

Module 4 Study Guide
Presentation
•
Science
•
12th Grade
•
Easy
Michael Baker
Used 2+ times
FREE Resource
77 Slides • 249 Questions
1

2

3

4

5

6

7
Multiple Choice
8

9

10

11

12

13

14

15

16
Multiple Choice
17

18

19

20

21

22

23

24

25
Multiple Choice
26
Multiple Choice
27

28

29

30

31
Multiple Choice

A cat is hanging out several feet from a fire in a fireplace. This causes the kitty cat to get warm. The kitty cat is warm because of...
Convection
Conduction
Radiation
The fur sweater he's wearing!
32
Multiple Choice

A certain type of decorative lamp contains colored liquids. These liquids form globs that break off and rise to the top of the liquid. The globs rise due to
Convection
Conduction
Radiation
Globulation
33
Multiple Choice

Near the ceiling of a room, the air is warmer. The warmer air is rising because of
Convection
Conduction
Radiation
It was told to by this crazy fool!
34
Multiple Choice

In a swimming pool, the water near the surface is slightly warmer. The temperature of the water near the top rises because of
Convection
Conduction
Radiation
Too many dogs being in the pool!
35
Multiple Choice

A fireman that is at a building that is on fire feels a door and it is hot. Why does his hand feel hot?
Convection
Conduction
Radiation
The Human Torch was on the other side!
36
Multiple Choice

A huge boulder at the state park in Arizona gets so hot during a mid summer's day. This is due to...
Convection
Conduction
Radiation
Boogie Fever
37
Multiple Choice

A house burns down. On the house next to it, the vinyl siding is twisted and warped by the heat. The heat was transferred across to the other house by
Convection
Conduction
Radiation
Marvin the Martian
38
Multiple Choice

As you hold a piece of ice, it begins to melt from the warmth of you hand on it. This is because of
Convection
Conduction
Radiation
the fact that you are the Human Torch from the Fantastic Four.
39
Multiple Choice

In heat transfer, heat always flows from the _______________ object to the _______________ object.
hotter / colder
colder / hotter
hotter / hotter
colder / colder
40
Multiple Choice

Which of the following statements is true regarding heat energy?
The quicker a molecule moves, the colder it is.
The slower a molecule moves, the warmer it is.
The quicker a molecule moves, the warmer it is.
The faster you dance, the quicker you get tired.
41
Multiple Choice
42
Multiple Choice
43
Match
Match the following Types of heat transfer to the appropriate example of it
Radiation
Conduction
Convection
Radiation
Conduction
Convection
44
Multiple Choice
Heat transfer by convection happens when:
there is a circulation of hot air/water rising and cold air/water sinking
there is space that heat travels through in waves outwardly
two objects are touching and heat moves from one object into the next object.
45
Multiple Choice
Heat transfer by radiation happens when:
there is a circulation of hot air/water rising and cold air/water sinking
two objects are touching and heat moves from one object into the next object
there is space that heat travels though in waves outwardly
46
Match
Thermal Energy
Conductor
Insulator
Thermal Energy
Conductor
Insulator
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice
50
Multiple Choice
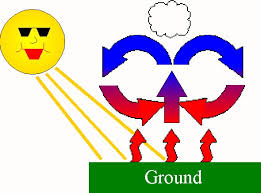
51
Multiple Choice
52
Multiple Choice
53
Multiple Choice

54
Multiple Choice
55
Multiple Choice
56
Multiple Choice
than in hot things.
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice

62
Multiple Choice
63
Multiple Choice
If you walk barefoot on hot asphalt, energy is transferred by which process?
Convection
Radiation
Conduction
Reflection
64
Multiple Choice

Throughout the day, Matthew and Mei measure the temperature of lake water as it is warmed by the sun. The following table shows their data at four different times.
At what time was the thermal energy of the lake water the greatest?
9 a.m.
11 a.m.
3 p.m.
5 p.m.
65
Multiple Choice
When energy from the sun reaches the air above land, the air warms and rises. Along a coastline, cooler air above the ocean flows toward the land to replace this rising air.
Which of the following best describe these processes?
Conduction and convection
Radiation and convection
Conduction, convection, and radiation
Radiation and conduction
66
Multiple Choice

Kumar is studying how Earth is warmed by sunlight. The following illustration shows his model of the sun and Earth.
Which process is mostly responsible for warming the soil in Kumar’s model?
Conduction between the lamp and the soil causes the soil to warm.
Convection currents between the lamp and the soil cause the soil to warm.
Expansion of particles in the soil causes the soil to warm.
Radiation from the lamp reaches the soil and causes the soil to warm.
67
Multiple Choice

Vivian placed an ice cube in a tray of warm water. The following picture shows her investigation. Which statement describes the energy transfer that happens in Vivian’s investigation?
Energy in the form of heat is transferred from the ice cube to the water by radiation.
Energy in the form of heat is transferred from the water to the ice cube by radiation.
Energy in the form of heat is transferred from the ice cube to the water by conduction.
Energy in the form of heat is transferred from the water to the ice cube by conduction
68
Multiple Choice
Sarah is investigating the transfer of energy in the form of heat. One process that transfers energy between two objects is conduction.
Which statement is NOT true about conduction?
It involves a transfer of energy in the form of heat.
It can transfer energy through empty space.
It requires direct contact between the objects.
It results in a change of temperature in both objects.
69
Multiple Choice

The following diagram shows water in contact with an ice cube and warmer air.
Which way will energy flow?
From the air to the water
From the water to the air
From the ice cube to the air
From the ice cube to the water
70
Multiple Choice
A warm rock is in direct contact with the cooler ground.
Which of the following will happen?
Energy in the form of heat will flow from the rock to the ground, and the temperature of the rock will increase
Energy in the form of heat will flow from the ground to the rock, and the temperature of the rock will increase
Energy in the form of heat will flow from the rock to the ground, and the temperature of the rock will decrease.
Energy in the form of heat will flow from the ground to the rock, and the temperature of the rock will decrease.
71
Multiple Choice
72
Multiple Choice
Energy is transferred as heat between two objects of ___________ temperatures.
differing
same
73
Multiple Choice
The measure of average kinetic energy of all the particles within an object is called ____________.
temperature
conduction
radiation
heat
74
Multiple Choice

75
Multiple Choice
76
Multiple Choice

77
Multiple Choice

78
Multiple Choice

79
Multiple Choice
80
Multiple Choice

81
Multiple Choice
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
85
Multiple Choice
86
Multiple Choice
87
Multiple Choice
88
Multiple Choice
89
Multiple Choice
90
Multiple Choice
91

92
Multiple Choice
93
Multiple Choice
94
Multiple Choice

What type of reaction occurs in a hand warmer?
exothermic
endothermic
95
Multiple Choice
What is the transfer of energy between objects at different temperatures called?
Heat
Temperature
Kinetic energy
Specific heat
96
Multiple Choice
What is the relationship between heat and temperature?
Heat is a measure of how hot or cold something is, while temperature is the transfer of energy
Heat and temperature are the same thing
Heat is the transfer of energy, while temperature is a measure of how hot or cold something is
Heat and temperature have no relationship
97
Multiple Choice
In an endothermic reaction, energy is _________.
absorbed
released
98
Multiple Choice

_____ reactions usually feel hot!
endothermic
exothermic
99
Multiple Choice

____ reactions usually feel cold.
endothermic
exothermic
100
Multiple Choice
Endo means in or _______.
absorbed
released
101
Multiple Choice
Exo means out or ________.
absorbed
released
102
Multiple Choice
What instrument would you use to detect an endothermic or exothermic reaction?
triple beam balance
ruler
thermometer
103
Multiple Choice
104
Multiple Choice
When a candle is lit hydrocarbons are combusting. An exothermic reaction is occuring. Energy is being _________________ .
absorbed
frozen
destroyed
released
105

106

107

108
Multiple Choice
109
Multiple Choice
110
Multiple Choice

111

112

113
Multiple Choice

114
Multiple Choice
it requires more energy to change temperature
it requires less energy to change temperature
115
Multiple Choice
You heat up 2 pans of water; one has 100 mL in it, and the other has 1000 mL. Which one heats up the fastest?
100 mL
1000 mL
They both heat up at the same speed
116
Drag and Drop

117
Multiple Choice
Which of the following best explains why the sand at the beach is hotter than the water?
Sand has a higher specific heat than water.
Sand has a lower specific heat than water.
Sand has a greater mass than water.
Water has a greater mass than sand.
118
Fill in the Blanks
119
Multiple Choice

Copper, Stainless Steel, Carbon Steel, and Zinc were all heated using the same amount of thermal energy. What material would be the coolest after being heated?
Copper
Carbon Steel
Zinc
Stainless Steel
120
Multiple Choice
Object A, it has a lower specific heat
Object B, it has a lower specific heat
Object A, it has a higher specific heat
Object B, it has a higher specific heat
121
Multiple Choice
Which one will have the SMALLEST change in temperature?
122
Multiple Choice
Heat will always transfer from the ___________ object to the ________ object.
The hottest object to the coolest object.
The coolest object to the hottest object.
The solid object to the liquid object.
The liquid object to the solid object.
123
Multiple Choice

Thermal energy
Temperature
Heat
Convection
124
Drag and Drop

125
Multiple Choice
126
Multiple Choice
127
Multiple Choice
128
Multiple Choice
129
Multiple Choice
130
Multiple Choice
131
Multiple Choice
132
Multiple Choice
133
Multiple Choice
If two objects have different temperatures when they come in contact, heat will flow from the warmer object to the cooler one UNTIL ____________
one reaches a temperature of zero
they both have an equal temperature
one runs out of energy
134
Multiple Choice
For a skillet, used for cooking, do you want a high or low specific heat
High, so that it will need more energy to heat up
Low, so that it will change temperature quickly
135
Multiple Choice

Which material would require the greatest amount of heat energy to raise its temperature from 5oC to 10oC? [Refer to the Earth Science Reference Tables.]
Iron
Lead
Granite
Dry Air
136
Multiple Choice

If the same amount of heat is added to each sample, the smallest change in temperature will occur in the
Iron
Dry Air
Granite
Water
137
Multiple Choice
Why does water heat heat up and cool down VERY SLOWLY?
High Specific Heat
Low Specific Heat
It's solid
It's always cold
138
Multiple Choice
139
Multiple Choice
140
Multiple Choice
Heat ALWAYS moves from
Low to High Temperature
High to Low Temperature
It doesn't move
High to Low Pressure
141
Multiple Choice
Specific Heat is....
The speed of the particles in a substance
The temperature of the substance
How much energy it takes to heat something up
Heat
142
Multiple Choice
When heat is absorbed by a substance, what happens to the temperature of the substance?
it increases
it stays the same
it decreases
it depends on the substance
143
Multiple Choice
When heat is released by a substance, what happens to its temperature?
it increases
it stays the same
it decreases
it depends on the substance
144

145

146

147

148

149

150

151

152

153

154

155

156

157

158

159
Multiple Choice
In the following equation, the symbol for specific heat is .......
Q = mcΔT
c
Q
m
ΔT
160
Multiple Choice
What unit is used to measure Thermal Energy?
J/g ºC
g
ºC
J
161
Multiple Choice
You should use scratch paper and a calculator
If you add 50 J of energy to a piece of iron, and the temperature rises (changes) by 25.0°C, what is the mass of the iron? (Iron has a specific heat of 0.10 J/g°C)
25 g
30 g
20 g
50 g
162
Multiple Select
How does heat flow, or get transferred?
(CHECK ALL STATEMENTS THAT ARE CORRECT)
Always from the colder object to the warmer object
Always from warmer object to the colder object
From the particles with more kinetic energy to the ones with less kinetic energy
From the particles with less kinetic energy to the ones with more kinetic energy
163
Multiple Choice
A high specific heat means...
a substance requires less energy for its temperature to change
a substance requires more energy for its temperature to change
a substance heats up very quickly
the substance's particles have a high average kinetic energy
164
Multiple Choice
You should use scratch paper and a calculator
The temperature of 20 grams of water changes from 25° C to 20° C. How much heat energy moved from the water to the surroundings? (The specific heat of water is 4.18 J/g°C)
418 J
209 J
83 J
4.18 J
165
Multiple Choice
The unit for temperature is
K or °C
J or cal
g°CJ or g°Ccal
g or kg
166
Multiple Choice
What does ΔT represent?
tf −ti
ti −tf
K = °C+273
celsius - t
167
Multiple Choice
168
Multiple Choice
169
Multiple Choice
How many joules of heat are needed to raise the temperature of 10.0 g of aluminum from 22.0°C to 55.0°C, if the specific heat of aluminum is 0.903 J/g°C?
298 Joules
0.003 Joules
297 J/g°C
0.003 J/g°C
170
Multiple Choice
20.0 g of water. specific heat of water is 4.184 J/g°C. temperature changes from 25.0° C to 20.0° C, how much heat energy (Q) moves from the water to the surroundings?
418 Joules
209 J
83 J
4.18 J
171
Multiple Choice
What does ΔT represent?
tf −ti
ti −tf
t - celsius
celsius - t
172
Multiple Choice
When is your answer negative?
When the sample is cooled or loses heat energy.
When the sample heats up or gains heat energy.
When the sample absorbs heat energy.
173
Multiple Choice
What letter do we use to represent change in specific heat?
Q
m
c
ΔT
174
Multiple Choice
What letter do we use to represent change in temperature?
Q
m
c
ΔT
175
Multiple Choice
What letter do we use to represent mass?
Q
m
c
ΔT
176
Multiple Choice
The unit for mass is
g or Kg
J
J/gC
°C
177
Multiple Choice
The unit for temperature is
K or °C
J
J/gC
g or kg
178
Multiple Choice
The unit for heat energy is
°C
J
J/gC
g
179
Multiple Choice
The unit for specific heat is
J
J/gC
g
°C
180
Multiple Choice
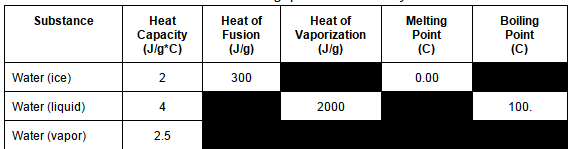
181
Multiple Choice

182
Multiple Choice

183
Multiple Choice
How many joules of heat are needed to raise the temperature of 10.0 g of aluminum from 22.0°C to 55.0°C, if the specific heat of aluminum is 0.903 J/g°C?
298 Joules
0.003 Joules
297 J/g°C
0.003 J/g°C
184
Multiple Choice
The specific heat of aluminum is 0.9025 J/g°C. How much heat(Q) is released when a 10.0 g piece of aluminum foil is taken out of the oven and cools from 100.0° to 50.0°?
451 J
45.1 J
400 J
185
Multiple Choice
186
Multiple Choice
187
Multiple Choice
188
Multiple Choice
189
Multiple Choice
Determine the specific heat of an unknown object that has a mass of 15g, released 2050J of heat and had a temperature change of 50C to 75C.
5.46 J
5.5 J/g ° C
768750 J
768750J/g ° C
190

191

192

193

194

195

196

197

198

199

200

201

202

203

204

205

206

207

208

209

210

211

212

213

214

215

216

217

218

219

220
Multiple Choice
221
Multiple Choice
222
Multiple Choice
223
Multiple Choice
224
Multiple Choice

225
Multiple Choice
226
Multiple Choice
227
Multiple Choice
228
Multiple Choice
229
Multiple Choice
230
Multiple Choice
231
Multiple Choice
232
Multiple Choice
233
Multiple Choice
234
Multiple Choice
235
Multiple Choice
236
Multiple Choice
237
Multiple Choice
238
Multiple Choice
239
Multiple Choice
240
Multiple Choice
241
Multiple Choice

242
Multiple Choice
243
Multiple Choice

244
Multiple Choice

245
Multiple Choice

246
Multiple Choice
247
Multiple Choice

248
Multiple Choice
249
Multiple Choice
250
Multiple Choice
251
Multiple Choice
252
Multiple Choice
253
Multiple Choice
254
Multiple Choice
255
Multiple Choice
256
Multiple Choice
257
Multiple Choice
258
Multiple Choice
259
Multiple Choice
260
Multiple Choice
261
Multiple Choice

262
Multiple Choice

263
Multiple Choice

264
Multiple Choice

265
Multiple Choice

266
Multiple Choice

267
Multiple Choice
The picture showing the energy conversion
Chemical → heat
is:
268
Multiple Choice
The picture showing the energy conversion
Light → chemical
is:
269
Multiple Choice
The picture showing the energy conversion
Chemical → light & heat
is:
270
Multiple Choice

271
Multiple Choice
A washing machine turns electrical energy into
Light energy
Chemical energy
Mechanical energy
272
Multiple Choice
273
Multiple Choice
Which law is this?
274
Multiple Choice
275
Multiple Choice
276
Multiple Choice
277
Multiple Choice
278
Multiple Choice

279
Multiple Choice

280
Multiple Choice

281
Multiple Choice

282
Multiple Choice

283
Multiple Choice

284
Multiple Choice

Pick the BEST Energy Conversion below for a Hydroelectric Dam.
285
Multiple Choice

A Steam Locomotive uses wood or coal, to create steam from water. This steam moves pistons, and thus, the train. Choose the best energy transformation below for a steam locomotive.
Thermal --> Mechanical --> Chemical (plus heat)
Chemical --> Thermal --> Mechanical (plus heat)
Mechanical --> Chemical --> Thermal (plus heat)
Electrical -> Chemical --> Thermal (plus heat)
286
Multiple Choice

Which location will a pendulum have the most kinetic energy?
the left side
the right side
The center/middle
either the right or left side
287
Multiple Choice

288
Multiple Choice
289
Multiple Choice

A hot air balloon rising is an example of ...
conduction
convection
radiation
connection
290
Multiple Choice

Heat always travels from...
Warm to cool
Cool to warm
Warm to warm
Cool to cool
291
Multiple Choice

A snowman melting in the sun
convection
conduction
radiation
connection
292
Multiple Choice

Heating a kettle on a hot stove (burner)
convection
conduction
radiation
connection
293
Multiple Choice

A stove top will cook your food because of ___________.
convection
conduction
radiation
connection
294
Multiple Choice

At what point does the biker have the greatest Kinetic Energy?
Point A
Point B
Point C
Point D
295
Multiple Choice
296
Multiple Choice
Heat moves by ______ when it travels by waves and doesn't require matter.
radiation
conduction
insulator
convection
297
Multiple Choice
298
Multiple Choice
299
Multiple Choice
To measure the temperature difference between two cups of water, you would use a _____.
calorimeter
ruler
thermometer
balance scale
300
Multiple Choice
301
Multiple Choice
302
Multiple Choice

303
Multiple Choice
304
Multiple Choice
305
Multiple Choice
306
Multiple Choice
307
Multiple Choice
308
Multiple Choice
309
Multiple Choice
310
Multiple Choice
Specific heat of water is 4,184 J/kg°C. Specific heat of wood is 1,760 J/kg°C What material needs more heat energy to raise the temperature?
Water
Wood
Both are same
311
Multiple Choice
312
Multiple Choice
313
Multiple Choice
Which of the following is an example of convection?
Touching a hot hair dryer
Laying down on a cold floor.
When smoke rises from a fire.
When you are making noodles and the water is boiling
314
Multiple Choice
315
Multiple Choice

316
Multiple Choice

317
Multiple Choice

318
Multiple Choice

319
Multiple Choice

320
Multiple Choice
321
Multiple Choice
322
Multiple Choice

323
Multiple Choice

324
Multiple Choice
325
Multiple Choice
326
Multiple Choice

Show answer
Auto Play
Slide 1 / 326
SLIDE
Similar Resources on Wayground

283 questions
TKA SMA
Presentation
•
9th - 12th Grade

284 questions
Untitled lesson
Presentation
•
6th Grade

272 questions
F5 Core 6 Revision Exercise on Unit 6.3 and 6.4
Presentation
•
11th Grade

301 questions
Python 2
Presentation
•
10th Grade - University

316 questions
7-Physical Science Review-TCAP
Presentation
•
6th - 8th Grade

296 questions
sory
Presentation
•
10th Grade

343 questions
ÔN TẬP CHƯƠNG 1
Presentation
•
7th Grade

291 questions
Untitled lesson
Presentation
•
7th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade