

G10_4.4: Molecular Shapes
Presentation
•
Science
•
10th Grade
•
Medium
+2
Standards-aligned
Oyerohunke oyediran
Used 5+ times
FREE Resource
28 Slides • 27 Questions
1

Molecular Shapes
Lesson 4
2


Focus Question
What shapes do molecules form?
3


New Vocabulary
VSEPR model
hybridization
4


Review Vocabulary
atomic orbital: the region around an atom’s nucleus
that defines an electron’s probable location
5
Multiple Choice

Region of high probability of finding an electron
atomic orbital
ground state
Heisenberg uncertainty principle
electron configuration
6

7

8
Multiple Choice
s orbitals are _____shaped
spherical
dumbbell
circular
9
Multiple Choice
p - orbitals are ______shaped
spherical
dumb-belled
circular
10

11
Multiple Choice
Valence Structure of Electron Pyramids and Regression
Varied Structures of Electrons Paired and Replaced
Varied Shell Energy of Protons and Radiation
Valence Shell Electron Pair Repulsion
12



VSEPR Model
• The molecular geometry
(shape) of a molecule can be
determined using the Valence
Shell Electron Pair Repulsion
model, or VSEPR model.
• It is based on an arrangement
that minimizes the repulsion of
shared and unshared electron
pairs around the central atom.
13




VSEPR Model
Bond Angle
• Electron pairs repel each other and cause
molecules to be in fixed positions relative to
each other.
• Unshared electron pairs called LONE PAIRS also determine the shape of a molecule.
• Electron pairs are located in a molecule as far
apart as they can be.
14
Multiple Choice
A pair of bonding electrons
One non-bonding electron
A pair of non-bonding electrons
A pair of electrons on the central atom
15
Multiple Choice

The ------ model helps predict the shape of a molecule.
VESPR
Electron
Lewis
16
Multiple Choice
The model is based on the principle that electron pairs around the central atom ------ each other.
bond
attract
repel
17
Multiple Choice
The ------ of the molecule determines the properties of the compounds.
density
shape
mass
18



Hybridization
• Hybridization occurs when two things are combined
and the result has characteristics of both.
• If electrons come from two different atomic orbitals,
such as a p or an s, they must rearrange or combine
into a hybrid orbital with the same shape and energy
level.
19
Multiple Choice

atomic orbital overlap to produce sigma bond
s orbital overlap to produce π bond
s orbital overlap with s orbital to produce sigma bond
p orbital overlap to produce π bond
20
Multiple Choice

formation of hybrid orbital
atomic orbital overlap to produce hybrid orbital
atomic orbital overlap to form sp hybrid orbital
s orbital overlap with p orbital to form sp hybrid orbital
21
Multiple Choice

s orbital overlap with p orbitals to form sp hybrid orbital
s orbital with two p orbitals to form one sp2 hybrid orbital
s orbital with two p orbitals to form two sp2 hybrid orbital
s orbital with two p orbitals to form three sp2 hybrid orbital
22
Multiple Choice
Bond Strength
Polarity
Molecular Shape
Electronegativity
23
Multiple Choice
The covalent bond forms between
a metal and nonmetal
a nonmetal and nonmetal
a metal and metal
24



Hybridization
• Consider the bonding involved in the methane
molecule (CH4) on the image below.
• The hybrid orbitals in a carbon atom are
shown in blue.
25



Hybridization
• Carbon initially has only 2
electrons in its p orbital.
• A 1 s electron is promoted
from the s to the p orbital
so a total of 4 unpaired
electrons can be shared.
• These electrons undergo
hybridization, which forms
four hybrid sp3 orbitals that
can bond with 4 hydrogen
atoms.
26
Multiple Choice

CO2
NH3
SO3
NO2
27
Multiple Choice
Molecule CO2
undergoes sp2 hybridisation
has 2 σ bond and 2 π bond
undergoes sp3d hybridisation
is trigonal planar molecule
28



Hybridization
• The number of atomic orbitals that mix and form the
hybrid orbital equals the total number of electron pairs.
• The number of hybrid orbitals formed equals the
number of atomic orbitals mixed (sigma bonds).
29



Hybridization
• For example, the table on the next slide shows that AlCl3
has a total of three pairs of electrons and VSEPR predicts
a trigonal planar shape.
• The shape results when one s and two p orbitals on the
central atom, Al, mix and form three identical sp2 hybrid
orbitals.
30
Multiple Choice

What is the the shape of this molecule according to VSEPR theory?
Linear
Tetrahedral
Trigonal Planar
Trigonal pyramidal
31
Multiple Choice
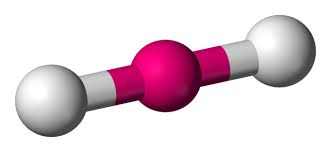
What is the hybridization of a linear molecule?
sp
sp2
sp3
sp3d
32
Multiple Choice
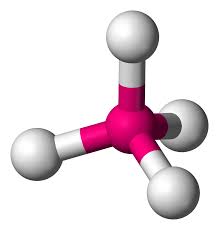
What is the hybridization of this molecule shown above
sp
sp2
sp3
sp4
33
Multiple Choice
Which of the following images represents a Trigonal Planar molecular geometry?
34




Hybridization
• BeCl2 has two pairs of electrons shared with the
central Be atom. The bonding electrons have
maximum separation and a bond angle of 180. The
shape is linear.
Molecule
Total
Pairs
Shared
Pairs
Lone
Pairs
Hybrid
Orbitals
Molecular Shape
BeCl2
2
2
0
sp
AlCl3
3
3
0
sp2
35
Multiple Choice

two sp hybrid orbitals
sp hybrid orbitals
one sp hybrid orbitals
sp2 hybrid orbitals
36
Multiple Choice

BeCI
BeCI2
BeCI3
BeCI4
37



Hybridization
• Lone pairs also occupy hybrid orbitals.
• Single, double, and triple bonds occupy only one hybrid orbital.
Thus, CO2, with two double bonds, forms sp hybrid orbitals.
38



Hybridization
• The H2O molecule has sp3 orbitals. There are two
lone pairs on the central oxygen atom in H2O.
Therefore, there must be four hybrid orbitals—two
for bonding and two for lone pairs. The bent shape
is shown on the next slide.
39
Multiple Choice
In a water (H2O) molecule, the polar bonds between the oxygen atom and the hydrogen atoms do not cancel out because the molecule is bent.
True
False
40





Hybridization
Molecule
Total
Pairs
Shared
Pairs
Lone
Pairs
Hybrid
Orbitals
Molecular Shape
CH4
4
4
0
sp3
NH3
4
3
1
sp3
H2O
4
2
2
sp3
41




Hybridization
Molecule
Total
Pairs
Shared
Pairs
Lone
Pairs
Hybrid
Orbitals
Molecular Shape
NbBr5
5
5
0
sp3d
SF6
6
6
0
sp3d2
42
Multiple Choice
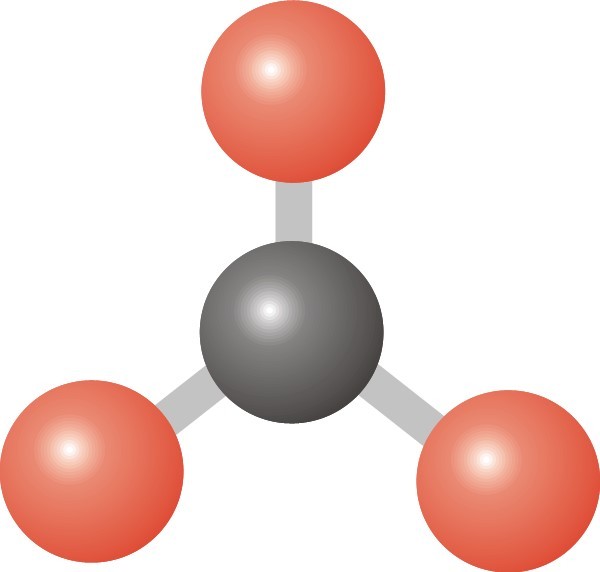
BF3
CH4
H2O
CO2
43
Multiple Choice
Bipyramidal
Bent
Trigonal Planar
Tetrahedral
44
Multiple Choice

structure A
structure B
structure C
structure D
45
Multiple Choice

determine the number of bonding and lone pairs electrons
determine the number of ECC
determine the shape and geometry of molecule
determine the lewis structure of molecule
46
Multiple Choice
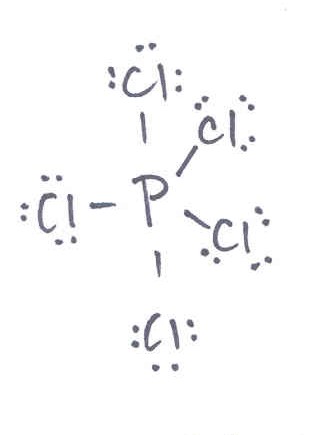
3 sigma and 2 pi
5 sigma and 5 pi
5 sigma and 0 pi
0 sigma and 5 pi
47


Think of the Quiz answers before moving
to the next slide
48

Quiz
It is based on an arrangement that maximizes the
attraction of shared electrons and the repulsion of
shared electron pairs.
D
It is based on an arrangement that maximizes the
repulsion of shared and unshared electron pairs.
C
It is based on an arrangement that minimizes the
repulsion of shared and unshared electron pairs.
B
It stands for Valence Shell Electron Pair Repulsion.
A
Which of the following is not true of the VSEPR
model?
1.
49

Quiz
It is based on an arrangement that maximizes the
attraction of shared electrons and the repulsion of
shared electron pairs.
D
It is based on an arrangement that maximizes the
repulsion of shared and unshared electron pairs.
C
It is based on an arrangement that minimizes the
repulsion of shared and unshared electron pairs.
B
It stands for Valence Shell Electron Pair Repulsion.
A
Which of the following is not true of the VSEPR
model?
1.
CORRECT
50

Quiz
trigonal planar
D
trigonal pyramidal
C
bent
B
tetrahedral
A
A molecule has three single covalent bonds,
one lone pair, and sp3 hybrid orbitals. What is
its likely shape?
2.
51

Quiz
trigonal planar
D
trigonal pyramidal
C
bent
B
tetrahedral
A
A molecule has three single covalent bonds,
one lone pair, and sp3 hybrid orbitals. What is
its likely shape?
2.
CORRECT
52

Quiz
180°
D
120°
C
109.5°
B
A
What are the bond angles in a trigonal planar
molecule?
3.
90°
53

Quiz
180°
D
120°
C
109.5°
B
A
What are the bond angles in a trigonal planar
molecule?
3.
CORRECT
90°
54

Quiz
octahedral
D
trigonal bipyramidal
C
trigonal pyramidal
B
tetrahedral
A
A molecule has 6 shared pairs of electrons and no
lone pairs of electrons. Its hybrid orbitals are sp3d2.
What is its likely shape?
4.
55

Quiz
octahedral
D
trigonal bipyramidal
C
trigonal pyramidal
B
tetrahedral
A
A molecule has 6 shared pairs of electrons and no
lone pairs of electrons. Its hybrid orbitals are sp3d2.
What is its likely shape?
4.
CORRECT

Molecular Shapes
Lesson 4
Show answer
Auto Play
Slide 1 / 55
SLIDE
Similar Resources on Wayground

50 questions
G10 Lesson 4.3: Molecular Structures
Presentation
•
10th Grade

51 questions
Virus Structure
Presentation
•
10th Grade

46 questions
Exploring Angle Pairs
Presentation
•
10th Grade

51 questions
Chemical Reactions
Presentation
•
10th Grade

51 questions
Chamisal Bonding Regents Review
Presentation
•
10th Grade

52 questions
Monday SEV2b
Presentation
•
9th Grade

51 questions
willowridge. Photosynthesis. Energy Conversion.
Presentation
•
9th - 10th Grade

52 questions
Protein & Enzymes DT 22-23 ADV
Presentation
•
9th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade