

1.2 Atomic Structure
Presentation
•
Chemistry
•
6th - 8th Grade
•
Medium
Lloyd Cascabel
Used 2+ times
FREE Resource
19 Slides • 78 Questions
1

2

Dimitri Mendeleev organized all the atoms of different elements into a table based on their atomic number.
3

4

5
Due to developments in available technology and with more information is generated from the creativity of scientists, the model of the atom grew in time.

6
Multiple Choice
Bohr Model of an atom
7
Multiple Choice
Quantum Mechanical Model of an Atom
8
Multiple Choice
Dalton's Atomic Model
9
Multiple Choice
Who was the first person to "think" of an atom?
Democritus
J.J. Thompson
Rutherford
Bohr
10
Multiple Choice
empty space, positively
empty space, negatively
full of protons, positively
full of neutrons, negatively
11
Multiple Choice
Who organized the elements into a table based on their atomic number?
John Dalton
Aristotle
Dimitri Mendeleev
Earnest Rutherford
12
Multiple Choice
Which of the following is similar to the models of Rutherford, Bohr, Shrodinger, and Thompson?
Atoms are indivisible
Atoms are all the same
Atoms have positive and negative charges
Atoms have layers of neutrons
13
Multiple Choice
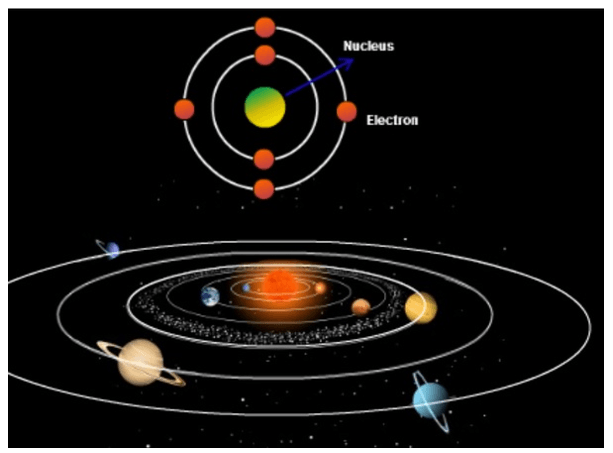
Who proposed this atomic model?
Earnest Rutherford
John Dalton
Democritus
Niels Bohr
14

There are three subatomic particles: protons (+), electrons (-), and neutrons (no charge).
The number of protons determine the atomic number and the identity of the element.
15

There are three subatomic particles: protons (+), electrons (-), and neutrons (no charge).
The number of protons determine the atomic number and the identity of the element.
Boron
.
.
.
Silver
16

17
Multiple Choice
An atom of an element is electrically neutral because the number of protons
is equal to the number of _____________.
A. electrons
B. neutrons
C. nucleons
D. nucleus
18
Multiple Choice
Which two particles would be attracted to each other?
A. protons and neutrons
B. electrons and protons
C. electrons and neutrons
D. All particles are attracted to each other.
19
Multiple Choice
What was Thomson's atomic model called?
A. Spherical Model
B. Solar System Model
C. Plum Pudding Model
D. Model of the Atomic Theory
20
Multiple Choice
Which of the following statements BEST describes the location of electrons in
Rutherford’s model of the atom?
A. The electrons are inside the nucleus.
B. The electrons are outside the nucleus.
C. There are no electrons in the Rutherford Model.
D. Electrons are both inside and outside of the nucleus.
21
Multiple Choice

The nucleus of an atom contains which subatomic particles?
protons and electrons
protons and neutrons
electrons and neutrons
protons, electrons, and neutrons
22
Multiple Choice

In a neutral atom, which two subatomic particles are equal in number?
protons and neutrons
all subatomic particles are equal in number
neutrons and electrons
protons and electrons
23
Multiple Choice

Which subatomic particle is counted to determine the type of element the atom represents?
proton
neutron
electron
boron
24
Multiple Choice
negatrons
electrons
neutrons
protons
25

26

27

28

Neon Ne-20 isotope
Neon Ne-21 isotope
29

30

31
Multiple Choice
Which particles have approximately the same size and mass?
A. protons and neutrons
B. electrons and protons
C. neutrons and electrons
D. None because all are different in terms of size and mass
32
Multiple Choice
the number of protons only
the number of neutrons only
the total number of protons and neutrons
the number of protons, neutrons, and electrons.
33
Multiple Choice
negatrons
electrons
neutrons
protons
34
Multiple Choice

How many neutron are in the atom "K"?
29
19
58
20
35
Fill in the Blanks
36
Fill in the Blanks
37
Multiple Choice
Potassium has a mass number of 39. How many protons does it have?
39
19
20
38
Multiple Choice
The smallest subatomic particle
Protons
Neutrons
Electrons
Atoms
39
Multiple Choice
An element has an atomic number of 9 and a mass of 19 amu. What is the number of protons this element has?
9
19
10
28
40
Multiple Choice

What is represented by the letter T?
Nucleus
Electron
Proton
Neutrons
41
Multiple Choice

What is represented by the letter S?
Nucleus
Electron
Proton
Neutrons
42
Multiple Choice

What is represented by the letter R?
Nucleus
Electron
Proton
Neutrons
43
Multiple Choice
What gives you the identity of an element?
Mass
Atomic number
Protons
Electrons
44
Multiple Choice
An element has 3 electrons and a mass of 7. How many protons does this element have?
3
7
4
10
45
Multiple Choice
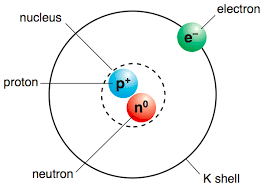
Protons, Neutrons, Electrons
Protons only
Protons and Electrons
Protons and Neutrons
46
Multiple Choice
How many electrons does an atom of Krypton have?
36
83.80
48
119.80
47
Multiple Choice
How many electrons does a Copper atom have?
63
29
92
34
48
Multiple Choice
Typically in an atom, which two subatomic particles are equal in number?
protons and neutrons
all subatomic particles are equal in number
neutrons and electrons
protons and electrons
49
Multiple Choice
How many protons are present in phosphorus?
15
16
31
46
50
Multiple Choice
Lithium has 7 a.m.u. How many neutrons does this lithium have?
8
3
4
5
51

52

53

54

55

56
Multiple Choice
Element A has a mass number of 100 while element B has an atomic number
of 118. What will be the number of electrons of element B?
A. 59
B. 60
C. 118
D. 120
57
Multiple Choice

Positive
Negative
Neutral
Invinsible
58
Multiple Choice
a winner
an isotope
an ion
a loser
59
Multiple Choice
When an atom loses an electron, it becomes a
positive ion
negative ion
neutral ion
neutral atom
60
Fill in the Blanks
61
Multiple Choice
If the number of protons and the number of electrons are equal, the net charge will be...
Positive
Neutral
Negative
62
Fill in the Blanks
63
Multiple Choice
An atom of oxygen has a - 2 charge, how many electrons does it have?
8
10
6
64
Multiple Choice
An atom of fluorine has a -1 charge, how many protons does it have?
10
8
9
65
Multiple Choice
Potassium has a mass number of 39. How many protons does it have?
39
19
20
66
Multiple Choice
An element has 3 electrons and a mass of 7. How many protons does this element have?
3
7
4
10
67
Multiple Choice
What is a positive ion called?
positron
pos-ion
anion
cation
68
Multiple Choice
What is a negative ion called?
negion
negation
anion
cation
69
Multiple Choice
Fluorine (F) has an atomic number of 9 and an atomic mass of 18.998. How many electrons does it have?
18
10
9
70
Match
Li
He
Na
Li
He
Na
71
Fill in the Blanks
72
Multiple Choice

How many protons are in Silicon?
7
42
28
14
73
Multiple Choice

How many neutrons are in Phosphorus?
16
15
31
46
74
Multiple Choice

How many electrons are in Aluminium?
40
27
13
14
75
Multiple Choice
Adding or subtracting electrons changes the elements overall charge and makes the element change into an...
ion
mixture
isotope
neutral atom
76
Multiple Choice
A neutral atom of the element sodium has ____ electrons.
22
23
8
11
77
Multiple Choice

63
29
92
34
78
Multiple Choice

1
2
3
0
79
Multiple Choice
15
16
31
46
80
Multiple Choice

Symbol
Name
Mass Number
Atomic Number
81
Multiple Choice
A rule that states that atoms are most stable when they have a full shell
of electrons in the outside electron shell.
Octet Rule
Golden Rule
Hund's Rule
All of the above
82
Multiple Choice
According to the Octet Rule, atoms of elements react with each other
in order to attain electrons in their outermost energy level or shell.
2
4
6
8
83
Multiple Choice
The valence electrons of representative elements are
In s orbitals only.
Located in the outermost occupied major energy level.
Located closest to the nucleus.
Located in d orbitals
84
Multiple Choice
How do you find the number of valence electrons in an atom?
Use the Atomic Number
Use the Group Number
Use the Mass - Atomic number
Use the Period Number
85
Multiple Choice
Which of the following elements GAINS 3 electrons in order to attain an octet?
nitrogen
lithium
boron
oxygen
fluorine
86
Multiple Choice
Which of the following elements can EITHER GAIN or LOSE 4 electrons in order to attain an octet?
silicon
beryllium
boron
selenium
chlorine
87
Multiple Choice
Each of the following elements will LOSE ELECTRONS in order to attain an octet EXCEPT _______
iodine
magnesium
potassium
copper
strontium
88
Multiple Choice
When atoms GAIN electrons, they get a...
Positive (+) charge
Negative (-) charge
No charge (zero)
89
Multiple Choice
Fluorine gains 1 electron to satisfy the Octet Rule. What kind of ion is formed?
Anion (-)
Cation (+)
90
Multiple Choice

What does Oxygen have to do to satisfy the Octet Rule? What would its charge be after? (TAKE YOUR TIME!)
Lose 6 electrons, + 6
Gain 8 electrons, - 8
Lose 2 electrons, + 2
Gain 2 electrons, - 2
91
Multiple Choice

Which group already satisfies the Octet Rule and therefore does not bond with other atoms?
Group 1 (H at top)
Group 2 (Be at top)
Group 13 (B at top)
Group 18 (He at top)
92
Multiple Choice
When anions and cations join together, what kind of bond is formed?
Ionic
Covalent
Metallic
Hydrogen
93
Multiple Choice

What is the charge of the Na ion based on its position on the Periodic Table?
Neutral (zero)
+1
-1
+11
94
Multiple Select
Beryllium (Be) is found in Group 2. What can Beryllium do in order to follow the Octet Rule? (Check ALL that work...)
Gain 6 more valence electrons
Gain 8 more valence electrons
Lose 2 valence electrons
Lose 8 valence electrons
95
Multiple Choice
Phosphorus (P) is in Group 5. How many MORE electrons does it need in order to follow the Octet Rule?
2 more valence electrons
5 more valence electrons
8 more valence electrons
3 more valence electrons
96
Multiple Choice

Do you think Nitrogen at the central already achieved an octet?
Yes
No
97
Multiple Choice

Do you think Sulfur at the central already achieved an octet?
Yes
No

Show answer
Auto Play
Slide 1 / 97
SLIDE
Similar Resources on Wayground

91 questions
Math 7 SOL Review
Presentation
•
7th Grade

91 questions
Unit 0 - 2 Vocab Review
Presentation
•
6th - 8th Grade

90 questions
Heat Energy Transfer
Presentation
•
6th - 8th Grade

95 questions
Review Skills-BOY-7 & 8th Grade
Presentation
•
6th - 8th Grade

89 questions
Math 7 SOL Review
Presentation
•
7th Grade

88 questions
“Teens Say They are Turning To AI for Friendship (Part 2)
Presentation
•
6th - 8th Grade

90 questions
East and Southeast Asia
Presentation
•
5th - 6th Grade

96 questions
Career Cluster Overload
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade

20 questions
Acids and Bases
Quiz
•
8th Grade

20 questions
Chemical Reactions
Quiz
•
8th Grade

12 questions
Rate of reaction
Quiz
•
8th - 9th Grade

20 questions
Counting Atoms
Quiz
•
8th Grade

10 questions
Exploring Solutions and Their Properties
Interactive video
•
6th - 10th Grade

5 questions
Balancing Chemical Equations
Interactive video
•
8th Grade