

Number of Neutrons
Presentation
•
Science
•
8th - 9th Grade
•
Hard
Joseph Anderson
FREE Resource
19 Slides • 27 Questions
1
proton number, nucleon number and isotopes

2

3
Multiple Choice
Which one of the below is the nucleon number of an atom?
he number of neutrons
the number of protons
the total number of protons and neutrons
the total number of protons and electrons
4

5
Multiple Choice

look at the table and answer the question. Which row describes the structure of the positively charged sodium atom? sodium atom's mass number is 23 and atomic number is 11
A
B
C
D
6
Multiple Choice

An isotope of chromium is represented in the picture. Which statement about an atom of this isotope of chromium is correct?
It contains 24 electrons
It contains 24 neutrons.
It contains 28 protons.
It contains 52 neutrons
7
Multiple Choice

How many neutrons are present in the atom represented in the picture
21
24
45
66
8
Multiple Choice
An aluminium atom has a nucleon number of 27 and a proton number of 13.
How many neutrons does this aluminium atom contain?
13
14
27
40
9
Multiple Choice
Which pair of atoms contains the same number of neutrons?
10
Multiple Choice

check the table and give the correct answer. Which row gives the number of protons, electrons and neutrons found in an atom of zinc? mass number of zinc is 65 and its atomic number is 30
A
B
C
D
11

12
Multiple Choice

What number should be written under the 33 in this isotope notation for sulfur?
-1
17
16
32
13
Multiple Choice

How many electrons does this isotope of beryllium have?
9
5
4
2
14
Multiple Choice

What is the mass number of this atom?
13
14
27
40
15
Multiple Choice
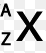
In a correctly written nuclear notation what would be located in the "A" position?
number of neutrons
atomic number
number of electrons
mass number
16
Multiple Choice

What do these isotopes of carbon all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and protons
protons, atomic number, and mass number
17
Multiple Choice

18
Multiple Choice

What is the atomic number of this atom?
2
4
6
none of the above
19
Multiple Choice

What is the atomic number of this atom?
2
4
6
none of the above
20

21

22

23
Multiple Choice
Which statements about isotopes of the same element are correct?
1. They are atoms which have the same chemical properties because they have the same number of electrons in their outer shell.
2. They are atoms which have the same number of electrons and neutrons but different numbers of protons.
3. They are atoms which have the same number of electrons and protons but different numbers of neutrons.
1 and 2
1 and 3
2 only
3 only
24
Multiple Choice
Which definition of isotopes is correct?
atoms of the same element that have the same number of electrons and nucleons
atoms of the same element that have the same number of neutrons and protons
atoms of the same element that have the same number of protons but a different number of electrons
atoms of the same element that have the same number of protons but a different number of
nucleons
25
Multiple Choice
Which statement about the isotopes of an element is correct?
Their physical properties are different because they have different proton numbers.
Their atomic masses are different because they have different numbers of electron shells.
They have the same chemical properties because they have the same number of electrons in their outer shells.
They have the same physical properties because they have the same number of neutrons in
their nuclei.
26
Multiple Choice
Which statement explains why isotopes of an element have the same chemical properties?
They have different numbers of neutrons.
They have the same number of electrons as protons.
They have the same number of electrons in the outer shell.
They have the same number of protons in the nucleus.
27
Multiple Choice

Which row describes isotopes of the same element in the table?
A
B
C
D
28
Multiple Choice

The number of particles in atoms W, X, Y and Z are shown in the table. which statement is correct.
W and X are isotopes of carbon.
X and Y are isotopes of nitrogen
X has a mass number of 12.
Z has an atomic number of 8
29
Multiple Choice

Carbon has several Isotopes, for example, carbon 12 and carbon 13 as shown in the table. which number of subatomic particle is same for these two atoms.
proton number
neutron number
nucleon number
boson number
30
Multiple Choice

Carbon has several Isotopes, for example, carbon 12 and carbon 13 as shown in the table. which number of subatomic particle is different for these two atoms.
proton number
neutron number
electron number
fermion number
31
Multiple Choice

which one of the below is an isotope of the atom in the picture.
32
Multiple Choice
33
Multiple Choice
34
Multiple Choice

What do these isotopes of carbon all have in common?
neutrons & mass number
atomic number and neutrons
atomic number and electrons
protons, atomic number, and mass number
35

36

37

38

39

40

41

42

43

44

45

46

proton number, nucleon number and isotopes

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

38 questions
Life Cycle of a Star
Presentation
•
8th Grade

44 questions
Biogeochemical Cycles
Presentation
•
8th - 10th Grade

41 questions
Earth, Sun, Moon Relationships
Presentation
•
9th Grade

39 questions
Mirror and Lenses
Presentation
•
8th Grade

44 questions
Animal Kingdom
Presentation
•
9th - 10th Grade

38 questions
PA - Linear Vs Nonlinear, That is the Question!
Presentation
•
8th - 9th Grade

40 questions
Causes of the Civil War Part 2 SHEET 2
Presentation
•
8th Grade

42 questions
1-Chemistry Basics Lesson
Presentation
•
8th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

30 questions
GMAS Physical Science Review
Quiz
•
8th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

18 questions
Carbon Cycle
Quiz
•
8th Grade

10 questions
Exploring Earth's Seasons and Their Causes
Interactive video
•
6th - 8th Grade

26 questions
Amplify Chemical Reactions
Quiz
•
6th - 8th Grade