

Last Chem Test Review!
Presentation
•
Chemistry
•
10th Grade
•
Medium
+2
Standards-aligned
Karina Farias
Used 6+ times
FREE Resource
22 Slides • 23 Questions
1
SE04 Multiple Choice Review!
2
Breakdown for today:
Review (this!) for the first 45 min.
Remember, this will be very similar to the exam, which has 16 questions!
Take the MC portion of the exam for 40 min.
Turn in materials/get ready to go.
3
Semester 1 Topics
4 Questions
4

When you write the atomic symbol, you include the mass number at the top left, the atomic number bottom left, and the symbol in the center.
Isotope Notation
5
You can use the APEMAN to create the symbol by finding the mass and atomic number, or deconstruct the symbol using APEMAN.
Isotope Notation
Atomic # = # Protons = # Electrons | Mass # = Atomic # + # Neutrons |
|---|---|
Since A is the bottom left, the atomic number is 6, as is the number of protons and electrons. | Since M = 14 and A = 6, we can find the neutrons using the equation: |

6

Elements that exist in pairs when they are alone are called diatomic elements. There are 7 elements that are diatomic:
H2, N2, O2, F2, Cl2, Br2, I2
Diatomic Elements
7
Name the metal (first element)
Name the nonmetal by changing to end in -ide, unless its an ion on the back of the periodic table.
Ionic Compounds
Naming Compounds
8
Fill in the Blanks

9
Fill in the Blanks

10
Multiple Choice

How many protons, neutrons, and electrons are in the symbol shown?
56 p, 81 n, 56 e
137 p, 56, 137 e
56 p, 137 n, 56 e
56 p, 193 n, 56 e
11
Quarter 3 Topics
3 Questions
12
Conversions
When there's no equation:
13
How many molecules (particles) are in 15 g O2?
Example
14
Conversions
When there's an equation:
15
Example
How many grams of Bromine are needed to create 150 grams of Sodium Bromide?
2Na + Br2 → 2 NaBr
16
Multiple Choice
48.86 g of cobalt is equal to how many moles of cobalt?
2879 moles
8.291 moles
.8291 moles
.829 moles
17
Multiple Choice
What is the mass of 4.98 x 1024 atoms (particles) of Zn?
541 g
8.27 g
.122 g
4.59 x 1046 g
18
Multiple Choice
2Al + 6HCl --> 2AlCl3 + 3H2
Aluminium reacts with hydrochloric acid. How many grams of aluminum are necessary to produce 11 L of hydrogen gas at STP?
13
8.8
0.99
1.0
19

Helps us determine if a reaction will occur for single replacement reactions.
The higher it is, it wants to be in a compound (more reactive it is).
Activity Series
20
Multiple Choice
21
Multiple Choice

Which metal is more reactive than calcium?
magnesium
potassium
silver
aluminum
22
Multiple Choice
If single element is above the element in compound, then the reaction ________________occur.
will
will not
23

We can use the relative amount as a percentage by making them into grams! Ex: 30% nitrogen and 70% oxygen would be 30 g N and 70 g O
Empirical Formulas
24
If a sample of a compound is 30% nitrogen by mass and 70% oxygen by mass, what would be the empirical formula?
Empirical Formulas
25
Multiple Choice
26
Quarter 4 Topics
9 Questions
27
Gas Laws
Gases follow a set of rules that allow for us to predict how they will exist or change given a set of conditions.
Variables to know:
P = Pressure; V = Volume; n = moles;
R = Gas Constant; T = Temperature
28




Pressure, volume, temperature, and the amount of particles are all related!
Ideal Gas Law
Volume and pressure are inversely related, so if one goes up, the other goes down.
Boyle's Law
Volume and temperature are directly related, so if one goes up, the other goes up.
Charles' Law
Temperature and pressure are directly related, so if one goes up, the other goes up.
Gay-Lussac's Law
29
EQUATIONS ARE ON THE BOARD!
30
Releases energy/heat to surroundings
Increases surrounding temps
-Q and -ΔH
Exothermic
Absorbs energy/heat from surroundings
Decreases surrounding temps
+Q and +ΔH
Endothermic
Thermochemistry
31
Heat flows from my cup to my water, making my cup colder and my water warmer. I should definitely put ice in it.
Ex 2
Heat flows from the oven to the pizza, which made my dinner. Don't judge me. It was from Walmart.
Ex 1
Heat flows from hot to cold (or less hot!)
32
Specific Heat Calculations!
Q = mcpΔT
Q = energy/heat (needs to + or -)
m = mass
cp = specific heat (use your chart!)
ΔT = change in temp (T2 - T1)
33
Multiple Choice
An ___________________ process is a process that releases energy.
exothermic
endothermic
34
Multiple Choice
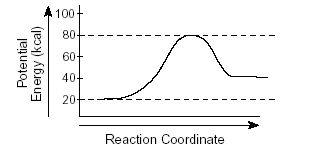
What is the ΔH of this reaction?
40 kJ
20 kJ
80 kJ
-60 kJ
35
Multiple Choice
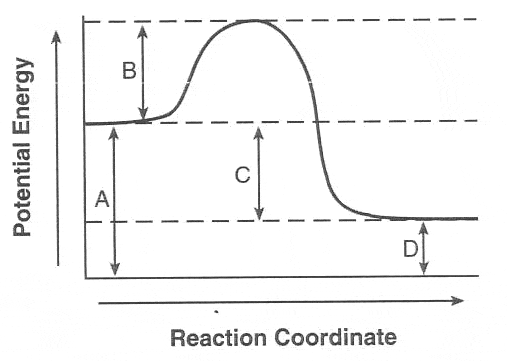
36
Multiple Choice
37
Multiple Choice
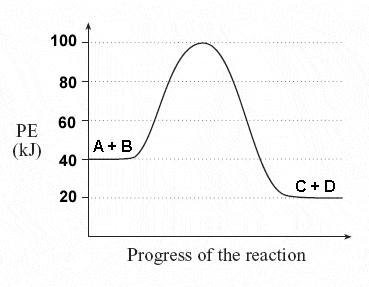
What is the change of the heat of the reaction (ΔH)?
-40 kJ
-20 kJ
100 kJ
60 kJ
38
Multiple Choice

39
Multiple Choice

What is the change of the heat of the reaction (ΔH)?
100 kJ
-175 kJ
-50 kJ
75 kJ
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
43
Multiple Choice
The specific heat of water is 4.18 J/g°C.
If 980. J of energy is added to 6.20 g of water at 18.0 °C, what is the final temperature of the water?
-55.8 °C
-19.8 °C
19.8 °C
55.8 °C
44
Multiple Choice

Lighting a match produces heat (energy) as the match stick burns. This is an example of an ____________ reaction.
endothermic
exothermic
45
Multiple Choice

Is the process shown to the right exothermic or endothermic?
endothermic
exothermic
SE04 Multiple Choice Review!
Show answer
Auto Play
Slide 1 / 45
SLIDE
Similar Resources on Wayground

39 questions
Naming Acids and Bases
Presentation
•
10th Grade

42 questions
Circle Vocabulary Part 2
Presentation
•
10th Grade

42 questions
Y4 Revision for EOY
Presentation
•
10th Grade

41 questions
Sentence Construction Errors
Presentation
•
10th Grade

39 questions
Only Americans use feets while world use meters.. SI
Presentation
•
10th Grade

42 questions
Density
Presentation
•
10th Grade

38 questions
Nuclear Chemistry Day 1
Presentation
•
10th Grade

42 questions
10/14/20 Periodic Trends
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade