

Heating Cooling Curves
Presentation
•
Science
•
9th Grade
•
Hard
Joseph Anderson
FREE Resource
7 Slides • 22 Questions
1
U01.L03 :
Heating and Cooling Curves
2

3
Fill in the Blanks
Type answer...
4
Fill in the Blanks
5

It shows how state of matter changes with temperature.
The positive slope means thermal energy is being added.
Heating
Curve
6
How to Read
During a phase change, the temperature does NOT change until every particle has switched phases.
Energy is being removed or added, but temperature does not change during the phase change.
Temperature changing is demonstrated with the sloped lines.
Phase changing is demonstrated with the flat lines.
7
Multiple Choice

What label is given to the first flat line on the graph?
temperature
boiling point
melting point
time
8
Multiple Choice

At the first flat line on the graph, what state of matter should we see?
mix of solid and liquid
mix of liquid and gas
mix of solid and gas
solid
9
Multiple Choice

What label is given to the second flat line on the graph?
temperature
boiling point
melting point
time
10
Multiple Choice

At the second flat line on the graph, what state of matter should we see?
solid
liquid
gas
mix of liquid and gas
11
Multiple Choice

The temperature of this substance is always increasing on this graph.
true
false
12
Multiple Choice

Thermal energy is always being added on this graph.
true
false
13

It shows how state of matter changes with temperature.
The negative slope means thermal energy is being removed.
Flat lines indicate the temperature is NOT changing, but the matter is changing states.
Cooling
Curve
14
Multiple Choice

What label is given to the first flat line on the graph?
temperature
boiling point
melting point
time
15
Multiple Choice

At the first flat line on the graph, what state of matter should we see?
mix of solid and liquid
mix of liquid and gas
mix of solid and gas
gas
16
Multiple Choice

What label is given to the second flat line on the graph?
temperature
boiling point
melting point
time
17
Multiple Choice

At the second flat line on the graph, what state of matter should we see?
solid
liquid
mix of solid and liquid
mix of liquid and gas
18
Multiple Choice

The temperature of this substance is always decreasing on this graph.
true
false
19
Multiple Choice

Thermal energy is always being removed on this graph.
true
false
20
Remember!
During a phase change, the temperature does NOT change until every particle has switched phases.
Energy is being removed or added, but temperature does not change.
Temperature changing is demonstrated with the sloped lines.
Phase changing is demonstrated with the flat lines.
21
Multiple Choice
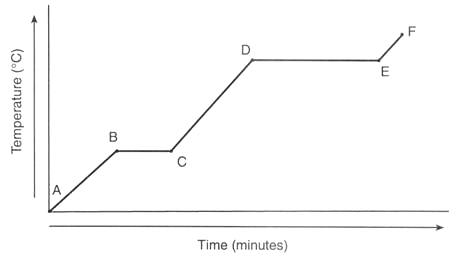
Between which points is the temperature of the substance remaining constant?
A-B only
A-B, C-D, and E-F
B-C only
B-C and D-E
22
Multiple Choice

The melting point of the sample is
-60 ºC
20 ºC
60 ºC
100 ºC
23
Multiple Choice

Which letter indicates where water is in both the solid and liquid phase at the same time?
A
B
C
D
E
24
Multiple Choice

In which region(s) does temperature increase?
Region C only
Regions B and D
Regions A, C, and E
Regions A and B
25
Multiple Choice

Describe the substance at letter A.
Solid
Liquid
Melting
Evaporating
26
Multiple Choice

What is the freezing point of the substance?
0oC
60oC
120oC
180oC
27
Multiple Choice

Describe the substance at letter E
28
Multiple Choice

Given the heating curve of a solid being heated at a constant rate, what is the boiling point of this substance?
20°C
50°C
110°C
170°C
29
You're finished!
Once you submit, check your grade. If you don't like it, you may redo this assignment to improve your score.
U01.L03 :
Heating and Cooling Curves
Show answer
Auto Play
Slide 1 / 29
SLIDE
Similar Resources on Wayground

23 questions
Evolutionary Relationships
Presentation
•
9th - 10th Grade

26 questions
Cell Membrane and Transport
Presentation
•
9th Grade

20 questions
Photosynthesis and Cellular Respiration
Presentation
•
9th Grade

23 questions
Physical vs. Chemical Properties/Changes (2022 Review)
Presentation
•
8th - 9th Grade

21 questions
Solar System Vocabulary
Presentation
•
8th Grade

21 questions
Net Force Review
Presentation
•
8th Grade

20 questions
Macromolecule review
Presentation
•
9th Grade

23 questions
Branches of Earth Science
Presentation
•
9th - 10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade