

STAAR Boot Camp - Day 8
Presentation
•
Science
•
8th Grade
•
Practice Problem
•
Easy
+8
Standards-aligned
Sarah Self
Used 2+ times
FREE Resource
11 Slides • 11 Questions
1
Properties of Matter Quizizz Lesson
STAAR Boot Camp Day 8
April 19, 2024
2

Everything is made of matter.
Matter can be measured, classified, and observed using its properties.
Properties of Matter
3
4
Categorize
Shiny
Conductors
Ductile
Malleable
Dull
Insulators
Brittle
Semiconductors
Can be shiny or dull
Can be malleable/ductile or brittle
Organize these properties into the correct classification.
5



Shiny (luster), conduct thermal energy and electricity very well (conductors), bend when hit with a hammer (malleable) and can be stretched into wires (ductile).
Metals are...
Not shiny (dull), cannot or are poor conductors of electricity and heat (insulators), shatter when hit with a hammer (brittle), and cannot be stretched into wires (brittle).
Nonmetals are...
Sometimes shiny or dull, can conduct thermal energy and electricity, but not very well (semiconductors), and can either be malleable/ductile or brittle.
Metalloids are...
6
Multiple Choice

Classify this element as either a metal, nonmetal, or metalloid.
7
Multiple Choice

Classify this element as either a metal, nonmetal, or metalloid.
Metalloid
8
Multiple Select
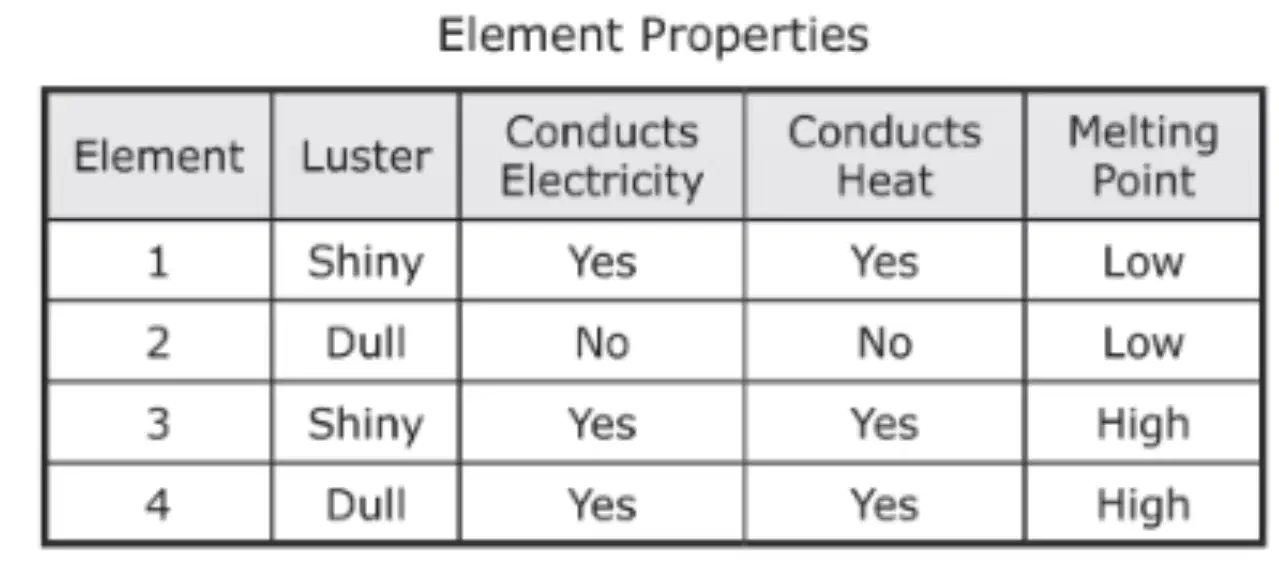
Based on the properties in the table, which TWO elements are most likely metals? (TWO Answers)
Element 1
Element 2
Element 3
Element 4
9
10
Dropdown
To solve for mass, the formula is...
To solve for volume, the formula is...
11

Density is the measurement of how much mass an object has per unit volume.
Mass is measured in grams (g).
Volume is measured in centimeters cubed (cm3) or milliliters (mL).
Density is measured in g/cm3 or g/mL
Density
12
Multiple Choice
Jack has a rock. The rock has a mass of 14 g and a volume of 2 mL. What is the density of the rock?
0.7 g/mL
1.2 g/mL
3.5 g/mL
11.4 g/mL
13
Multiple Choice
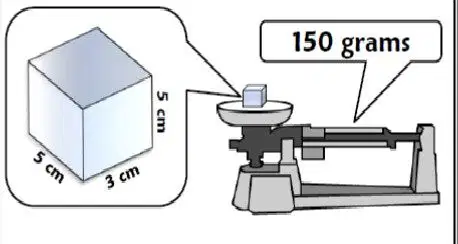
The image shows the an object on a triple beam balance. Its length, width, and height is provided. Based on these measurements, what would be the density of this object?
2 g/cm3
4 g/cm3
6 g/cm3
8 g/cm3
14


Physical changes happen when matter changes form but not chemical identity.
Physical Changes
Chemical changes happen when a chemical reaction forms new products.
Chemical Changes
15
Categorize

Iron rusting

Cooking an egg

Rotting Banana

Ice cube melting

Boiling water

Dissolving sugar in tea
Organize these changes in matter as either a chemical or physical change.
16
17
Match
Match the following images with what signs of a chemical change are presented.
Baking soda and vinegar react and fizz
A clear liquid changes to purple
A thick, solid substance is formed
A glow stick is cracked and glows
Warm steam is produced
Gas Production
Color Change
Precipitate
Light Production
Temperature Change
Gas Production
Color Change
Precipitate
Light Production
Temperature Change
18

Chemical reactions produce new substances.
Evidence a new substance is being produced are
(1) Color Change
(2) Gas
(3) Light
(4) Temperature Change
(5) Precipitate (solid)
(6) Odor
This can be remembered with City Girls Love Their Phones On!
Chemical Reactions
19
Multiple Select
Which TWO statements describe an action that most likely resulted in a chemical reaction? (TWO answers)
A pan of water was heated on a stove, and steam formed.
A liquid was mixed with another liquid, and a precipitate formed.
Leaves use carbon dioxide and water to make sugar.
A cup of saltwater evaporates, leaving behind salt crystals.
20
Multiple Choice
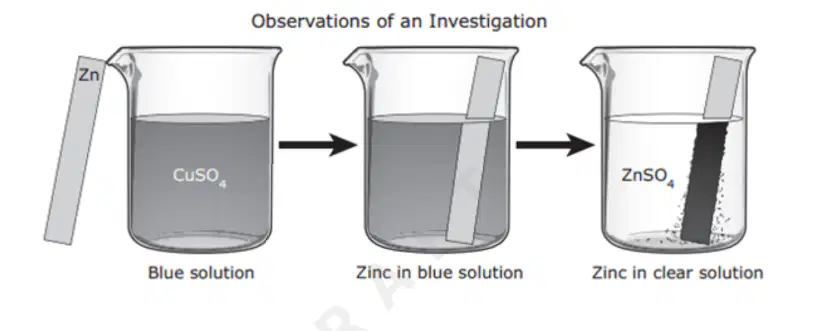
For an investigation a student poured a blue solution of CuSO4 into a beaker. The student placed a shiny, silver-colored strip of zinc metal in the solution and observed the changes.
The student inferred that a chemical reaction occurred. What evidence supports this inference?
A dark solid formed on the zinc metal.
The zinc metal remained silver-colored and shiny.
The CuSO4 solution turned blue when the zinc metal was added.
None of these
21
STAAR Question Practice Time!
Go to Canvas and complete Day 8 Questions.
22
Properties of Matter Quizizz Lesson
STAAR Boot Camp Day 8
April 19, 2024
Show answer
Auto Play
Slide 1 / 22
SLIDE
Similar Resources on Wayground

17 questions
Making an Electromagnet
Presentation
•
8th Grade

19 questions
Compounds
Presentation
•
8th Grade

21 questions
SSA Review
Presentation
•
8th Grade

15 questions
Rocks and Minerals
Presentation
•
7th Grade

19 questions
Human Effects on Water
Presentation
•
7th Grade

17 questions
Elements, Compounds, Molecules, oh my!
Presentation
•
8th Grade

16 questions
ENERGY TRANSFORMATION
Presentation
•
8th Grade

16 questions
8.E.2.2
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

45 questions
8th Grade Science - EOG Review
Quiz
•
8th Grade

49 questions
NC Released EOG 8 Science (2024)
Quiz
•
8th Grade

48 questions
8th Grade NC Science EOG Review 2
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

10 questions
The Ocean's Role in Weather and Climate
Interactive video
•
6th - 8th Grade

10 questions
Evidence of Evolution
Presentation
•
6th - 8th Grade