

Teach .Ed. Periodic table/Mixtures
Presentation
•
Science
•
8th Grade
•
Medium
Standards-aligned
Aguado Angelica
Used 7+ times
FREE Resource
21 Slides • 34 Questions
1

2

3
Periods
The horizontal rows on the periodic table
All elements in the same period have the same number of energy levels in their electron cloud
4
Groups
The vertical columns on the periodic table
All elements in the same group have the same number of valence electrons
5
Multiple Choice

The positively charged particle in an atom.
Proton
Neutron
Electron
Nucleus
6
Multiple Choice
The unit used to express the mass of the particles in an atom.
Proton
AMU
Neutron
Electron
7
Multiple Choice
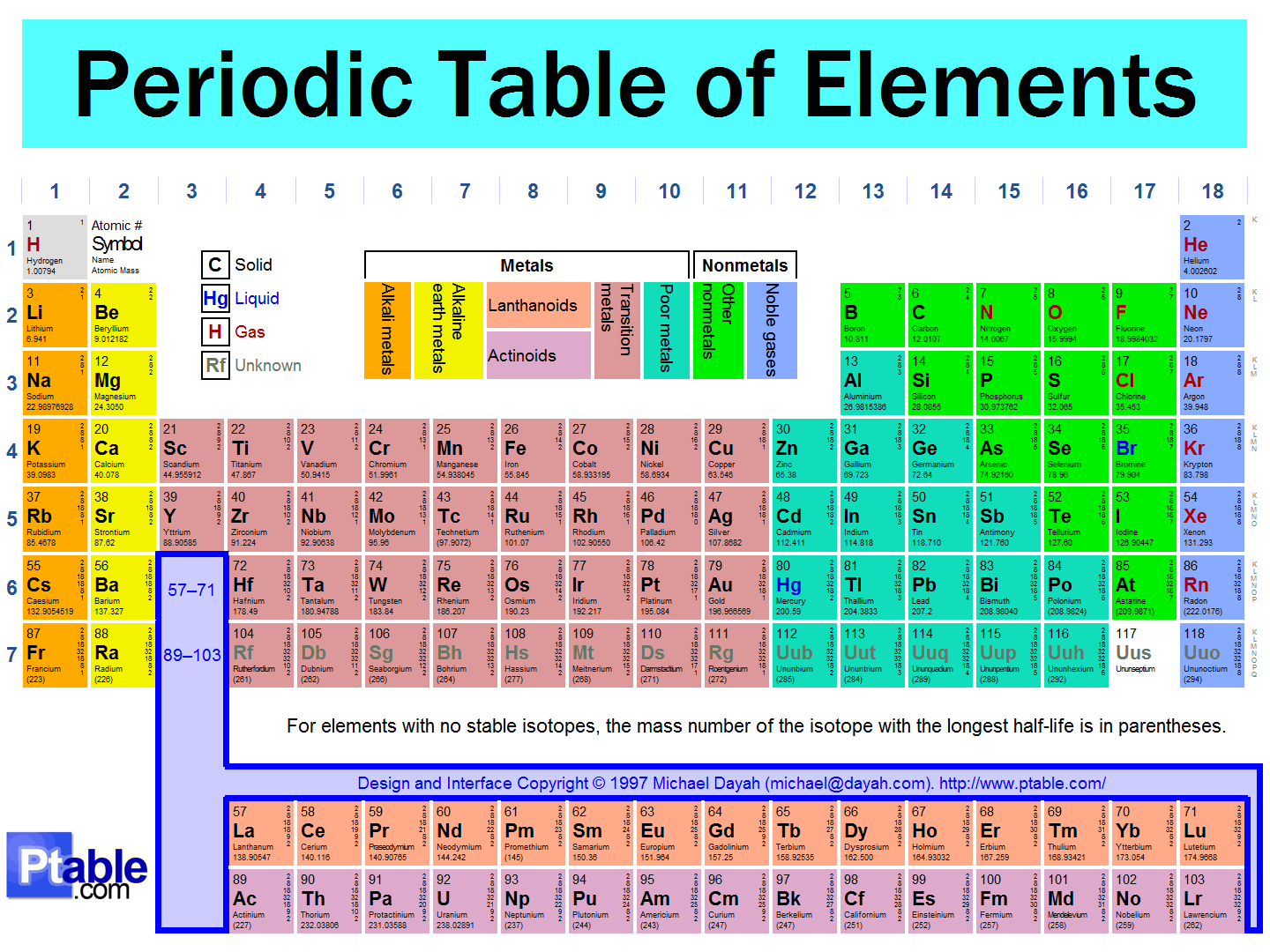
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
8
Multiple Choice
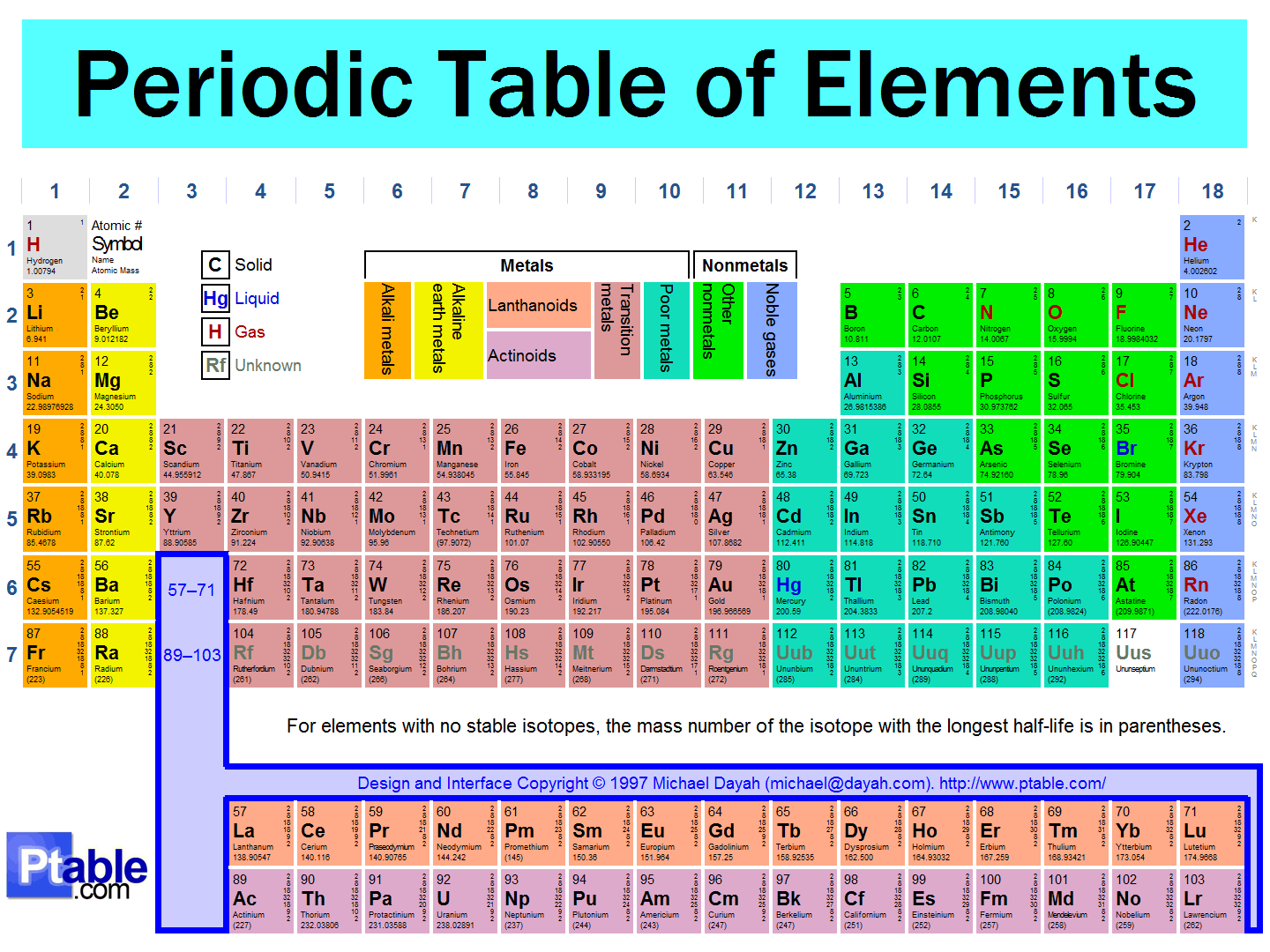
9

10

11

12

13

14

15
Multiple Choice

16
Multiple Choice
Elements in the periodic table are arranged by...
Atomic mass
Atomic number
Number of neutrons
Chemical reactivity
17
Multiple Choice
The rows of the periodic table are called periods.
True
False
18
Multiple Choice
Which of these is NOT a family of elements?
Halogens
Alkali Metals
Transition Metals
All of these are families
19
Multiple Choice
You will find metals on the far right column of the periodic table.
True
False
20
Multiple Choice

Atoms of elements in the same group have the same number of what?
protons
protons and neutrons
valence electrons
neutrons
21
Multiple Choice

Which two elements share common characteristics based on the periodic table?
Sodium and Magnesium
Chlorine and Sulfur
Argon and Neon
Calcium and Bromine
22
Multiple Choice
What is a group (also called a family)?
Number of valence electrons
Orbital levels/energy levels
elements with similar properties
Number of elements
23
Multiple Select
The subatomic particle(s) found in the nucleus of an atom
Proton and electron
Neutron and electron
Protons and neutron
Neutron and proton
24
Multiple Choice

25
Multiple Choice

26
Multiple Choice

27
Multiple Choice
one or two letters that represent the name of an element
element name
element symbol
atomic number
atomic mass
28
Multiple Choice
Name this group: These metals are the most reactive.
29
Multiple Choice
Name this group: These metals are the second most reactive.
30
Multiple Choice
Name this group: These metals contain familiar metals such as gold, iron, and copper.
31
Multiple Select
Locate Lanthanides and Actinides.
32
Multiple Choice
33
Explanation Slide...
Noble gases are found on the far right of the periodic table in group VIIIA. Halogens are in the second group from the right. Metals of all types are found around the left side of the periodic table. There may be earth, transition, or alkaline earth metals across the table.

34
Explanation Slide...
All of these choices are different types and families of elements. Inert gases can be found on the far right of the periodic table in group VIIIA. Halogens are just to the left in VIIA. Metals make up the center and left side of the table in a variety of groups.

35
Explanation Slide...
The rows of the periodic table are called periods. The columns of the periodic table are called groups. Periods are loosely organized by the number of atomic orbitals found in each element. For example, hydrogen (H) in the first row has only one orbital. Lithium (Li) in the second row has electrons in two orbitals. Francium (Fr), in row seven, has electrons in seven orbitals.

36
Multiple Choice
6.15: Which of these is not a property of noble gases?
Colourless
Odourless
Flammable
Gas at room temperature
37
Multiple Choice

38
Compound
Elements can chemically combine to form new, pure chemical substances.
A chemical substance that is made of two or more different elements that chemically combine together is called a compound
The force between compounds is STRONGER than between mixtures


39
Contain one or more substances NOT bonded together
Can be separated physically
Retains many of the properties of the components
Record the information above on your note-taker.
Mixtures

40
Mixture
A compound is NOT the same as a mixture
A mixture is a physical combination of two or more different chemical substances.
A compound is a chemical combination of two or more different chemical substances (specifically, elements)
Unlike an element and compound, a mixture is NOT a pure chemical substance


41

42

43
Multiple Choice
A combination of two or more substances that can be separated is a
potion
mixture
solute
compound
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice
In sweet tea the solvent would be
water
tea
sugar
lemon
47
Multiple Choice
48
Concept Map of Classifying Matter

49
Recall
Mixtures are a combination of substances
Can heterogenous or homogenous
Heterogenous Mixtures: large particles that can be easily seen and separated
Suspensions: a heterogenous mixture that separates on its own over time
Clay in water
Colloids: a heterogenous mixture that does not separate on its own over time
Milk


50
Homogenous solutions are Solutions
Solution: any mixture that is homogenous on a microscopic level
Usually a liquid, but can be solid or gas as well
Solid solutions tend to be alloys like brass
Liquids Solutions have two parts
Solvent: The main ingredient that you dissolve substances in
Water is called the universal solvent because it dissolves so many things into aqueous solutions
Solute: the ingredient you are dissolving

51
Multiple Choice
The ______ contains a dispersed phase and a dispersing medium.
colloids
solute
solvent
solution
52
Multiple Choice
Which of the following is not an example of a colloid?
whipped cream
mayonnaise
jelly
sand
53
Multiple Choice
Which of the following is not an example of suspension?
Flour suspended in water
Chalk suspended in water
Sand suspended in water.
Rocks in water
54
Multiple Choice
Which of the following is an example of a solution?
salt in water
sand and water
rocks and water
pebbles and water
55
Multiple Choice

Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

51 questions
Chemistry Review
Presentation
•
8th Grade

54 questions
MMS L1
Presentation
•
8th Grade

51 questions
Hurricanes and El Nino
Presentation
•
8th Grade

48 questions
Introduction to Electromagnetic Waves
Presentation
•
8th Grade

51 questions
Microorganism- friend and foe
Presentation
•
8th Grade

50 questions
Lotus Intro
Presentation
•
8th Grade

51 questions
Speed, Velocity and Acceleration IA Review #3
Presentation
•
8th Grade

51 questions
Energy, Motion, & Newton's Laws
Presentation
•
8th Grade
Popular Resources on Wayground

25 questions
The Ultimate College Knowledge Quiz
Quiz
•
8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Math Review
Quiz
•
6th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

52 questions
8th Grade NC Science EOG Review 1
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

14 questions
6.8 - The Space Race
Quiz
•
6th - 8th Grade

53 questions
7.6 Earth Resources/Human Impact Review
Quiz
•
6th - 8th Grade

33 questions
Human Body Systems
Quiz
•
8th Grade