

The Brainery Intro Chemistry Class
Presentation
•
Science
•
9th - 12th Grade
•
Hard
Standards-aligned
Sharon Tan
FREE Resource
26 Slides • 59 Questions
1

Secondary Three Chemistry

2
Open Ended
What is Chemistry?
3

Think that it is all about reactions and mixing of chemicals?
What is Chemistry?
4
5

You can open this webpage in a new tab.
https://www.seab.gov.sg/docs/default-source/national-examinations/syllabus/olevel/2024syllabus/5078_y24_sy.pdf
6
Building on Lower Secondary Science
C8 Atomic Structure: Protons, Electrons, Neutrons
Mass number, Atomic/Proton Number
C11 Chemical Changes: Acids, Bases/Alkalis
C3 Chemical Composition: Elements, Mixtures, Compounds, Periodic Table
C4 Separation Techniques: Filtration, Evaporation, Chromatography, Distillation
C7 Particulate Nature of Matter: Particles of Solid, Liquid, Gas

7

8

9

10

11

number is a double digit, we subtract 10.
12
Multiple Choice
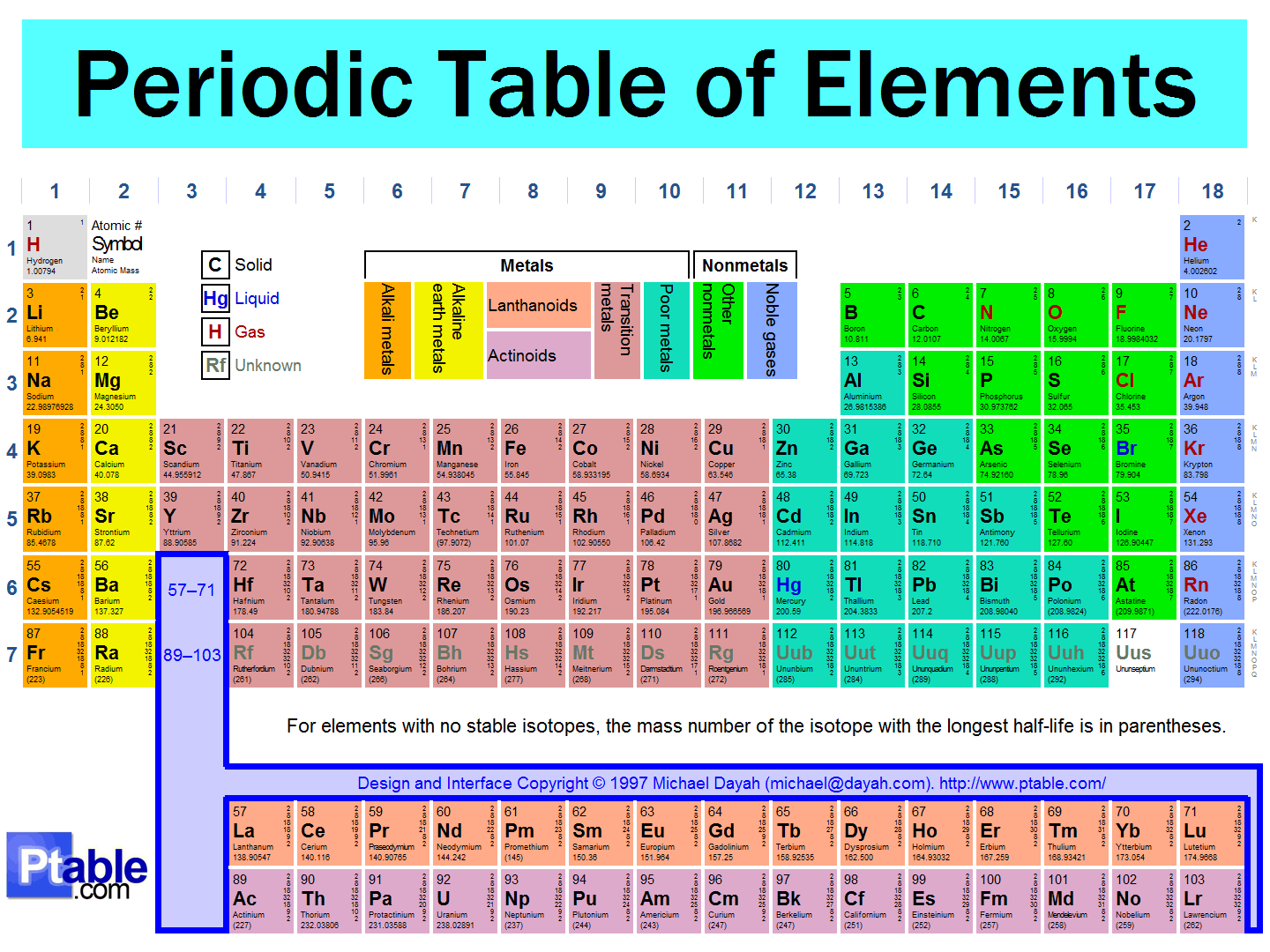
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
13
Multiple Choice
Elements in a .................. have similar chemical properties.
period
group
row
14

15

16
Multiple Choice
Which group of the periodic table is composed of inert (not reactive) gases?
answer choices
alkali metals
alkaline earth metals
halogens
noble gases
17

18

19

20
Multiple Choice
21
Multiple Choice

22

23
Reorder
A mixture contains sulfur powder, iron filings and solid table salt. Order the steps to show the correct sequence to separate the mixture
Use a magnet to remove the iron filings.
Add some water to dissolve the table salt.
Stir with a glass rod.
Filter the mixture.
Heat the filtrate to evaporate the water.
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice

28
Match
pure element
pure compound
mixture of elements
mixture of compounds
mixture of elements and compounds
pure element
pure compound
mixture of elements
mixture of compounds
mixture of elements and compounds
29
Multiple Choice

30




31
Multiple Choice
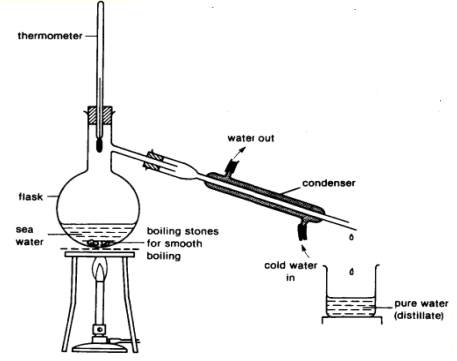
What is the name of this separation technique?
Simple distillation
Fractional distillation
Boiling
Evaporation
32
Multiple Choice
Which of the following statements tells us that a sample of liquid is pure water?
It is colourless.
It leaves no residue when filtered.
It boils at 100oC.
It is odourless.
33
Multiple Choice
Which separation technique is used to obtain pure water from seawater?
Simple distillation
Filtration
Chromatography
Evaporation
34
Multiple Choice
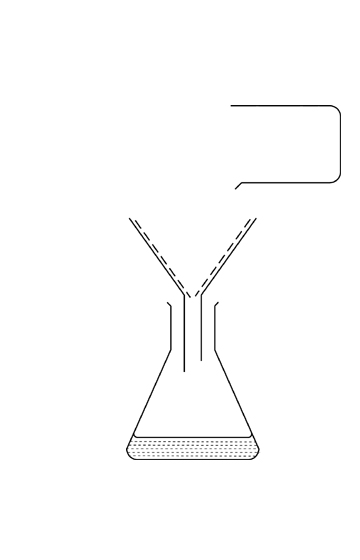
What is the name of this separation technique?
distillation
filtration
evaporation
chromatography
35
Multiple Choice
What is the physical property on which fractional distillation depends?
different boiling points
different melting points
different solubilities
different densities
36
Multiple Choice
For paper chromatography to work,
the dyes must have different boiling points.
the dyes must have different solubilities in the solvent.
the starting line on the chromatogram must be drawn using pen.
The starting line must be below the level of the solvent.
37
Multiple Choice
Which separation technique is used to obtain pure oxygen from liquid air?
Simple distillation
Filtration
Chromatography
Fractional distillation
38
Multiple Choice
Which separation technique is used to obtain salt from seawater?
Simple distillation
Filtration
Chromatography
Evaporation
39
Multiple Choice
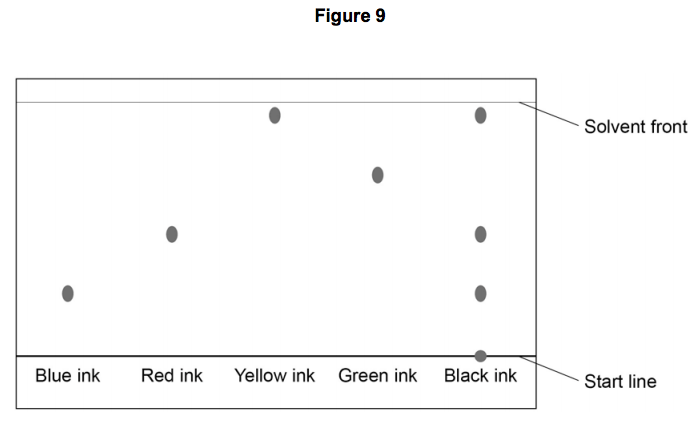
Which ink is most soluble?
Blue
Red
Yellow
Green
40
Multiple Choice

Which ink is a mixture?
Blue
Red
Yellow
Black
41
Multiple Choice

Referring to the chromatogram, black ink contains
Blue, red and yellow ink only
Blue, red, yellow and green ink
Blue and yellow ink
Black ink only
42
Multiple Choice
During filtration, the solid that is trapped in the filter paper is called ..A.. and the liquid that has passed through the filter paper is called ..B..
A: residue B: filtrate
A: residue B: filterate
A: filtrate B: residue
A: filterate B: residue
43

44

45
Multiple Choice
Which statement best describes the fact that gases can be compressed?
The particles in a gas do not have a fixed volume.
The particles in a gas are very far apart.
The particles in a gas have no force of attraction (bonds) between them.
The particles in a gas are constantly moving very rapidly.
46
Multiple Choice
Which statement best describes the fact that a liquid cannot be compressed?
The particles in a liquid are all close and touching.
The particles in a liquid are constantly moving.
The particles in a liquid have weak attractive forces between them.
The particles in a liquid can change position.
47
Multiple Select
Diffusion occurs in which states of matter?
Solids
Liquids
Gases
48
Multiple Choice

The three states of matter are represented by diagrams X, Y and Z. Which change occurs during condensation?
X to Y
X to Z
Y to X
Z to X
49
Multiple Choice
50
Multiple Choice
These particles are held together by very strong forces of attraction
Solid
Liquid
Gas
Plasma
51
Multiple Choice
The particles of solids ________________
vibrate about their fixed postions
are able to slide over each other
move about freely at high speeds and in random direction
do not move
52
Multiple Choice
The particles of liquids ________________
vibrate about their fixed postions
are able to slide over each other
move about freely at high speeds and in random direction
do not move
53
Multiple Choice
The particles of gases ________________
vibrate about their fixed postions
are able to slide over each other
move about freely at high speeds and in random direction
do not move
54
Multiple Select
Which statements describe the arrangement of particles in solids?
particles are moving in all directions
Particles are arranged in an orderly manner
particles can slide over one another.
particles vibrate about their positions
55
Multiple Select

The diagram represent the arrangement of particles in ____________.
gas
liquid
solid
56
Multiple Select

The diagram represent the arrangement of particles in ____________.
gas
liquid
solid
57
Multiple Select

The diagram represent the arrangement of particles in ____________.
gas
liquid
solid
58


59
Multiple Choice
What is atomic number?
Number of protons in an atom
Number of neutrons in an atom
Mass of an atom
Charge on an atom
60
Multiple Choice
61
Multiple Choice

What is the charge formed on the particle shown?
1+
0
1-
2+
62
Multiple Choice
The total number of protons and neutrons in an atom is called the...
Atomic number
Proton number
Nucleon number
Weight number
63
Multiple Choice
64
Multiple Choice
The __________ of an atom is the sum of the protons and neutrons in the nucleus of that atom.
nucleon number
atomic number
atomic weuight
isotope weight
65
Multiple Choice
66
Multiple Choice
For an atom to be electrically neutral, it must contain the same number of _____
protons and neutrons
neutrons and electrons
protons and electrons
nucleons and electrons
67
Multiple Choice
An atom of an element has the electronic configuration 2,8,5. Which group and period is the element placed in the Periodic Table?
Group 13, Period 3
Group 13, Period 5
Group 15, Period 3
Group 15, Period 5
68



Hydrogen Gas Test
Bring a lighted splint near the gas.
Flame extinguished with a 'pop' sound.
Hydrochloric acid
Nitric acid
Sulfuric acid
Sodium hydroxide
Potassium hydroxide
aqueous ammonia
69

Neutralisation
70


Test for CO2
Bubble gas into limewater.
White precipitate formed.
71
Multiple Choice
72
Multiple Choice
73
Multiple Choice
74
Multiple Choice
75
Multiple Choice
What type of reaction is the following:
HCl + Zn --> ZnCl2 + H2
Neutralisation
Acid and a metal
Acid and a carbonate
Physical reaction
76
Multiple Choice
Complete the following reaction:
Sulfuric acid + sodium carbonate
-->
Sodium sulfate + water + carbon dioxide
77
Multiple Choice
Hydrochloric acid + magnesium hydroxide -->
78
Multiple Choice
Bitter, slippery, pH above 7
Sour, reacts with metals, pH below 7
Sour, reacts with metals, pH above 7
Bitter, pH of 7, does not react with metals
79
Multiple Choice
Which of the following is the chemical formula of an acid?
NaOH
HCl
H2O
Ca(OH)2
80
Multiple Choice
Which of the following is the chemical formula of a base/alkali?
CaSO4
H2SO4
Ba(OH)2
KCl
81
Multiple Choice
What is the test for carbon dioxide produced from a reaction?
the burning splint test
the glowing splint test
bubbling the gas through limewater
82
Multiple Choice
Which gas gives out a 'pop' sound when a lighted splint is placed in it?
hydrogen
oxygen
carbon dioxide
nitrogen
83
New!
Chemical formula and equations!
Chemical bonding!
Preparing salts!
Mole calculations!
Redox reactions!
Metal reactivity series!
Period Table Group Properties!
Energetics!
Rate of reactions!
Electrochemistry!
Organic chemistry!
and more depth on sec 2 topics!
84

85


Secondary Three Chemistry

Show answer
Auto Play
Slide 1 / 85
SLIDE
Similar Resources on Wayground

83 questions
Spanish 3 - Week 1 - Unit 1 - Identity
Presentation
•
9th - 12th Grade

81 questions
26108-20 Section 1
Presentation
•
KG - University

80 questions
Lo más importante de las ciudades españolas
Presentation
•
9th - 12th Grade

79 questions
PS - Force and Motion
Presentation
•
8th - 11th Grade

79 questions
Geologic Processes
Presentation
•
9th - 12th Grade

80 questions
Prior Learning
Presentation
•
9th - 12th Grade

80 questions
Blood Vessels and Heart Anatomy
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

61 questions
End of the Year Trivia Fun
Quiz
•
9th Grade

15 questions
Protein synthesis
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade