

11.1 Gas Laws
Presentation
•
Science
•
9th - 12th Grade
•
Hard
Standards-aligned
Stacy King
FREE Resource
15 Slides • 8 Questions
1
Gas Laws
School Announcements:
Boyle's Law
Charles’s Law
Avogadro’s Law
Ideal Gas Law


2

3

4
Multiple Choice
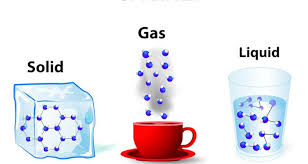
5

6
Pressure
Pressure: The amount of force applied per unit of area.
In gases, as pressure increases, volume decreases.
Why?
When the particles in a gas are compressed, they have less space to move around. As a result, the particles start to collide with one another more often — this increases pressure!

7

8
Pressure
⦿ Pressure- Force per unit area. Dependent on volume temperature. (Gas Laws)
1 atm
0.3 atm
0 atm
DOES AIR WEIGH ANYTHING?


9
Multiple Choice

When the area decreases what happens to the gas particles?
The pressure increases due to the applied force
The pressure decreases due to the applied force
The force and pressure are unrelated
10

Units of Pressure
Pascal - Pa (SI Unit)
Atmospheric – atm (we will use this unit)
Torr – torr
Millimeters of mercury – mmHg
Pounds per square inch - psi
11

12

13

14
Multiple Choice
15
Multiple Choice
16

Volume and pressure have an INVERSE RELATIONSHIP.
If pressure increases, volume decreases (gas is compressed).
If pressure decreases, volume increases (gas is released/expands)
17

BOYLE'S LAW Equation
18

BOYLE'S LAW Equation
P1 is initial Pressure
V1 is initial Volume
P2 is Final Pressure
V2 is Final Volume
19
Match
P1
V1
P2
V2
Initial pressure
Initial volume
Final pressure
Final Volume
Initial pressure
Initial volume
Final pressure
Final Volume
20
Multiple Choice

The formula for BOYLE'S LAW is?
P1V1=P2V2
T1V1=T2V2
pV=nRT
21
22
Multiple Choice
What happens to the balloon when the syringe plunger is pulled upwards?
The balloon remained unchanged
The balloon increased in size
The balloon decreased in size
23
Multiple Choice
According to the video what keeps the mass of the air molecules constant?
The size of the syringe
The temperature of the air
Covering the opening of the syringe
Water in the balloon
Gas Laws
School Announcements:
Boyle's Law
Charles’s Law
Avogadro’s Law
Ideal Gas Law


Show answer
Auto Play
Slide 1 / 23
SLIDE
Similar Resources on Wayground

21 questions
Atmosphere Refresher
Presentation
•
9th - 12th Grade

17 questions
Water uses, cycle, and conservation
Presentation
•
9th - 12th Grade

19 questions
3.4 Behavior of Gases
Presentation
•
9th - 12th Grade

18 questions
Intro to Nucleic Acids
Presentation
•
9th - 12th Grade

20 questions
Cell Division
Presentation
•
9th - 12th Grade

16 questions
Bioaccumulation vs. Biomagnification
Presentation
•
9th - 12th Grade

16 questions
The Theory of Evolution
Presentation
•
8th Grade

19 questions
Layers of the Earth
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade