

Phy Sci Sem 1 Exam Review
Presentation
•
Science
•
10th Grade
•
Medium
John Oglesby
Used 1+ times
FREE Resource
50 Slides • 89 Questions
1
Physical Science
Semester 1 Exam Review - 2023-24
2
Topic Questions Pts/each Total
Metric System 2 2 4
Properties & Change 5 2 10
Per. Table Organization 8 2 16
Ionic Bonding 10 2 20
Covalent Bonding 8 2 16
Acids 4 2 8
Essay Questions 2 5 10
total 47 100
3
Metric System
Units of Measure
4
Base Units
Volume = Liters = L
Mass = Grams = g
Length = meters = m

5
The Metric System

6
Prefixes
The Metric system is based on multiples of tens.

7
Remember
“King Henry Doesn’t (Usually) Drink Chocolate Milk”

8
Multiple Select
Which units below are NOT part of the metric system? Check all that apply.
Inches
Feet
Meters
Grams
9
Multiple Select
Which units are the BASE units in the metric system? Check all that apply.
Meters
Liters
Grams
Inches
Miles
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Converting Units
Changing a Metric Units to another Metric Unit
Moving BIG unit to small Multiply
Moving small to BIG Divide

14
Multiple Choice
42 L = ? mL
4,200
.0042
4.20
42,000
15
Multiple Choice
16
Multiple Choice
Convert:
3 kg = ? g
3
30
300
3,000
17
Physical Properties of an Element
Physical Property: Characteristics that can be observed or measured without changing the composition (identity) of a substance
You don't have to change what an apple is made of to see that it is red and being able to hold it in your hand

18
Multiple Select
Density is a physical property that describes the relationshipep between the ________ of its _________.
matter, volume
mass, volume
volume, matter
none of the above
19
Physical Propeties
Density is a property that describes the relationship between the mass of a material and its volume. It does not change the substance.
Substances that have a higher density contain more matter (stuff) in a given volume.
The density of a substance will stay constant/the same.

20
Multiple Choice
The physical property melting point is the
temperature at which a liquid changes to a solid
temperature at which a liquid boils
temperature at which a solid can change to a liquid
21
Multiple Choice
The tranisition metal copper has luster. This means
is is very dull
it is very shiny
it is very thin
it is very dirty
22
Physical Property-Boiling Point
The temperature at which liquid boils.
A substance can change from a liquid to a gas.
Boiling temperature is unchanging under constant conditions for a given substance.

23
Open Ended
Explain malleable and ductile metals. What can be done to them?
24
Physical Properties of Metals
Good conductor of heat and electricity.
Conductivity- does it heat up or allow electricity to pass through it.
Is it ductile- can it be stretched out into a wire.
Can it be hammered into sheets or pounded into shapes- Malleable.
Do they have luster- are they Shiny?
They are solid at room temperature. Except for Mercury (Hg).

25
Multiple Choice
Which is a Physical property?
A metal can react with water to become a metal oxide (rust)
A metal can be hammered into a shape (malleable)
26
Multiple Choice
Which is a Chemical property
A metal can react with water to form a metal oxide (rust)
A metal can be hammered into a shape (malleable)
27
Chemical Properties
Flammability- the ability of a chemical to burn or ignite.
Combustibility-how easily a substance can burst into flames, through fire or combustion.
Toxicity-an elements ability to become toxic or poisonous
pH- how acidic or basicity something is. Acidic can have a sour taste, smell that burns, and a sticky texture. Basicity can be bitter like baking soda, no smell, and slippery.

Describes the ability of a substance to react with another substance to become something else
Magnesium can react with oxygen (burn) to become magnesium oxide (let me demonstrate)
28
Chemical Properties Continued
Reactivity- the ability of matter to react chemically with other substances.
Oxidize- ability to react with oxgyen
Ability of substances to react with acids or water but not all substances do this.
29


Anther physical property is
MAGNETISM
• Whether or not an
object is attracted
to a magnet
30

More EXAMPLES -
Physical
• size, shape, freezing point, boiling
point, melting point, magnetism,
viscosity, density, luster and many
more.
– Viscosity - The resistance of a liquid to
flowing.
– Examples:
– Low viscosity-water, rubbing alcohol
– High viscosity-honey
31



Chemical Properties
• When a substance interacts with
another substance to change its
chemical make-up.
32

Chemical properties include:
• Flammability
• Rusting
• Reactive to water
• pH
• odor
• Produces gas bubbles
• New product is created
33


Flammability
the ability to catch on fire
34
Multiple Choice
Which is an example of a chemical change?
ripping paper
chopping wood
baking a cake
stretching a rubber band
35
Multiple Choice
If I take a piece of paper and rip it up into pieces, is that a physical or chemical change?
Chemical
Physical
36

37
Multiple Choice
A Log chopped into firewood is an example of a _________ change
Physical
Chemical
38

39
Multiple Choice
Burning firewood in the fireplace is an example of a ________ change
Physical
Chemical
40
Atomic Structure

41
Atomic Structure

42
Electron

43
Multiple Choice
What is the charge of a proton?
Negative
Positive
Neutral
44

45

46
Multiple Choice
What is the charge of a neutron?
Negative
Positive
Neutral
47

48
Open Ended
What are the particles circulating around the Nucleus
49

50

51
Multiple Choice
A charged atom is called a(n)...
electron.
ion.
proton.
acid.
52
Atoms & their parts
The center of the atom is called the nucleus. The nucleus of most atoms is made up of two kinds of particles, protons & neutrons. Protons carry a positive (+) charge. Charge is an electrical property that can by either positive or negative.
Neutrons have no charge. The masses of protons and neutrons are measured in the atomic mass units (amu). Each proton or neutron has a mass of about 1 amu.
Electrons are subatomic particles that exist in an area outside the nucleus called the electron cloud. Electrons have a negative (-) charge. The mass of electrons is insignificant compared to the mass of protons & neutrons.

53
Multiple Choice
Which of the following parts of an atom are positively charged?
neutron
proton
electron
nucleus
54
Multiple Choice

What is the charge on an electron?
+1
0
-1
-2
55
Multiple Choice

What is the centre of an atom called?
Nucleus
Electron
Proton
Neutron
56
Multiple Choice
1) What is the charge of the nucleus of an atom?
negative
positive
neutral
none
57
Multiple Choice

What word describes two atoms of the same element but a different number of neutrons.
Isomer
Isolate
Isotope
Isopale
58
Multiple Choice

This is a neutrally-charged lithium ion. It has 3 protons and 3 neutrons. How many electrons does it have?
9
0
6
3
59
Multiple Choice
60
Multiple Choice

If the atomic number is 7 and the mass number 17 how many protons does the atom have?
17
7
10
0
61
Multiple Choice
62
Multiple Choice
Which of the following determines the identity of an element?
number of protons
atomic mass
number of neutrons
number of shells
63
Multiple Choice
Valence electrons are the...
Innermost electrons
Middle electrons
Outermost electrons
Any electrons
64

65
Multiple Choice
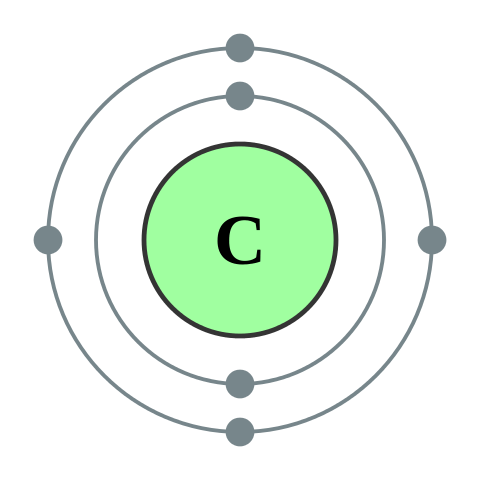
How many valence electrons does carbon have?
4
5
6
7
66

67
Multiple Choice

2
8
3
13
68

69

70
Multiple Choice
Which element is similar to Li?
Be
K
71
Multiple Choice

How many valence electrons does an atom of Fluorine have?
11
2
9
7
72
Multiple Choice
Which element is similar to Mg?
Ca
B
73
Groups & Periods
Groups: Columns; up & down, similar properties. ⬆️ ⬇️
Periods: Rows; left to right; increasing atomic ➡️ ⬅️

74
Multiple Choice
Students in Mrs. Clark's science class are investigating a substance that has the following properties
-shiny
-bends easily
-hard, not easily broken
Based on these properties how would you classify the substance?
metal
nonmetal
metalloid
75
Multiple Choice
The majority of the periodic table is made up of
Metals
Nonmetals
Metalloids
Noble gases
76
Multiple Choice
Which element is similar to F?
Ne
Cl
77
Multiple Choice

78
Metals, Nonmetals, Metalloids
Metals: ductile, malleable, luster, solids
Metalloids: properties of metals & nonmetals
Nonmetals: brittle & dull

79
Multiple Choice

80
Multiple Choice
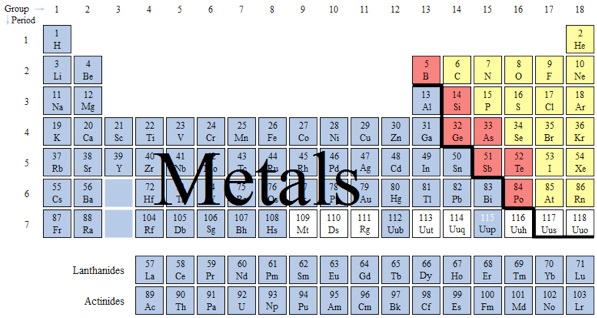
Which color identifies as the group non-metals?
neither color
blue
red
yellow
81

82

83
Multiple Choice
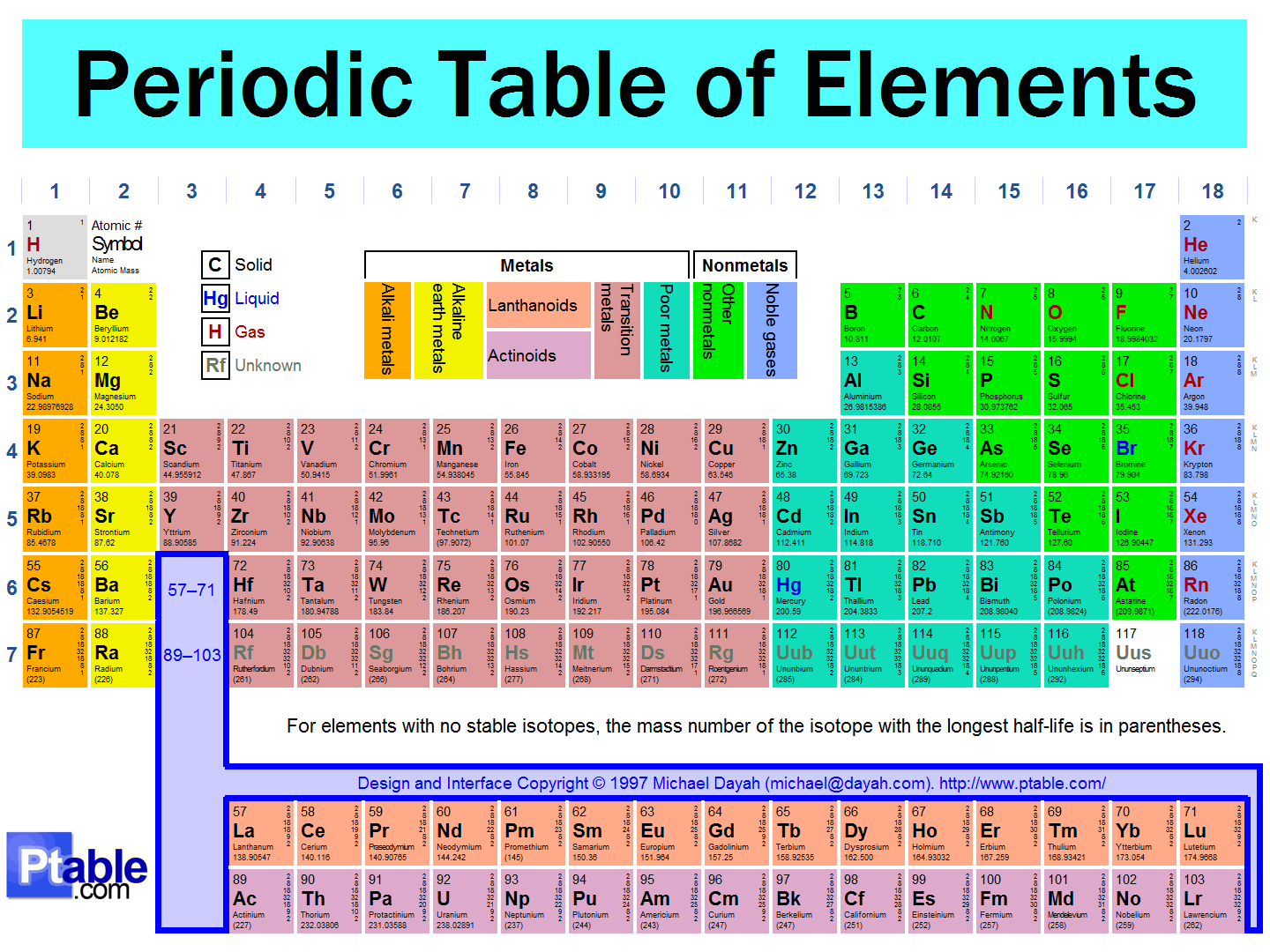
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
84

85
Multiple Choice

How many valence electrons are found in atoms of group 1?
7
6
4
1
86
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does Phosphorus have?
31
5
15
4
87
Multiple Choice

How many valence electrons does carbon have?
4
5
6
7
88
Multiple Choice
If we combine Zn2+ with O2- , what will be the chemical formula and name of the compound?
Zn2O2 -zinc dioxide
ZnO - zinc oxygen
ZnO2 - zinc (II) oxide
ZnO - zinc oxide
89
Naming ionic compounds is pretty simple.
Cation + Anion = ionic compound

90
Multiple Choice
91
Multiple Choice
Lithium Chloride
LiCl
Li3Cl
LiClO3
LiClO4
92
Multiple Choice
AlPO4
Aluminum Phosphide
Aluminum Phosphoxide
Aluminum Phosphorus Oxide
Aluminum Phosphate
93
Aluminum + Chlorine
Aluminum ion has a 3+ charge after giving its valence electrons to Chlorine
Chlorine has 7 valence electrons on each atom, only having room for 1 more
This electron transfer forms Chloride ions
It takes 3 Chlorine atoms to absorb the electrons from Aluminum
Aluminum Chloride is formed
AlCl3

94
Multiple Choice
95

96
Multiple Choice
Cobalt (II) Oxide
Co2O
CoO2
CoO
Co2O2
97
Fill in the Blanks

98
Match
Match each compound with the correct name
Fe(NO3)3
Fe(NO3)2
Fe(NO2)3
Fe(NO2)2
iron(III) nitrate
iron(II) nitrate
iron(III) nitrite
iron(II) nitrite
iron(III) nitrate
iron(II) nitrate
iron(III) nitrite
iron(II) nitrite
99
Multiple Choice
Which of the following is the correct ionic formula for a Barium and Phosphate compound?
Ba(PO4)
Ba3(PO4)
Ba3(PO4)2
Ba2(PO4)3
100
Match
Match the following compounds with the correct name
Au3N
AuN
AuNO3
AuNO2
Au(NO3)3
gold(I) nitride
gold(III) nitride
gold(I) nitrate
gold(I) nitrite
gold(III) nitrate
gold(I) nitride
gold(III) nitride
gold(I) nitrate
gold(I) nitrite
gold(III) nitrate
101
Naming compounds with Transition Metals
Use a roman numeral after the cation to show the charge of the transition metal
Fe3+ is named Iron (III)
Fe2+ is named Iron (II)
Everything else stays the same

102
Examples: Chromium and Oxygen
Chromium 2+ becomes Chromium (II)
Cr2+ + O2- --> CrO
Chromium (II) oxide
Chromium 3+ becomes Chromium (III)
Cr3+ + O2- --> Cr2O3
Chromium (III) oxide
103
Multiple Choice
FeCl3
Iron (III) Chloride
Iron (III) Chlorate
Iron (II) Chloride
Iron (II) Chromate
104
Mixed Practice
Binary Ionic
Ionic with Polyatomics
Ionic with Transition Metals
Iionic with Transition metals and Polyatomics
105
Multiple Choice
Name the following ionic compound: MgSO4
magnesium (II) sulfide
magnesium sulfide
magnesium sulfate
magnesium (II) sulfate
106
Multiple Choice
107
Multiple Choice
Name the following compound: FeCl3
iron chloride
iron III chloride
iron II chloride
iron III chlorate
108
Multiple Choice
The formula for Magnesium phosphide is:
Mg3(PO4)2
MgP
MgPO4
Mg3P2
109
Multiple Choice
What is the formula for manganese(III) oxide?
MnO
MnO3
Mn3O
Mn2O3
110
Multiple Choice
WHAT IS THE FORMULA FOR COPPER(II) SULFATE?
CuS
Cu(SO4)4
CuSO4
CuSO
111
Multiple Choice
WHAT IS THE FORMULA FOR LITHIUM OXIDE?
LiO2
Li2O
Li2O3
LiO
112
Multiple Choice
WHAT IS THE NAME FOR Ti(ClO3)2
TITANIUM CHLORATE
TITANIUM CHLORIDE
TITANIUM(II) CHLORATE
TITANIUM(II) CHLORIDE
113
Multiple Choice
114
Multiple Choice
WHAT IS THE NAME FOR Cr(NO3)2?
CHROMIUM NITRATE
CHROMIUM(II) NITRATE
CHROMIUM DINITRATE
CHROMIUM(II) NITRIDE
115
Multiple Choice
116
G10 Review on Covalent Bonding
Unit 3 Session 2

117
Multiple Choice
118
Covalent Compounds
A covalent compound is usually composed of two or more nonmetal elements.
119
Multiple Choice
120

121
Multiple Choice
122
Multiple Choice
What is the correct chemical formula for sulfur hexafluoride?
SF6
S6F6
S6F
SF
123
Multiple Choice
Name the molecular compound CO
carbon monoxide
monocarbon monoxide
carbide
oxygen carbide
124
Fill in the Blanks
125
Multiple Choice
Name the following compound:
CO2
monocarbon dioxide
carbon oxide
carbon dioxide
oxygen carbonide
126
Multiple Choice
Covalent Bonds are between...
a metal and a metal
a metal and a non-metal
a nonmetal and a nonmetal
127
Multiple Choice
128
Multiple Choice
What is the name for SiCl4?
silicon tetrachloride
silicon quadchloride
monosilicon tetrachloride
silicon chloride
129
Naming Binary Acids
Contains just two elements
One of those elements is hydrogen
Use prefix "hydro" and suffix "ic"
HCl = hydrochloric acid
HF = Hydrofluoric acid
130
Multiple Choice
What is the formula for hydrobromic acid acid?
HBr
HB
HBrO3
HBO3
131
Multiple Choice
What is the formula for hydroiodic acid acid?
HIO2
HIo
HI
IO2
132
Multiple Choice
Carbon and Oxygen will make a ___________ bond.
ionic
covalent
metallic
133
Acid with Polyatomic Ions
NO "HYDRO" IN NAME
Use polyatomic ion list to identify anion
Change "ate" to "ic"
Change "ite" to "ous"
134
Multiple Choice
Beryllium and sulfur will form a ______ bond
ionic
covalent
metalloid
135
Multiple Choice
Name the acid: HC2H3O2
Acetic Acid
Acetous Acid
Hydroacetic acid
Acetate Acid
136
Multiple Choice
What is the correct name of HClO3?
hydrochloric acid
chloric acid
chlorous acid
hydrochlorous acid
137
Multiple Choice
Name the following: CO
Carbon Oxide
Carbon Monoxide
Carbon(II) Oxide
138
Multiple Choice
What is the formula for nitrous acid?
HNO2
H3NO2
H2NO2
HNO3
139
Multiple Choice
carbonic acid
H2CO3
H2CrO4
H2C2O4
HCO3
Physical Science
Semester 1 Exam Review - 2023-24
Show answer
Auto Play
Slide 1 / 139
SLIDE
Similar Resources on Wayground

132 questions
Holt Ch 18
Presentation
•
9th - 12th Grade

130 questions
Human Innovations
Presentation
•
9th Grade

139 questions
Farang Farang
Presentation
•
9th - 12th Grade

140 questions
connection
Presentation
•
KG

139 questions
Unit 2 Study Guide
Presentation
•
KG

134 questions
ELPAC Practice
Presentation
•
KG - University

133 questions
Marine Science Ch 5
Presentation
•
9th Grade

138 questions
INFINITIVES-GERUNDS
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

40 questions
NCFE Earth and Environmental Science Released Test
Quiz
•
9th - 12th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

20 questions
Final Review Thermochemistry
Quiz
•
9th - 12th Grade

10 questions
Exploring Animal Adaptations: Physical, Behavioral, and Life Cycle Changes
Interactive video
•
6th - 10th Grade

11 questions
Explore Homeostasis and Feedback Mechanisms
Quiz
•
9th - 12th Grade

99 questions
PS Semester 2 Review
Quiz
•
9th - 12th Grade

49 questions
Florida Biology EOC Practice Test
Quiz
•
9th - 12th Grade