

Introduction to Atomic Theory
Presentation
•
Chemistry
•
11th Grade
•
Hard
Joseph Anderson
FREE Resource
16 Slides • 44 Questions
1
Timeline and Scientists

2
Democritus (400 B.C. / Greece)
Known For: naming the atom
Atomos: means uncuttable in Greek
Experiment: Guessed (no scientific data)
Atomos
3
Multiple Choice

4
Aristotle (384 B.C. to 322 B.C.)
Said all materials on Earth were not made of atoms, but of the four elements, Earth, Fire, Water, and Air.
His theory held true for the next 2000 years.

5
Open Ended
Why do you think Aristotle's theory of the atom was more accepted than Democritus' theory?
6
Multiple Choice
Which of the following scientists was the first to consider atoms made up matter?
Democritus
Aristotle
Dalton
Chadwick
7
John Dalton (1808 / England)
Known For: Atoms were indivisible (cannot be divided)
4 Postulates (laws/rules)
See next slide
Billiard Ball or Solid Sphere Model

8
Dalton's Contributions
Elements consist of indivisible small particles (atoms).
All atoms of the same element are identical; different elements have different types of atom.
Atoms can't be created nor destroyed. (Law of Conservation of Mass)
Compounds are formed when atoms of different elements join in simple ratios to form molecules (Law of Definite Proportions).
9
Multiple Choice
10
J.J. Thomson (1897 / England)
Known For: electrons were smaller particles of an atom and were negatively charged.
Experiment:
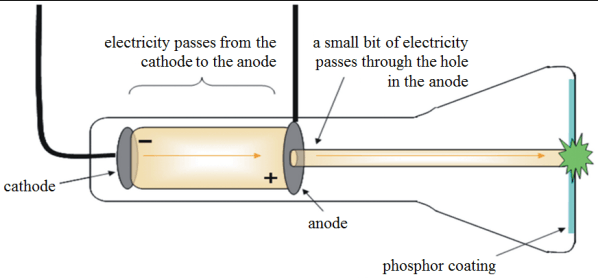
Cathode Ray Tube Experiment
Plum Pudding or Chocolate Chip


11
12
Multiple Choice
13
Multiple Select

What were J. J. Thomson's three contributions to the atomic theory?
Discovered the electron
Discovered the nucleus -did the gold foil experiment
Used the cathode ray tube in his discovery
Created a model of the atom with electrons moving around the nucleus in fixed orbits
Created the "plum pudding" model of the atom
14
Multiple Choice
J. J. Thomson's model of the atom includes all BUT ONE of these features. That is
electrons revolved around the positive nuclear sphere.
the positive atomic charge is spherically distributed.
electrons radiated throughout the positive sphere.
an atom contains both positive and negative charges.
15
Ernest Rutherford (1911 / England)
Known For: Protons (positive charged) and atoms were mostly empty space
Experiment:
Gold Foil Experiment
1. What was expected
2. What actually happened
Nuclear Model


16
Vocabulary
Alpha particles (a)
Particles consisting of two protons and two neutrons tightly bound together (this is a Helium atom - He).
17
18
Categorize
Atomos
Uncuttable

Uncuttable


Billiard ball model or Solid Sphere model
Discovered electrons
Cathode Ray Tube
Plum Pudding
all matter is composed of 4 elements.
4 postulates
19
Multiple Choice
20
Multiple Choice
Which of the following scientists was the first to determine the existence of electrons?
Dalton
Thomson
Bohr
Chadwick
21
Multiple Choice
Which scientists introduced to the idea of a nucleus in the atom using the gold foil experiment?
Democritus
Aristotle
Thomson
Rutherford
22
Multiple Choice

23
Multiple Choice
24
Multiple Choice
25
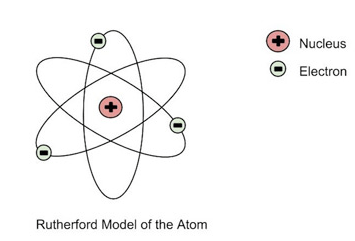
Niels Bohr (1913 / England)
Known For:
1. Determining that electrons have distinct amounts of energy.
2. Electrons absorb energy and jump to higher energy levels. (unstable)
3. Electrons release energy in the form of light when they go back to the original orbital. (stable)
Experiment:
Emissions Spectrum (color of light)
"Planetary model"

26
27
Multiple Choice
Which scientist theorized electrons orbit the nucleus in the atom like planets around the sun?
Rutherford
Chadwick
Dalton
Bohr
28
Multiple Choice
29
Multiple Choice

30
Multiple Choice
According to the model Bohr created, electrons orbit the nucleus in defined locations known as
carapaces
integuments
cartridges
shells
31
Erwin Schrödinger (1926 / Austria)
A.K.A.: Modern Model of the Atom
Known For: Quantum mechanics
electrons can only exist in specified energy states
Experiment:
mathematical equations
"Electron Cloud model"

32
James Chadwick (1932 / England)
Known For: Discovered neutrons
neutral particles in the nucleus of an atom
Experiment:
mathematical equations (atom was too heavy with just protons)
"Quantum model" or Modern Model of the Atom


33
Summary of Current Atom Model
Subatomic Particle | Charge | Relative Mass (amu) | Location |
|---|---|---|---|
Proton | +1 | 1 | in the nucleus |
Neutron | -1 | 1 | in the nucleus |
Electron | 0 | 1/1840 | outside the nucleus |

34
Multiple Choice
35
Multiple Choice
36
Theories
Dalton’s Atomic Theory: Dalton
All matter is composed of atoms.
Atoms of a given element are identical
Today: Not true - there are isotopes
Atoms cannot be divided, created, or destroyed
Technically, no, but there are smaller parts (p, n, and e)
Atoms of different elements combine in simple whole number ratios to form chemical compounds.
In chemical reactions, atoms are separated, rearranged, or combined.
Quantum Theory
1. Atoms are made of smaller particles
2. Atoms of the same element (isotopes) can vary in mass (neutrons)
37
Multiple Choice
38
Multiple Choice

Which scientist introduced the atom model shown?
Dalton
Thomsom
Rutherford
Heisenberg
39
Multiple Choice
Which of the following is NOT included in our modern day model of the atom?
Protons and Neutrons in the center
Electrons orbit in energy levels like planets around the sun
The atom is mostly empty space.
Electrons can be found in orbitals outside the nucleus
40
Multiple Choice
Which of the following represents our modern day model of the atom?
41
Multiple Choice
Which of the following models is considered the Plum Pudding Model?
42
Multiple Choice

43
Multiple Choice
Which experiment listed below did Thomson use to discover electrons?
Cathode Ray Tube
Oil Drop Experiment
Cat Experiment
Gold Foil Experiment
44
Multiple Choice
What is the name of our modern day model of the atom?
Plum Pudding Model
Electron Orbit Model
Pool Ball Model
Quantum Mechanical Model
45
Multiple Choice
Who was the first contributor to the atomic theory?
John Dalton
Ernest Rutherford
J.J. Thomson
Democritus
Niels Bohr
46
Multiple Select

What were Ernest Rutherford's three contributions to atomic theory?
Discovered that the atom is mostly empty space
Discovered the nucleus- did the gold foil experiment
Used the cathode ray tube in his discovery
Discovered the electron
Discovered that the proton had a positive charge
47
Multiple Choice
48
Multiple Choice
49
Multiple Choice

50
Multiple Choice
51
Multiple Choice

What explains the path of the cathode ray in the image above?
Gravity pulls the particles down.
The particles are repelled by the positive electrode.
The particles are attracted towards the negative electrode.
The particles are deflected away from the negative electrode and attracted to the positive electrode.
52
Multiple Choice

53
Multiple Choice

54
Multiple Choice
Sir J.J. Thompson discovered the electron in 1897. How did Thompson explain the overall neutral charge of the atom?
Electrons are present all over the atom, even in the nucleus.
Electrons alternate between positive and negative charges.
Negative electrons were inside a cloud of positive charge.
Electrons travel around the nucleus at great speeds.
55
Multiple Choice
In the famous gold foil experiment, radioactive particles were fired at a very thin sheet of gold foil. Most of the particles passed straight through the gold foil. Only about 1 in every 8000 particles were deflected. The gold foil experiment showed the researchers that the atom is made mostly of
protons
electrons
neutrons
empty space
56
Multiple Choice
These particles are found in the nucleus of the atom.
protons only
protons and electrons
electrons only
protons and neutrons
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
Subatomic particles with a negative charge
Electrons
Neutrons
Protons
Quarks
60
Multiple Choice
Timeline and Scientists

Show answer
Auto Play
Slide 1 / 60
SLIDE
Similar Resources on Wayground

54 questions
Our Beautiful Planet
Presentation
•
11th Grade

58 questions
Unit 3: Chemical Bonds
Presentation
•
10th Grade

57 questions
Module 6: Lesson 3: Names & Formula for Ionic Compounds (pg 180)
Presentation
•
10th Grade

57 questions
Reading strategies
Presentation
•
11th Grade

55 questions
Unit 7 lesson part 4 Presidents
Presentation
•
11th Grade

56 questions
Edexcel IAL S1 Representation of Data
Presentation
•
11th Grade

54 questions
C1-UNIT 10- CLAUSES OF REASON, PURPOSE, RESULT AND CONCESSIO
Presentation
•
11th Grade

51 questions
Average Atomic Mass
Presentation
•
11th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade