

Finals M4 Chemistry covalent,ionic,metallic
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Hard
Lady Alias
Used 1+ times
FREE Resource
42 Slides • 42 Questions
1

2

3

4

5
Covalent Bonds
Covalent bonding occurs in elements and compounds containing non-metallic elements only and is formed when pairs of electrons are shared.
A shared pair of electrons is known as a single bond.
All which form a covalent bond join together to form uncharged molecules. All the atoms involved achieve a full outer shell of electrons.
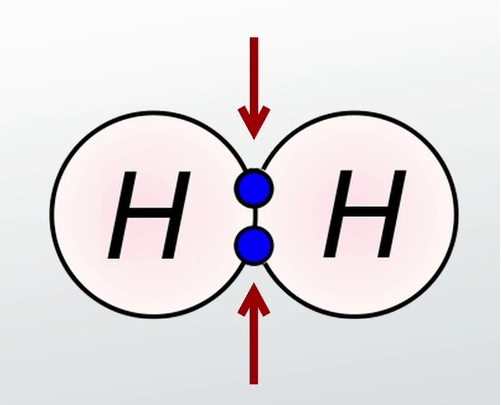
for Hydrogen, it has only 1 electron which make it share the only electron. The total electrons after bonded for hydrogen is only 2 electrons (duplet).
the rest atoms must be octet (8 electrons), but for period 3 and above, they can make more than 8 electrons.
Some text here about the topic of discussion
6
1 electron shared by each hydrogen
Some text here about the topic of discussion.
Hydrogen (H2)

7
Cl2 = 1 electron shared by each chlorine atom
H2O = 1 electron shared by each hydrogen, to form two single covalent bonds
Some text here about the topic of discussion.
Cl2 and H2O

8
THREE electrons are shared by two atoms
e.g. N2
Triple bonds
TWO electrons are shared by two atoms
e.g. CO2
Double bonds
Double Bonds and Triple bonds


9
Covalent Bond
Electron Dot or Lewis Dot
signifies the bonding pair of electrons

10
Polyatomic ions
a bonded set of 2 or more atoms that function as a single unit

11
Octet Exceptions
Free Radicals have odd number of electrons

12
Octet exceptions
electron deficient the central atom does not have a filled valence shell

13
Multiple Choice
14
Multiple Choice

15
Multiple Choice
16
Multiple Choice
17
Multiple Choice

18
Multiple Choice

19
Multiple Choice
20
Multiple Choice
21
Multiple Choice
Valence electrons are found
in the innermost energy level of an atom
in the middle energy levels of an atom
in the outermost energy levels of an atom
22
Multiple Choice
23
24
Ionic bonds
THe CATion transfers valence electrons to the Anion. A a Crystalline lattice holds it together.
25

26
Ionic bonds
How can you tell if it is an Ionic bond?
You look for the Metal!!! A Metal and a non metal is required to make an Ionic bond.
This is also called a SALT.
27
Ionic bonds are strong
Salts have very high melting points. This means that you need a LOT of heat to melt salt. Think of your French fries. The salt is a crystal not a puddle.

28
Multiple Choice
What temperature do Ionics melt?
Very low temperatures
They don't ever melt
Very high temperatures
They always melt
29
Ionic bonds
Are good conductors
But ONLY WHEN dissolved in water!!!
(aq) aqueous mixture
30
When a salt dissolves in water (aq)
IONS are FREE to MOVE!!

31
THE MORE FREE MOVING IONS
the Brighter the light!!!!!!!!!
This is called an ELECTROLYTE

32
Multiple Choice
When do Ions conduct electricity?
When they are in a crystal structure
When they are free moving in solution
When they are solids
When they are bonded together.
33

34

35

36

37

38

39

40

41

42
Multiple Choice
Which pair of atoms will form an ionic bond?
Li and Ne
K and Br
K and Cs
S and Cl
43
Multiple Choice
Which pair of atoms will form an ionic bond?
Li and Be
Na and Mg
K and Ca
Na and Cl
44
Multiple Choice
An ionic bond would form between Sc and F
True
False
45
Multiple Choice
Which of the following compounds would you expect to be ionic?
H2O
CO2
SrCl2
SO2
46
Multiple Choice
Which of the following compounds would you expect to be ionic?
CO
NO
LiCl
SiO2
47
Octet Rule
elements tend to bond in such a way that each atom has eight electrons in its valence shell
48
Ionic Bonds
metal and non metal

49
Ionic Bonds
complete transfer of electron

50
Ionic Bond
Forms cation and anion that are held together due to electrostatic attraction forces

51
Multiple Choice
Chemical bond is ...
the physical mixing of two different atoms.
the force that holds two atoms together
the energy used up when two atoms combined.
the temperature that changes the phase of matter.
52
Multiple Choice
Predict the bond that will form between Sr and S.
Ionic
Covalent
53
Multiple Choice
54
Multiple Choice
The octet rules states that most elements want to have _____ valence electrons.
2
4
6
8
55
Multiple Choice
If an atom gains two electrons what charge will it have?
-2
-1
+1
+2
56
Multiple Choice
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
60
Multiple Choice
61
Multiple Choice
62
Multiple Choice
Why do elements bond?
To be friends
To create a new element
To become stable
To get bigger
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
Metals are hard because metals have a
sea of electrons that tightly hold the nuclei together
they form triple bonds
low melting point
high luster
66
Multiple Choice
Why are metals malleable?
The metal ions can easily slide past one another
The metal ions cannot easily slide past one another
The metal ions repel one another
The metal ions attract one another
67
Multiple Choice
What does malleable mean?
Breaks easily
Always stays in the same shape
Can be drawn into wires
Can be forced into a certain shape without breaking
68
Multiple Choice
Metals conduct electricity because
the metal ions are free to travel
their electrons are free to travel
the bonds between metal atoms can easily be broken
of witchcraft
69
Multiple Choice
Which of the following would be held together by the metallic bond?
Atoms of iron (Fe)
Molecules of CH4
Atoms of sulfur (S)
Units of NaCl
70
Electrons are the Key
If valence electrons are shared by atoms, it is called COVALENT bonding
If valence electrons move freely between atoms, it is called METALLIC bonding
If valence electrons are transferred from one atom to another, it is called IONIC Bonding

71
ELECTRONEGATIVITY
DEN Range is the difference in electronegativity values between atoms
Determines the type of bond
Covalent bonds have similar electronegativities
Ionic bonds have different electronegativities

72
What you need to remember...
Metals + Nonmetals
Make IONIC bonds
Transfer electrons
High electronegativity difference

73
Ionic Bonds
Form Crystalline Lattice (crystals)
Have high melting and boiling points
Hard
Brittle
Conduct electricity when dissolved in water

74
What you need to remember
Nonmetal + Nonmetal
Make COVALENT bonds
Share electrons
Low electronegativity difference

75
Covalent Compounds
Have Lower melting and boiling points
Tend to be flammable
When dissolved in water they do not conduct electricity
Soft or brittle solid
Can also form crystals

76
What you need to remember
Metal ions near each other share valence electrons
These valence electrons flow freely between all of the ions
These delocalized electrons are why metals conduct heat and electricity
It is also why metals are malleable and ductile

77
Metallic Bonds
good conductors of heat and electricity
malleable and ductile
lustrous
opaque

78
Multiple Choice
Identify the following compound as ionic or covalent: Na2SO4
ionic
covalent
79
Multiple Choice
What type of bond involves the sharing of electrons between atoms?
covalent bond
ionic bond
metallic bond
transfer bond
80
Multiple Choice
Identify the following compound as ionic or covalent: CF4
ionic
covalent
81
Multiple Choice
A bond between a nonmetal and a nonmetal is called a(n)
covalent bond
ionic bond
metallic bond
transfer bond
82
Multiple Choice
Identify the following compound as ionic or covalent: MgO
ionic
covalent
83
Polyatomic Ions
Certain atoms bond covalently to form a group of atoms that have both molecular and ionic characteristics
Polyatomic Ions: A charged group of covalently bonded atoms is known as a polyatomic ion

84
Multiple Choice
What is A charged group of covalently bonded atoms?
A polyatomic ion
A cation
An anion
a crystal ion

Show answer
Auto Play
Slide 1 / 84
SLIDE
Similar Resources on Wayground

74 questions
Identifying DNA as the genetic material
Presentation
•
KG

78 questions
midterm review: Combined
Presentation
•
9th Grade

73 questions
PhySci Final Review 5: Matter
Presentation
•
9th Grade

82 questions
Introduction to Covalent Bonding
Presentation
•
9th - 12th Grade

84 questions
Writing Chemical Equations from Word equations
Presentation
•
9th - 12th Grade

79 questions
Unit 2 review
Presentation
•
9th - 10th Grade

83 questions
Chemistry Unit 2 Summative Review - Atomic Structure & Molar Qua
Presentation
•
10th Grade

78 questions
Chemistry U3: Valence Electrons & Coulombic Attraction Notes
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade