

Phase changes and States of Matter
Presentation
•
Science
•
8th Grade
•
Medium
+9
Standards-aligned

Laura Terry
Used 30+ times
FREE Resource
0 Slides • 56 Questions
1
Multiple Choice

What state of matter does the picture below represent?
solid
liquid
gas
plasma
2
Multiple Choice
The state of matter that keeps its shape and volume when it is placed in a different container is called ______.
gas
liquid
solid
3
Multiple Choice
The state of matter that expands to fill its container is called ______.
gas
liquid
solid
4
Multiple Choice
What is a Solid?
Matter that has a fixed shape and size.
Matter that has a fixed size and no fixed shape
Matter that has no fixed shape or size
Matter that has no fixed shape and size
5
Multiple Choice
Which state of matter has its own shape
Gas
Solid
LIquid
6
Multiple Choice
Define a liquid.
Matter that has fixed shape but no fixed volume
Matter that has fixed shape and fixed volume
Matter that has fixed volume, but no fixed shape
7
Multiple Choice

The particles in the picture are tightly held together with very tiny movement. They have shape and fixed volume. Therefore, the picture represents _________.
solid
liquid
gas
plasma
8
Multiple Choice
In which state of matter will there be the LARGEST space between molecules?
Solid
Liquid
Gas
9
Multiple Choice
What is a gas?
Matter that has fixed shape but no fixed volume
Matter that has no fixed shape and no fixed volume
Matter that has no fixed shape but fixed volume
compounds
10
Multiple Choice

What state is represented in the gif?
Solid
Liquid
Gas
11
Multiple Choice
What determines the states of matter a material is in?
solid
liquid
temperature
gas
12
Multiple Choice
Liquid has a fixed shape
True
False
13
Multiple Choice
Solid has a fixed shape
True
False
14
Multiple Choice

Evaporation is the change of state of a
Gas to solid
Liquid to Gas
Solid to liquid
Liquid to Solid
15
Multiple Choice

What is the state change that occurs, when ice turns into a liquid?
Boiling
Condensation
Melting
evaporation
16
Multiple Choice

Condensation is when
Liquid turns to a solid
Gas turns to liquid
Solid turns to liquid
Liquid turns to a gas
17
Multiple Choice
Solid particles are
Far apart
Close, but able to move
Closest together
Fast moving
18
Multiple Choice

This image is an example of what particle
Solid
Liquid
Gas
19
Multiple Choice
What particles move the fastest?
Solid
Liquid
Gas
20
Multiple Choice
What is the state of matter of ice
Gas
Liquid
Solid
21
Multiple Choice
What is the name of the change when a liquid becomes a solid?
Precipitation
Freezing
boiling
evaporation
22
Multiple Choice
Why do solids have a fixed shaped?
The particles vibrate
The particles are fixed in place
The particles are closely packed together
The particles are free moving
23
Multiple Choice
When a gas reaches its condensation point what does it become?
Solid
Gas
Liquid
24
Multiple Choice
When a liquid reaches its freezing point what does it become?
Liquid
Gas
Solid
Plasma
25
Multiple Choice
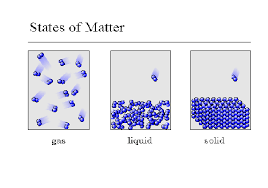
26
Multiple Choice
The slower the particles in a substance move,
the colder it is.
the warmer it is.
the more energy it has.
27
Multiple Choice
28
Multiple Choice
29
Multiple Choice

30
Multiple Choice

31
Multiple Choice
What is matter?
Only the things we can see with our eyes.
Anything that has mass and takes up space.
Only liquids and solids.
The things that are important to us like happiness.
32
Multiple Choice

What effect is heat having on the ice?
Thermal expansion.
Temperature increase.
Temperature decrease.
Change in state.
33
Multiple Choice
What is the freezing in celsius
32°
100°
0°
965°
34
Multiple Choice
What is boiling in celsius
212°
32°
100°
1,000,000°
35
Multiple Choice
When it is cold the atoms move...
slow
fast
still
there are no atoms
36
Multiple Choice
when it is hot the atoms move...
slow
fast
still
there are no atoms
37
Multiple Choice
in the kelvin scales what degree stands for when the atoms are not moving
50°
32°
0°
100°
38
Multiple Choice
100 Celsius is ? Kelvin (K = Celsius + 273)
173 K
373K
273 K
0 K
39
Multiple Choice
Absolute Zero is which value:
0 K
0 °F
0 °C
40
Multiple Choice
41
Multiple Choice
the temperature scale on which 0 is the temp at which no more energy can be removed
absolute zero
Fahrenheit scale
Kelvin scale
Celsius scale
42
Multiple Choice
the temperature scale on which water freezes at 0 and boils at 100
Kelvin scale
conduction
Fahrenheit scale
Celsius scale
43
Multiple Choice
°C = (°F-32) x (5/9)
°F = (9/5) x °C + 32
Celsius = Kelvin - 273
Kelvin = Celsius + 273
Convert the temperature:
25 C = _______ F
77 °F
32 °F
28 °F
44
Multiple Choice
°C = (°F-32) x (5/9)
°F = (9/5) x °C + 32
Celsius = Kelvin - 273
Kelvin = Celsius + 273
Convert the temperature:
97 °C = _______ °F
97.3 °F
10.2 °F
206.6 °F
45
Multiple Choice
°C = (°F-32) x (5/9)
°F = (9/5) x °C + 32
Celsius = Kelvin - 273
Kelvin = Celsius + 273
Convert the temperature:
2 °C = _______ °F
56.9 °F
35.6 °F
0 °F
46
Multiple Choice
°C = (°F-32) x (5/9)
°F = (9/5) x °C + 32
Celsius = Kelvin - 273
Kelvin = Celsius + 273
Convert the temperature:
-40 °C = _______ °F
-40 °F
32 °F
0 °F
47
Multiple Choice
°C = (°F-32) x (5/9)
°F = (9/5) x °C + 32
Celsius = Kelvin - 273
Kelvin = Celsius + 273
Convert the temperature:
25 °F = _______ °C
-3.9 °C
-10 °C
13 °C
48
Multiple Choice
What is the freezing temperature of water in Kelvin?
0
32
273.1
49
Multiple Choice
What is the boiling point of water in Kelvin?
212
100
373.1
50
Multiple Choice
51
Multiple Choice
A thermometer takes the temperature as the liquid inside will _______ or _______ as the temperature changes.
expand or contract
move faster or move slower
become more or become less
52
Multiple Choice
Thermal expansion happens when _____________.
temperatures increase
temperatures decrease
temperatures fluctuate
53
Multiple Choice
Heat loss causes matter to ________. Heat gain causes matter to ________.
contract ; expand
expand ; contract
stay ; change
change ; stay
54
Multiple Choice
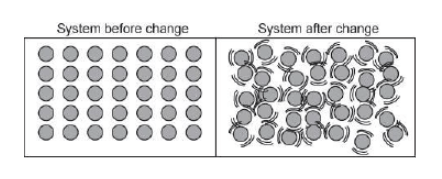
The picture below shows a closed system before and after undergoing a change.
What was changed in the system?
Particles were added to the system.
Water was added to the system.
Heat was added to the system.
Air was added to the system.
55
Multiple Choice

56
Multiple Choice


What state of matter does the picture below represent?
solid
liquid
gas
plasma
Show answer
Auto Play
Slide 1 / 56
MULTIPLE CHOICE
Similar Resources on Wayground

53 questions
EID 1.3-1.4 Competition / Biodiversity and Human Impact
Presentation
•
8th Grade

49 questions
Wave Quiz #1 Lesson Review
Presentation
•
8th Grade

50 questions
ElectromagneticSpectrum 2024
Presentation
•
8th Grade

47 questions
SVA- Speed, Velocity, Acceleration
Presentation
•
8th Grade

50 questions
Universal Solvent - Factors Affecting Solubility - Concentration
Presentation
•
8th Grade

52 questions
Science 8 Year End Review- Unit B
Presentation
•
8th Grade

53 questions
EM Spectrum EOU Assignment
Presentation
•
8th Grade

51 questions
Topographic maps
Presentation
•
8th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

19 questions
Renewable and Nonrenewable Resources (CK)
Quiz
•
8th Grade

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade