
![G7- Final Review Chapter 3 [Term1]](https://quizizz.com/media/resource/gs/quizizz-media/quizzes/056d6a80-2aeb-491d-823b-a10c503f92dd?w=200&h=200)
G7- Final Review Chapter 3 [Term1]
Presentation
•
Science
•
7th Grade
•
Hard
Mai Hamza
Used 8+ times
FREE Resource
4 Slides • 88 Questions
1

2

3

4
Multiple Choice
Which scientist discovered the electrons?
Dalton
Thomson
Atristotle
Bohr
5
Multiple Choice
Bohr discovered that electrons are located in
the nucleus
inside protons
electron cloud
circular orbits around the nucleus
6
Multiple Choice
The smallest particle of an element that still represents that element.
Nucleus
electron
proton
atom
7
Multiple Choice
What is the center of an atom called?
nucleus
electron cloud
proton center
photon center
8
Multiple Choice
9
Multiple Choice
Who was the Greek philosopher who called the smallest particle of matter an "atom"?
Democritus
Aristotle
J.J Thompson
Einstein
10
Multiple Choice
Which of the following is/are conclusions based on Rutherford’s gold foil experiment?
Atom is mostly empty space
The nucleus is positively charged
The atom has a small dense nucleus
All answers are correct
11
Multiple Choice
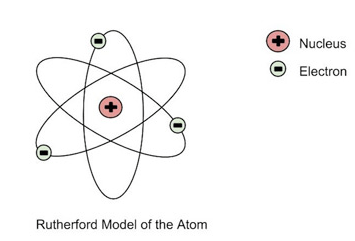
12
Multiple Choice

13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
Which one of the following is the correct order of scientific contribution of the atom?
Chadwick, Dalton, Bohr, Rutherford, Democritus,Thomson
Thomson, Democritus,Rutherford, Bohr, Dalton ,Chadwick
Dalton, Chadwick ,Bohr, Rutherford, Democritus,Thomson
Democritus, Dalton, Thomson, Rutherford, Bohr, Chadwick
16
Multiple Choice
Who was the first person to come up with the Atomic Theory which included four parts.
John Dalton
Democritus
Ernest Rutherford
Niels Bohr
17
Multiple Choice
18
Multiple Choice
Used the word "atomos" to decsribe the uncuttable (indivisible) atom.
Democritus
Thomson
Bohr
Dalton
19
Multiple Choice

Proposed that electrons move around the nucleus in specific layers, or shells.
Bohr
Rutherford
Chadwick
Thomson
20
Multiple Choice

This picture best represents ___________________ atomic model.
Thomson's
Dalton's
Bohr's
Chadwick's
21
Multiple Choice
Who proposed the 'plum pudding' model of the atom?
o Dalton
o Thomson
o Rutherford
o Bohr
22
Multiple Choice
Which of the four Physicists was first to work with cathode rays?
o Dalton
o Thomson
o Rutherford
o Bohr
23
Multiple Choice
Which model of the atom was created directly after the cathode ray tube experiment?
plum pudding
quantum mechanical model
atomic model
bohr model
24
Multiple Choice
Which experiment discovered the nucleus of the atom?
cathode ray tube
gold foil experiment
heisenberg's uncertainty principle
flame test experiment
25
Multiple Choice

Whose model suggested that negative particles were mixed in with positively charged material - like seeds in a watermelon?
J.J. Thomson
Ernest Rutherford
Niels Bohr
Albert Einstein
26
Multiple Choice
Place the following atomic models in order, from earliest to latest:
A) Rutherford B) Thomson C) Dalton
B, C, A
C, A, B
A, C, B
C, B, A
27
Multiple Choice
28
Multiple Choice

In the atomic model nicknamed the "plum pudding" model, what do the plums represent?
the nucleus
the atom
the electrons
the positive material
29
Multiple Choice
All matter is made of what
energy
atoms
electrons
compounds
30
Multiple Choice
31
Multiple Choice
32
Multiple Choice
A particle that circles around the nucleus is a(n)...
Proton
Neutron
Electron
33
Multiple Choice
34
Multiple Choice
The central region (middle part) of an atom where its neutrons and protons is its
nucleus
electron cloud
core
center
35
Multiple Choice
36
Multiple Choice
37
Multiple Choice
38
Multiple Choice
39
Multiple Choice

What is the atomic number for an element with three protons?
2
1
3
6
40
Multiple Choice

41
Multiple Choice

How many neutrons are in the atom "K"?
7
2
39
20
42
Multiple Choice
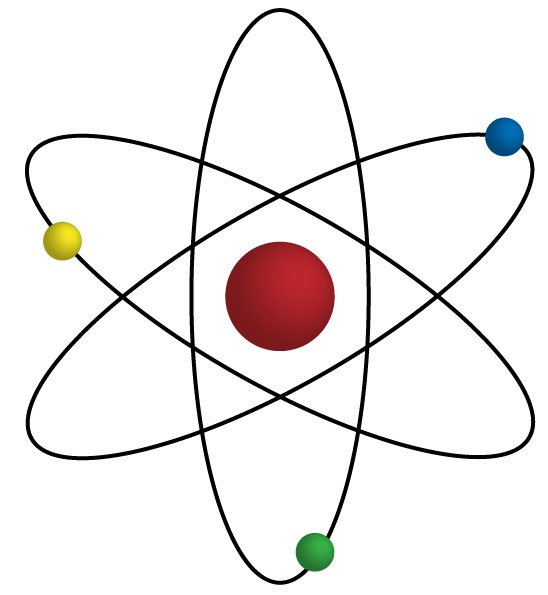
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
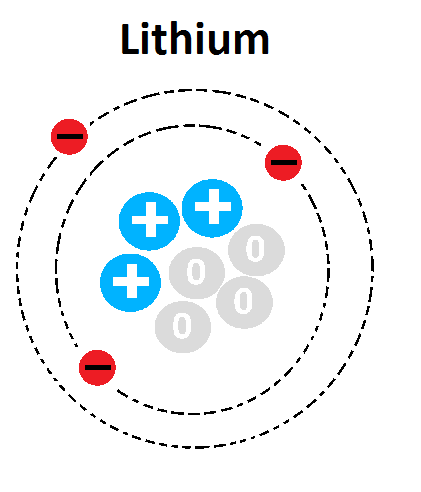
46
Multiple Choice
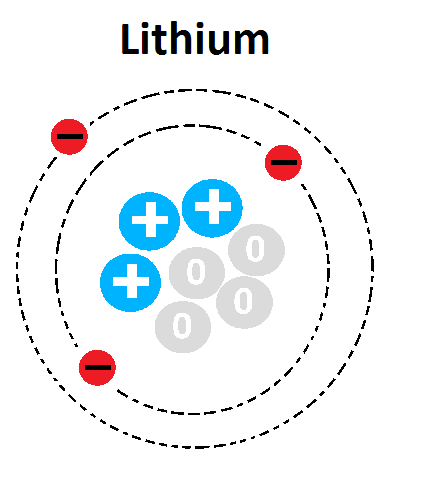
What is the mass number of this atom?
1
3
4
7
47
Multiple Choice

The fluorine atom has an atomic number of
19
9
48
Multiple Choice

What is the mass number and atomic number ?
Mass number = 7, atomic number = 14
Mass number = 14, atomic number = 7
49
Multiple Choice
50
Multiple Choice
51
Multiple Choice

52
Multiple Choice
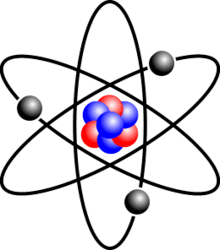
Which subatomic particle has a negative charge in the atom?
proton
neutron
electron
quark
53
Multiple Choice

How many neutrons are there in one atom of magnesium?
12
6
24
54
Multiple Choice
Which element has a mass number of 12 and contains 6 neutrons?
Gallium
Copper
Carbon
Sodium
55
Multiple Choice
ALL atoms of the same element have:
same number of proton
same number of nucleon
different number of neutron
different number of electron
56
Multiple Choice
57
Multiple Choice
What is the average mass of this element?
58
Multiple Choice

59
Multiple Choice
Calculate the average atomic mass of the element iron (Fe) using the following data:
[Isotope / % abundance]
[Iron 54 / 6% ] [Iron 56 / 92% ] [ Iron 57 / 2% ]
53.7 amu
54.9 amu
5592.0 amu
55.9 amu
60
Multiple Choice
61
Multiple Choice
62
Multiple Choice

63
Multiple Choice

64
Multiple Choice

65

66
Multiple Choice
What is a positive ion called?
anion
cation
isotope
covalent
67
Multiple Choice
What is a negative ion called?
anion
cation
covalent
isotope
68
Multiple Choice
Which types of elements from cations?
metals
nonmetals
metalloids
noble gases
69
Multiple Choice
Which types of elements from anions?
metals
nonmetals
metalloids
noble gases
70
Multiple Choice
If an atom loses electrons, the charge will be positive.
true
false
71
Multiple Choice
A Bromine ion gains 1 electron, which of the following is the correct symbol for a Bromine ion?
Br-1
Br+1
Br+7
Br-7
72
Multiple Choice

73
Multiple Choice

74
Multiple Choice

75
Multiple Choice

76
Multiple Choice

77
Multiple Choice

P+5
78
Multiple Choice
atoms lose or gain electrons
atoms lose electrons
atoms gain electrons
79
Multiple Choice

80
Multiple Choice
81
Multiple Choice
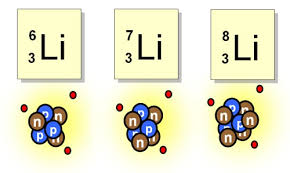
82
Multiple Choice
83
Multiple Choice
84
Multiple Choice
85
Multiple Choice
86
Multiple Choice
Isotopes of an element have a different number of...
Protons
Neutrons
Electrons
Mass
87
Multiple Choice
88
Multiple Choice
89
Multiple Choice
90
Multiple Choice
91
Poll
How do you Evaluate yourself....Are you ready for the Final Exam
92
Open Ended

What are the points you still need help to understand
ما النقاط التي لازلتي تحتاجين لمساعدة حتى تتمكنين من فهمها

Show answer
Auto Play
Slide 1 / 92
SLIDE
Similar Resources on Wayground

95 questions
Chapter 2 Section 9 Lesson
Presentation
•
7th Grade

78 questions
AKIDAH ISLAM
Presentation
•
7th Grade

78 questions
UNIT 2 REVIEW 10-18-21
Presentation
•
7th Grade

79 questions
GA Milestones Physical Science
Presentation
•
8th Grade

79 questions
Ecology
Presentation
•
9th Grade

87 questions
Form 1 Chapter 6: Periodic Table
Presentation
•
7th Grade

86 questions
PhySci Final Review 7: Compounds
Presentation
•
9th Grade

96 questions
Matter, Elements, Compunds and Mixtures
Presentation
•
7th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
genetics, punnett squares, heredity
Quiz
•
7th Grade

20 questions
Food Webs + Energy Pyramids
Quiz
•
7th Grade

10 questions
Exploring Weather Influences and Map Reading
Interactive video
•
6th - 10th Grade

43 questions
Amplify Earth's Changing Climate Unit Review
Quiz
•
6th - 8th Grade

62 questions
Super Science Trivia
Quiz
•
6th - 8th Grade