

Covalent Bond Lesson
Presentation
•
Science
•
10th Grade
•
Hard
Joseph Anderson
FREE Resource
29 Slides • 21 Questions
1

The Covalent Bond
Lesson 1
2


Focus Question
How do atoms bond in covalent
molecules?
3


New Vocabulary
covalent bond
molecule
Lewis structure
sigma bond
pi bond
endothermic
exothermic
4


Review Vocabulary
chemical bond: the force that holds two atoms
together
5


Why do atoms bond?
Gaining Stability
•
The stability of an atom, ion, or compound is
related to its energy: lower energy states are
more stable.
•
Ionic bonds, metals, and nonmetals gain stability
by transferring (gaining or losing) electrons to
form ions with stable noble-gas electron
configurations.
•
Valence electron sharing allows atoms to acquire
the stable electron configuration of noble gases.
6
Multiple Choice
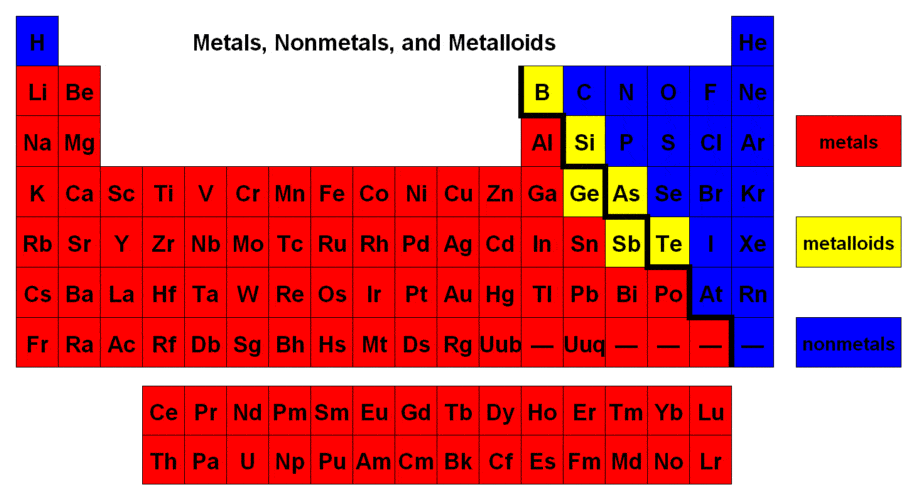
metal and nonmetal
2 or more nonmetals
metal
none of the above
7
Multiple Choice
The outermost electrons of an atom are __________.
outermost electron
Lewis electrons
valence electrons
octet electrons
8
Multiple Choice
How many valence electrons should an element have for it to be considered as a STABLE element?
2
10
6
8
9


What is a covalent bond?
Shared Electrons
• Atoms in non-ionic compounds share
electrons.
• A covalent bond is a chemical bond that
results from electron sharing.
• A molecule is formed when two or more
atoms bond covalently.
• The majority of covalent bonds form between
atoms of nonmetallic elements.
10



What is a covalent bond?
Covalent Bond Formation
• Diatomic molecules (H2, N2, F2, O2, I2, Cl2, Br2)
exist because the two-atom molecules are
more stable than the individual atoms.
11



What is a covalent bond?
• The most stable arrangement of atoms exists
at the point of maximum net attraction,
where the atoms bond covalently and form a
molecule.
12
Multiple Choice
Name the type of bonding in which the electrons are shared.
Ionic bonding
Covalent bonding
Metallic bonding
13
Multiple Choice
What are diatomic elements?
Elements that form two-atom molecules when not combined in a compound.
Two elements that combine with one atom from each element.
Elements that are always in pairs no matter what.
14
Multiple Choice
Is Fe2 a diatomic element?
Yes
No
15
Multiple Choice
Is O2 a diatomic element?
Yes
No
16
Multiple Choice
The covalent bond forms between
a metal and nonmetal
a nonmetal and nonmetal
a metal and metal
17



Single Covalent Bonds
•
When only one pair of electrons is shared, the
result is a single covalent bond.
•
The figure shows two hydrogen atoms forming a
hydrogen molecule with a single covalent bond,
resulting in an electron configuration like helium.
•
In a Lewis structure,dots or a line are used to
symbolize a single covalent bond.
18


TYPES OF COVALENT BONDS
SINGLE Covalent Bonds
DOUBLE Covalent Bonds
TRIPLE Covalent Bonds
19


Single Covalent Bonds
Group 17 and Single Bonds
•
The halogens—the group 17 elements—have 7
valence electrons and form single covalent bonds
with atoms of other non-metals.
Group 16 and Single Bonds
•
Atoms in group 16 can share two electrons and
form two covalent bonds.
•
Water is formed from one oxygen with two
hydrogen atoms covalently bonded to it.


20


Single Covalent Bonds
Group 15 and Single Bonds
•
Atoms in group 15 form three single covalent
bonds, such as in ammonia.
Group 14 and Single Bonds
•
Atoms of group 14 elements form four single
covalent bonds, such as in methane.


21



Single Covalent Bonds
The Sigma Bond
•
Single covalent bonds are called sigma bonds.
•
They occur when the pair of shared electrons is in
an area centered between the two atoms.



22
Multiple Choice

When one atom takes the other atom's electron
When the atom shares an electron with an another atom
When the two nucleus merge
When the neutrons leave the nucleus
23
Multiple Choice

2 pairs
4
2
1
24
Multiple Choice

James
Ionic
Covalent
25
Multiple Choice

1
2
3
4
26
Multiple Choice

How many sigma bonds are in the following molecule?
0
4
8
12
16
27


Multiple Covalent Bonds
Double Bonds
•
A double bond forms when two pairs of electrons are shared
between two atoms.
•
In a. of the figure below, two oxygen molecules form a
double bond.

28



Multiple Covalent Bonds
Triple Bonds
•
A triple bond forms when three pairs of electrons are shared
between two atoms.
•
In b. of the figure below, a triple bond forms between
two nitrogen atoms.
29



Multiple Covalent Bonds
The pi Bond
•
A multiple covalent bond
consists of one sigma bond
and at least one pi bond.
The figure shows the
multiple bond between
carbon atoms in C2H4.
•
A pi bond forms when
parallel orbitals overlap
and share electrons.
30
Multiple Choice

1
2
3
4
31
Multiple Choice

1
2
3
4
32
Multiple Choice

1 sigma and 1 pi
2 sigma and 1 pi
1 sigma and 2 pi
2 sigma and 2 pi
33
Multiple Choice
0
1
2
3
34
Multiple Choice
0
1
2
3
35
Multiple Choice

How many sigma bonds are in the following molecule?
0
1
2
5
6
36


The Strength of Covalent Bonds
Bond Length
•
The strength depends on the distance between the two nuclei, or
bond length.
•
As length increases, strength decreases.
Bonds and Energy
•
The amount of energy required to break a bond is called the bond
dissociation energy.
•
The shorter the bond length, the greater the energy required to
break it.
37


The Strength of Covalent Bonds
• An endothermic reactionoccurs when a greater
amount of energy is required to break the existing
bonds in the reactants than is released when the new
bonds form in the products.
• An exothermic reactionoccurs when more energy is
released during product bond formation than is
required to break bonds in the reactants.
38
Multiple Choice

39
Multiple Choice

6
3
6 pairs
5
40


Think of the quiz answers before moving
to the next slide
41

Quiz
Atoms in nonionic pairs gain or lose electrons.
D
Atoms with lower energy states are more stable.
C
Atoms with higher energy states are more stable.
B
Metals gain stability by sharing electrons to form ions.
A
Which of the following is true?
1.
42

Quiz
Atoms in nonionic pairs gain or lose electrons.
D
Atoms with lower energy states are more stable.
C
Atoms with higher energy states are more stable.
B
Metals gain stability by sharing electrons to form ions.
A
Which of the following is true?
1.
CORRECT
43

Quiz
molecule
D
covalent bond
C
metallic bond
B
ionic bond
A
Which term refers to a chemical bond that
results from sharing valence electrons?
2.
44

Quiz
molecule
D
covalent bond
C
metallic bond
B
ionic bond
A
Which term refers to a chemical bond that
results from sharing valence electrons?
2.
CORRECT
45

8
D
3
C
1
A
2
B
How many electrons do group 17 elements need to form
an octet?
3.
Quiz
46

8
D
3
C
1
A
2
B
How many electrons do group 17 elements need to form
an octet?
3.
CORRECT
Quiz
47

Quiz
sigma
D
Lewis
C
ionic
B
pi
A
Which term refers to single covalent bonds?
4.
48

Quiz
sigma
D
Lewis
C
ionic
B
pi
A
Which term refers to single covalent bonds?
4.
CORRECT
49

None of the above is true.
D
It consists of two sigma bonds and more than two pi
bonds.
C
It consists of one sigma bond and at least one
pi bond.
A
It consists of more than one sigma bond.
B
Which of the following is true of a multiple covalent
bond?
5.
Quiz
50

None of the above is true.
D
It consists of two sigma bonds and more than two pi
bonds.
C
It consists of one sigma bond and at least one
pi bond.
A
It consists of more than one sigma bond.
B
Which of the following is true of a multiple covalent
bond?
5.
CORRECT
Quiz

The Covalent Bond
Lesson 1
Show answer
Auto Play
Slide 1 / 50
SLIDE
Similar Resources on Wayground

47 questions
Energetics 22-23
Presentation
•
9th - 10th Grade

45 questions
Organic and Intermolecular Forces
Presentation
•
10th Grade

45 questions
Electronegativity
Presentation
•
10th Grade

46 questions
Cladograms and Levels of Organization
Presentation
•
9th Grade

44 questions
Unit 2 Review
Presentation
•
9th Grade

44 questions
6.9 Quiz Review
Presentation
•
11th Grade

43 questions
Chemistry Review for Final 3: Periodic Trends and Ions
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

10 questions
Exploring Air Masses and Weather Fronts
Interactive video
•
6th - 10th Grade

20 questions
Cladograms and Phylogenetic Trees
Quiz
•
9th - 12th Grade

31 questions
STAAR Strand 1 TEKS(11,12)
Quiz
•
9th - 12th Grade