

7.1.1-Substances & Mixtures
Presentation
•
Science
•
6th - 8th Grade
•
Medium
+16
Standards-aligned
Jessica Freeman
Used 6+ times
FREE Resource
39 Slides • 54 Questions
1

2

3

4
Atom - The smallest unit of an element. (Non-Living)
Made of 3 parts
Protons (+) and Neutrons (-).`
Found in the center (Nucleus)
Electrons
Found outside around the Nucleus.
Element - Anything that can not be broken down into a smaller unit.
Made of all the same atoms
If something is made of all the same atoms and no able to be broken down into anything smaller - Pure substance.
Making matter

5
Multiple Select
What are atoms made of
Protons
Nuetrons
Electrons
Robotics
6
Multiple Choice
What is an element?
An element is a substance that cannot be broken down into simpler substances .
7
Multiple Choice

Is gold a pure element?
YES
NO
8
3 Make up 93% of your body mass (weight).
There are 6 Elements that make up 99% of the bodies mass (weight).
Elements

9
Multiple Choice

What three elements make up living organisms?
10
3 Make up 93% of your body mass (weight).
There are 6 Elements that make up 99% of the bodies mass (weight).
Elements

11
Anything that is made of 2 at least or more atoms of the same element or of atoms of different element by some kind of bond.
Chemical Bonds
act like a glue to hold atom of different elements in place
Compounds
When many of the same type of Molecules are present.
Molecule



12
Multiple Choice

Why is this a molecule?
it has more then 2 atoms
not a pure substance
It has more than one atom.
it is not a molecule.
13
Match
Match the molecules
H2O
O2
CO2
NaCl
C₆H₁₂O₆
H2O
O2
CO2
NaCl
C₆H₁₂O₆
14
Fill in the Blanks

15
Labelling
What Molecules make up 93% of the body?
oxygen 65%
Carbon 18.5%
Hydrogen 9.5%
16

17

18
Multiple Choice
Which substance is a compound?
Neon (Ne)
Oxygen (O2)
Carbon Dioxide (CO2)
Nitrogen (N2)
19
Multiple Choice
Is Sodium (Na) an element or a compound?
Element
Compound
20
Multiple Choice
Is Sodium Chloride (NaCl) an element or compound?
Element
Compound
21
Multiple Choice
Is Iron (Fe) an atom or a molecule?
Atom
Molecule
22
Multiple Choice
Is Chlorine (Cl2) an atom or a molecule?
Atom
Molecule
23

24
Multiple Choice
True or False: The tiniest bit of water that still is water is a molecule of water.
True
False
25
Multiple Choice
True or False: Hydrogen is a compound.
True
False
26
Multiple Choice
What is a compound?
Two different elements bonded together.
Two of the same elements bonded together.
A mixture of different substances.
The substances stuck together with glue.
27
Multiple Choice
Two or more atoms join up to make a(n)
element
compound
molecule
28
Multiple Choice
Two or more different elements bond to form a(n)
element
compound
molecule
29

30
Multiple Choice

Which is it?
Mixture of Elements and Compounds
Compound
Molecule
Mixture of Elements
Mixture of Compounds
31
Multiple Choice

Which is it?
Mixture of Elements and Compounds
Compound
Molecule
Mixture of Elements
Mixture of Compounds
32
Multiple Choice

Which is it?
Mixture of Elements and Compounds
Compound
Molecule
Mixture of Elements
Mixture of Compounds
33

34
Pure Substances & Mixtures
Some text here about the topic of discussion

35
Multiple Choice
Both elements and compounds are pure substances.
AGREE
DISAGREE
36
Multiple Choice
Which of these is a compound?
Aluminium (Al)
Carbon Dioxide (CO2)
Nitrogen (N)
Sand
37

Homogeneous Mixture is uniform and evenly mixed!
38
Heterogeneous Mixture is NOT uniform and NOT evenly mixed!

39
Multiple Choice

Elements and Subatomic Particles
Protons and Electrons
Atoms and Compounds
Homogenous and Heterogeneous
40
Multiple Choice
In which mixture can you not see all of the parts?
homogeneous
heterogeneous
41
Open Ended
Type 3 examples of a homogeneous mixture
42
Multiple Choice
In which mixture can you see all of the parts?
homogeneous
heterogeneous
43
Open Ended
Type 3 examples of a heterogeneous mixture
44

45

Some mixtures might seem to change properties because the particles are evenly spread out, but the particles don't change.
The mixture might seem like a new substance but the particles can still be separated.
Solutions
46

47

48
Multiple Choice
What is a combination of two types of matter that can easily be taken apart?
Mixture
Solution
Dissolve
Oso
49
Fill in the Blanks
Type answer...
50
Multiple Choice
What word means to have something "disappear" inside of something else?
dissolve
magic
leave
go away
51

52

53
Multiple Choice

Mixture or solution?
Mixture
Solution
54
Multiple Choice

Mixture or solution?
Mixture
Solution
55
Multiple Choice

Mixture or solution?
Mixture
Solution
56
Multiple Choice

Mixture or solution?
Mixture
Solution
57
Multiple Choice

Mixture or solution?
Mixture
Solution
58
Multiple Choice

Mixture or solution?
Mixture
Solution
59
Day 1 Recap

Let's take a poll on the next slide.
60
Poll
What do you think so far? Do you think you understand...
PURE SUBSTANCES: Atoms/Elements, Molecules/Compounds VS. Mixtures: Heterogeneous/Homogenous and Solutions
Smarty Pants
Awesome!
Let me help!
I've been there.
61
Separation of Mixtures
How do we separate Homogeneous and Heterogeneous mixtures??

62
Multiple Choice
Are made from a combination of 2 or more elements...
Molecules
Pure Substances
Compounds
Elements
63
Multiple Choice
A combination that can be separated by physical processes is a .....
Pure Substance
Glucose
Element
Mixture
64
Multiple Choice
A mixture that is NOT evenly distributed is called ....
Compounded
Homogenous
Heterogenous
Salty
65
Multiple Choice
A substance with atoms that are all alike.
element
compound
homogeneous mixture
heterogeneous mixture
66

67

68

69
Chromatography
Chromatography SEPARATES substances as they are drawn across a barrier.
Chromatography happens when different colors from an ink pen spread out on a paper towel.

70
Distillation
Separates mixtures based on differences in substances' boiling points.
Distillation is used to process crude oil into its many different parts such as gasoline.

71

72

73
Multiple Choice
If a solid doesn't dissolve in a liquid we say that it is
Soluble
A solvent
Insoluble
Undissolveable
74
Multiple Choice
What happens when salty water is boiled?
The salt becomes a new compound and water evaporates as it has a low boiling point
The water evaporates and the salt remains in the beaker as it has a high melting point
The water becomes a new compound and evaporates while salt remains in the beaker
The water will disappear and the salt remains in the beaker as it is a solid
75
Multiple Choice
The process used to separate heterogeneous mixtures of solids and liquids is called
filtration
crystallization
distillation
chromatography
76
Multiple Choice

If you spilled nails in a sandbox, what separation tool/method could you use to clean out the sandbox so it would be safe for children to play in?
Filter
Screen
Magnet
Evaporation
77
Multiple Choice

This type of paper allows liquid to pass through it. What tool is it?
sieve
filter
78
Multiple Choice
Mixtures can be separated .......................
Physically
Chemically
79

80

81

• Two or more types of atom chemically
combined
• Two or more capital letters
• One substance with different elements
HBr
NH3
H2O NaHCO3
Examples:
Visual Representation
How to Distinguish
82

83
Multiple Choice

Is this a mixture?
Yes
No
84
Exit Slip- Match It!

85
Multiple Choice

Study the diagram. Which of the following is represented in the diagram?
monoatomic element
elemental molecules
compound molecules
86
Match
Match the following models to their correct descriptions.
mixture of elements
mixture of elements and compounds
mixture of compounds
elements
compounds
mixture of elements
mixture of elements and compounds
mixture of compounds
elements
compounds
87
Multiple Choice
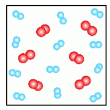
88
Multiple Choice

What type of substance is pictured?
element
compound
mixture
solution
89
Poll
What do you think so far? Do you think you understand...
PURE SUBSTANCES: Atoms/Elements, Molecules/Compounds VS. Mixtures: Heterogeneous/Homogenous and Solutions
Smarty Pants
Awesome!
Let me help!
I've been there.
90

91
Mixtures
Made up of many different kinds of atoms and molecules.
Form when two or more different substances combine physically but not chemically.
You can separate a mixture relatively easily

92

93
Day 2 Recap



Show answer
Auto Play
Slide 1 / 93
SLIDE
Similar Resources on Wayground

89 questions
B1 Language Exam Guide B1P1
Presentation
•
6th - 8th Grade

85 questions
Force as a vector
Presentation
•
7th Grade

83 questions
RFMS Science: Fall Benchmark Review/Preview 24
Presentation
•
6th - 8th Grade

90 questions
Basic English
Presentation
•
6th - 8th Grade

85 questions
Communism in China
Presentation
•
7th Grade

87 questions
P6A_Reported Speech
Presentation
•
6th - 7th Grade

90 questions
Great Depression
Presentation
•
8th Grade
![G7- Final Review Chapter 3 [Term1]](https://cf.quizizz.com/image/image-loader.svg)
92 questions
G7- Final Review Chapter 3 [Term1]
Presentation
•
7th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

12 questions
PS.8.1.2, PS.8.1.3, PS.8.1.5 Review
Quiz
•
8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade