

Isotope Lesson
Presentation
•
Chemistry
•
10th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
12 Slides • 21 Questions
1
Isotopes Lesson
2
Objectives
Read through the slides to be able to answer the following questions.
By the end of the lesson, students will be able to explain how the atomic structure of isotopes are similar and different from one another.
3


ISOTOPES
Atoms with the same number of protons and a
different number of neutrons
4
Multiple Choice

What is the main difference between the Hydrogen atoms shown in the picture?
The number of protons
The number of neutrons
The electron shell
There is no difference between the atoms of Hydrogen
5
Fill in the Blanks
6
Fill in the Blanks
Type answer...
7

8
Multiple Choice
An isotope is an atom of the same element that has a different number of _____.
protons
neutrons
electrons
9

10

11
Multiple Choice
The biggest difference between isotopes of the same element are the masses.
False
True
12
Multiple Choice
An isotope of an element will react the same as a different isotope of the same element.
true
false
13

14
Multiple Choice

Carbon has an atomic number of 6. Which isotope has 8 neutrons?
Carbon-12 or C-12
Carbon-13 or C-13
Carbon-14 or C-14
15
Multiple Choice

Lithium has 3 isotopes, which isotope has 4 neutrons?
Lithium-6 or Li-6
Lithium-7 or Li-7
Lithium-8 or Li-8
16


ISOTOPE NOTATION:
17
Multiple Choice
What does the 14 represent in the isotope notation?
atomic number
protons
electrons
Mass Number
18


19
Labelling
Label the Isotope Notation correctly
mass number
atomic number
symbol
20
Fill in the Blanks

Type answer...
21
Fill in the Blanks

Type answer...
22
Fill in the Blanks

Type answer...
23
Multiple Choice

How many protons are in a Chlorine atom? (Mass number = 35)
17
35
35.45
18
24
Multiple Choice

How many neutrons are in a Phosphorus atom? (Mass Number = 31)
15
30.9738
16
31
25

26
Reading the Symbol
Note: This is not reflective of the Periodic table Organization.
Mass Number provided at the top and atomic number sometimes at the bottom.
The only change should be the top number.

27
Anything else?
Sometimes it is given in Hyphen notation.
Element symbol or name hyphen (dash) Mass Number.

28
Multiple Choice
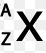
In a correctly written symbol what would be located in the "A" position?
number of neutrons
atomic number
number of electrons
mass number
29
Multiple Choice
What quantities vary between isotopes of an element?
protons, electrons, and atomic mass
protons, electrons, and atomic number
neutrons and electrons
neutrons and atomic mass
30
Multiple Choice

What do these isotopes of carbon all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and electrons
protons, atomic number, and mass number
31
Multiple Choice

How many protons does this isotope of titanium have?
48
22
26
70
32
Multiple Choice
Isotopes are atoms of the same element with different #’s of __________ & therefore different __________.
n0 ; atomic #’s
p+ ; atomic #’s
e- ; atomic masses
n0 ; atomic masses
33
Multiple Choice

If X is the symbol for an element, which of the following two symbols represent isotopes of the same element?
I and II
III and IV
I and IV
I and III
Isotopes Lesson
Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

27 questions
Molecular Geometry
Presentation
•
11th - 12th Grade

25 questions
2.9 Covalent Bonding
Presentation
•
9th - 12th Grade

28 questions
Atomic Structure Notes
Presentation
•
9th - 12th Grade

28 questions
Stoichiometry
Presentation
•
9th - 12th Grade

24 questions
Atomic Models
Presentation
•
9th - 12th Grade

26 questions
Periodic Table Day 4-Trends in Electronegativity & Ion Size
Presentation
•
10th - 12th Grade

21 questions
Dilutions and Molarity Calculations
Presentation
•
10th - 12th Grade

21 questions
Limiting Reagents and Percentage Yield
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Unit 7 Chemical Reactions
Quiz
•
10th Grade

25 questions
Unit 8 Stoichiometry Review
Quiz
•
10th Grade

22 questions
Unit 9 Gas Law Quiz
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

15 questions
Nuclear Chemistry: Balancing
Quiz
•
10th Grade

20 questions
Naming Acids and Bases
Quiz
•
10th Grade

10 questions
Exploring Stoichiometry in Chemistry
Interactive video
•
6th - 10th Grade