

Ion, Isotopes, and AAM
Presentation
•
Science
•
11th Grade
•
Medium
+8
Standards-aligned
Sean Sandvick
Used 5+ times
FREE Resource
7 Slides • 30 Questions
1
Isotopes, Ions and Average Atomic Mass
By TROB

2
Ion Notation
Same as isotope
includes charge in upper right
+ means LESS ELECTRONS than PROTONS
(-) means MORE ELECTRONS than PROTONS

3
Ions
Ion means charged atom
To be charged it needs more or less electrons than protons
more electrons = NEGATIVE
less electrons = POSITIVE

4
ISOTOPES:
Same Element
Different Mass Number
WHICH MEANS DIFFERENT # of NEUTRONS

5
Average Atomic Mass
DIFFERENT than Mass Number
Average of all of the isotopes of an atom found in nature.

6
How to tell which isotope is more abundant?
CLOSEST TO ROUNDED AVERAGE ATOMIC MASS
So the most abundant isotope of Gold would be?
Gold - 197

7
Multiple Choice
8
Multiple Choice
What is the average mass of this element?
9
Multiple Choice
How many protons does this element have?
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice

How many electrons?
9
4
5
2
16
Multiple Choice

How many neutrons?
9
4
5
2
17
Multiple Choice
18
How they get that number
% abundance of that isotope x actual mass of that isotope
add together
divide by 100 (if you didn't put into percentage first)

19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice
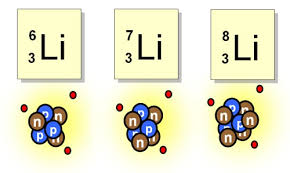
24
Multiple Choice

25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

28
Multiple Choice
An atom with the same number of protons and electrons (no charge) and the expected atomic mass or number of neutrons from the periodic table.
Neutral/Normal Atom
Ion
Isotope
29
Multiple Choice
An atom that has gained or lost electrons in an attempt to become stable/bond with another atom.
Normal/Neutral Atom
Ion
Isotope
30
Multiple Choice
A negatively charged ion that has gained electrons to satisfy the octet rule (metals do this)
Anion
Cation
Neutral Atom
Isotope
31
Multiple Choice
A positively charged ion that has lost electrons to satisfy the octet rule (non-metals do this)
Anion
Cation
Neutral Atom
Isotope
32
Multiple Choice
Atoms of the same element that have different mass than expected because they have a different number of neutrons.
Neutral Atom
Ion
Isotope
Cation
33
Multiple Choice
If an atom has 9 protons and 10 electrons it has a charge of ____.
0
-1
+1
-2
34
Multiple Choice
If an atom has 4 protons and 2 electrons it has a charge of ____.
0
-2
+2
+1
35
Multiple Choice
If an atom has a charge of +3 and an atomic number of 5 it should have _______ electrons.
1
2
3
4
36
Multiple Choice
If an atom has a charge of -2 and an atomic number of 16 it should have _______ electrons.
14
16
18
20
37
Multiple Choice
If an atom has a charge of 0 (neutral) and an atomic number of 8 it should have ____ electrons.
0
8
7
9
Isotopes, Ions and Average Atomic Mass
By TROB

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

29 questions
Plate Tectonics & Boundaries
Presentation
•
11th Grade

32 questions
Introduction to Equilibrium Chemistry
Presentation
•
11th Grade

32 questions
3.1 Cell Membrane & Nucleus 2025-26
Presentation
•
11th Grade

33 questions
Year 11 Science - Giant covalent structures
Presentation
•
11th Grade

25 questions
Physical and Chemical Changes Review
Presentation
•
8th Grade

32 questions
Periodic trends
Presentation
•
10th Grade

34 questions
Blood Types
Presentation
•
11th - 12th Grade

34 questions
Alternative Resources
Presentation
•
11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

49 questions
AP Environmental Science Final Exam Review
Quiz
•
10th Grade - University

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

28 questions
Greenhouse effect
Quiz
•
9th - 12th Grade