron in a lower energy level to a higher energy level
52
Multiple Choice
Of the following metals, which will be the MOST reactive? (Hint: reactive metals have low ionization energies)
gold
cobalt
strontium
tungsten
53
Multiple Choice
54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
57
Multiple Choice
58
Multiple Choice
59
Chemistry I Final Exam Review Part 1
By llanham summitt
60

Timers are turned off so that you can relax and think about the questions and take your time.
Chemistry take thought and carefully reading.
Take you time and read carefully!
61
The periodic table. the polyatomic ion chart, the mole conversion chart and your calculator are available to you; USE THEM.
rely on your resources not GOOGLE or others.
Use your resources!

62
You can do this if you try!
Effort is an important part of anything that you do..
“The average person puts only 25% of his energy and ability into his work. The world takes off its hat to those who put in more than 50% of their capacity, and stands on its head for those few and far between souls who devote 100%.” ~ Andrew Carnegie
Where do you want to be?
63

64

65

66

67

68

69

70

71

72

73

74

75

76

77

78

79

80

81

82
Multiple Choice

What is the volume of a piece of zinc with measurements 5.0 cm x 5.0 cm x 5.0 cm?
15 cm3
125 cm3
15 cm
125 cm
83
Multiple Choice

What is the volume of the fluid in the graduated cylinder shown?
12.0 ml
13.0 ml
13.5 ml
12.5 ml
84
Multiple Choice

Which of the following is NOT a chemical property of sugar?
it decomposes readily
It is a white, crystalline powder
It has a composition of carbon, hydrogen, and oxygen
It will burn when heated
It does not conduct electricity so is non-ionic
85
Multiple Choice

1
10
100
1000
86
Multiple Choice

Matter is made of small indestructible particles called:
protons
electrons
atoms
neutrons
87
Multiple Choice

Matter takes up space or ______, and has mass or quantity, measured in ______.
area, grams
mass, milliliters
volume, grams
88
Multiple Choice

Different substances can often be identified by their density. Density is a derived unit equal to:
mass x volume
volume / mass
length x width x height
mass /volume
89
Multiple Choice

All of the following are physical changes except:
Melting
burning
condensing
vaporizing
90
Multiple Choice

If the graph represents the energy of the reactants and products of a chemical reaction, which of the following is true of the reaction?
there is an exothermic reaction
there is an endothermic reaction
the activation energy is lower than the energy released
the total energy is higher than the starting energy
91
Multiple Choice

Atoms in a _______ are closely packed vibrating particles.
plasma
liquid
solid
gas
92
Multiple Select

1 ml is the same as which af the following? (Mark all that apply)
1 cc
1 cm3
1 μl
1 dm3
93
Multiple Select

Increasing the temperature of a substance causes the particles to speed up and can cause which of the following to occur? (Multiple answers)
condensing
vaporizing
melting
boiling
94
Multiple Choice

Which is NOT a chemical property?
iron will react with oxygen
white phosphorus is very flammable
copper is malleable (can be bounded into sheets) and ductile (can be pulled into wire)
lead is toxic
95
Multiple Choice

Which is NOT a Chemical Change?
iron is oxidized to become rust (iron oxide)
carbon products are usually combustible
Decomposition is a necessary reaction
dissolving substances is often the 1st step in any procedure
96
Multiple Choice

There are seven naturally occurring diatomic elements; which of the following is NOT a diatomic element?
H
O
Cl
N
C
97
Multiple Choice

A _________ is a pure substance made of different elements that are chemically combined into a new substance.
mixture
ion
compound
98
Multiple Choice

A material that is made of different substances that are NOT chemically combined is called a(an)
compound
mixture
ion
99
Multiple Choice

In a solution, the substance that causes the dissolving is known as a ________
solute
solvent
suspension
colloid
100
Multiple Choice

_________ mixtures are evenly blended and appear to be the same throughout.
homogeneous
heterogeneous
101
Multiple Select

A mixture, like salt water, containing one substance dissolved in another, is known as a ____________ (mark all that apply)
homogeneous mixture
solution
suspension
colloid
102
Multiple Choice

The two main parts of an atom are the:
principle energy levels and sublevels
nucleus and energy levels
planetary electrons and energy levels
nucleus and kernel
103
Multiple Choice

The number of electrons in the 2nd principal energy level, n=2, of an atom is: 2n2
4
2
8
18
104
Multiple Choice

When the particles of a liquid are heated, they begin to expand and the phase changes to a _____. These particles then move in straight lines until they bounce off of something. When the particles cool back down to a liquid it is called _________
Gas, condensation
Plasma, condensation
Gas, Sublimation
Plasma, vaporization
105
Multiple Choice

An atom of beryllium contains 4 protons, 5 neutrons, and 4 electrons. What is the mass number of this atom?
13
9
8
5
106
Multiple Select

A substance that contains only one type of atom is known as a(n) ______ (choose all that apply)
compound
element
Pure substance
Mixture
107
Multiple Choice

A can of baking powder reads: "Ingredients- cornstarch, sodium bicarbonate, calcium acid phosphate, and sodium aluminum sulfate." Baking soda is a
compound
mixture of compounds
molecule
mixture of elements
108
Multiple Choice

A 220 g piece of metal was submerged in a graduated cylinder containing 50 ml of water. The level of the water rose to 150 ml. What is the density of the metal?
2.2 g/ml
0.45 ml/g
320 g/ml
20 g/ml
109

110

111

112

113

114

115

116

117

118

119

120

121
Chm.1.3.1 Classify the components of a periodic table (period, group, metal, metalloid, nonmetal, transition).
Periods
Groups
Valence Electrons

122

123
The Periodic Table
Group- Up and Down
Main group elements in the same group have similar properties, the same number of valence electrons, and the same
Period- Left to Right
Reactivity increases as you go down within a group for metals and decreases for nonmetals.
Valence Electron Cheat Code!

124

125
Element Classification
Representative Elements(main group)- A groups or as groups 1, 2, 13-18.
Transition elements as B groups or as groups 3-12.

126
Chm.1.3.2 Infer the physical properties (atomic radius, metallic and nonmetallic characteristics) of an element based on its position on the Periodic Table.
Periodic Trends
Electron Configuration

127
Electronegativity
A measure of the ability of an atom to attract the electrons.
Fluorine F is the most electronegative and Francium is the least electronegative.
Electronegativity is not measured in energy units, but is rather a relative scale
Increase L to R, this is due to an increase in nuclear charge
Electronegativities generally decrease from top to bottom within a group due to the larger atomic size.

128

129
Electron Configuration
Understanding Electron Configuration and Notation helps understand bonding and chemical reactions by indicating where electrons are located in a specific atom.
Hunds Rule
Aufubau's Principle
Pauli Exclusion

130
Hunds Rule
Every orbital in a sublevel is singly occupied before any orbital is doubly occupied.

131
Aufbau Principle
Electrons fill lower-energy atomic orbitals before filling higher-energy ones in the pattern shown. This pattern can be memorized or you can follow the Periodic table.









1s
2s & 2p
3s & 3 p
4s 3d & 4p
5s 4d & 5p
6s 4f 5d & 6p
7s 5f 6d & 7p
d orbitals drop & are 1 behind
f orbitals drop & are 2 behind
132

133
Pauli Exclusion Principle
States that, in an atom or molecule, no two electrons can have the same four electronic quantum numbers. As an orbital can contain a maximum of only two electrons, the two electrons must have opposing spins

134
Metallic Character vs Non Metallic
Metallic character refers to the level of reactivity of a metal; Metals tend to lose electrons in chemical reactions. Metallic character increases as you go down a group
Nonmetals tend to gain electrons in chemical reactions and have a high attraction for electrons within a compound. The most reactive NM: upper right portion of the periodic table

135

136
Multiple Select

Chlorine is represented by the electron dot structure shown in the picture. Which other atom(s) would have the same atomic dot structure? (Mark all that apply)
Iodine
Oxygen
Sodium
Fluorine
137
Multiple Choice
Elements in the same column of the periodic table always have the same # of _______ as one another.
Protons
Neutrons
Electrons
Valence Electrons
138
Multiple Choice
Which has the greater Electronegativity:
N or C?
C
N
139
Multiple Choice
Which group of the periodic table is composed of inert (not reactive) gases?
alkali metals
alkaline earth metals
halogens
noble gases
140
Multiple Choice
+ 2
- 2
+1
-1
141
Multiple Select

Which two statements are correct about the figure shown?
It is a covalent bond.
Hydrogen atoms loss electrons and Carbon gains the atoms.
It happens on metal and on another non-metal.
The valence electrons are shared by both atoms.
142
Multiple Choice
Ionic
Covalent
143
Multiple Choice

An electron dot diagram consists of the symbol representing the element and an arrangement of dots that usually shows
the atomic number
the atomic mass
the number of neutrons
the number of valence electrons
144
Multiple Choice

What element on the periodic table has 16 protons?
germanium Ge
phosphorus P
oxygen O
sulfur S
145
Multiple Choice

Which element easily gives up 1 electron to form a cation with +1 charge?
F
Cl
Mg
Li
146
Multiple Choice

Which atom will have the strongest affinity for electrons, becoming an anion with a -1 charge?
K
F
O
Fe
147
Multiple Choice

Where on the periodic table do you find elements that form the strongest bases?
left
right
middle
inert gases
148
Multiple Choice

If two atoms bond in such a way that one-member beomes a positive cation and the other a negative anion, what type of bond is formed?
ionic bond
covalent bond
polar bond
Vanderwaals bond
149
Multiple Choice

Isotopes are atoms with different numbers of ________.
protons
neutrons
electrons
150
Multiple Choice

The first scientist to show that atoms emit tiny negative particles was
J J Thomson
Ernest Rutherford
James Chadwick
Neils Bohr
151
Multiple Choice

The scientist whose alpha-particle scattering experiment led him to conclude that the nucleus of an atom contains a dense center of positive charge is
JJ Thomson
Ernest Rutherford
Neils Bohr
John Dalton
152
Multiple Choice

This scientist is responsible for the model that we use today to illustrate and help understand the atoms' energy levels.
JJ Thomson
Ernest Rutherford
Neils Bohr
John Dalton
153
Multiple Select
Which of the following parts of Dalton's atomic theory are no longer entirely correct? (Mark all that apply)
atoms are small indestructible particles
all atoms of an element are alike and different from atoms of other elements
compounds are made of elements combined in different ratios of elements
chemical reaction occur that rearrange elements but do not destroy them
154
Multiple Choice
An atom has 10 protons, 15 neutrons and 10 electrons what is its mass number.
20
10
35
25
155
Multiple Choice
12 neutrons
12 electrons
12 protons
24 protons and neutrons
156
Multiple Choice
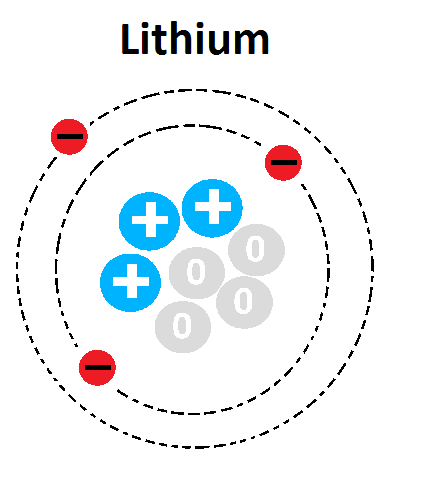
What is the mass number of this atom?
1
3
4
7
157
Multiple Choice
Which Bohr model represents Neon?
158
Multiple Choice
What atom matches this electron configuration?
1s2 2s2 2p6 3s2
Neon
Magnesium
Aluminum
Potassium
159
Part 1 Ends Here

Take a break. Part two contains formulas, balanced equations and mole calculations. Be sure to have your calculator and charts ready.
Chemistry Final Exam Review Part 1
By llanham summitt
Show answer
Auto Play
Slide 1 / 159
SLIDE
Similar Resources on Wayground

144 questions
MODUL GENERATIVE AI
Presentation
•
10th Grade

161 questions
SP - S8 2026
Presentation
•
KG

151 questions
DiHybrid Crosses
Presentation
•
9th Grade

157 questions
Intro to Government
Presentation
•
11th - 12th Grade

153 questions
4E Nutrition & Transport in Plants
Presentation
•
10th Grade

155 questions
Medical Term - Digestive System
Presentation
•
10th Grade

154 questions
Q4_W3_M3_Pagbása at Pagsusuri
Presentation
•
11th Grade

149 questions
Math 8 Module 15
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade