

Specific Latent Heat Review
Presentation
•
Science
•
9th - 11th Grade
•
Hard
Joseph Anderson
FREE Resource
7 Slides • 15 Questions
1
Specific Latent Heat
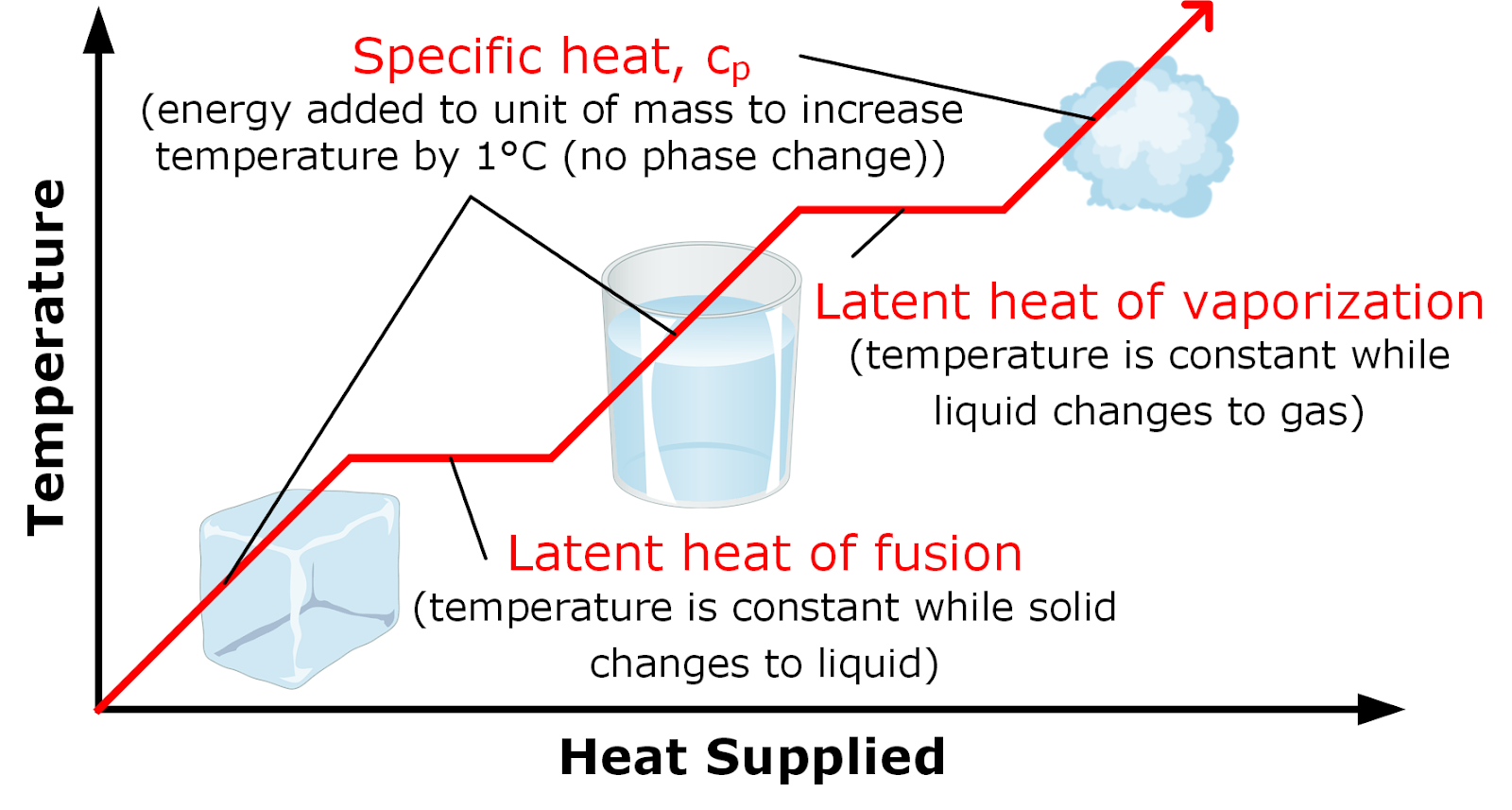
2

3

4
Multiple Choice

The diagram shows a cooling curve for water. Which line represents the water in a solid form?
A
B
C
D
E
5
Multiple Choice

The diagram shows a cooling curve for water. Which line represents the water freezing?
A
B
C
D
E
6
Multiple Choice

The diagram shows a cooling curve for water. Which line represents the it in a liquid state (water)?
A
B
C
D
E
7
Multiple Choice
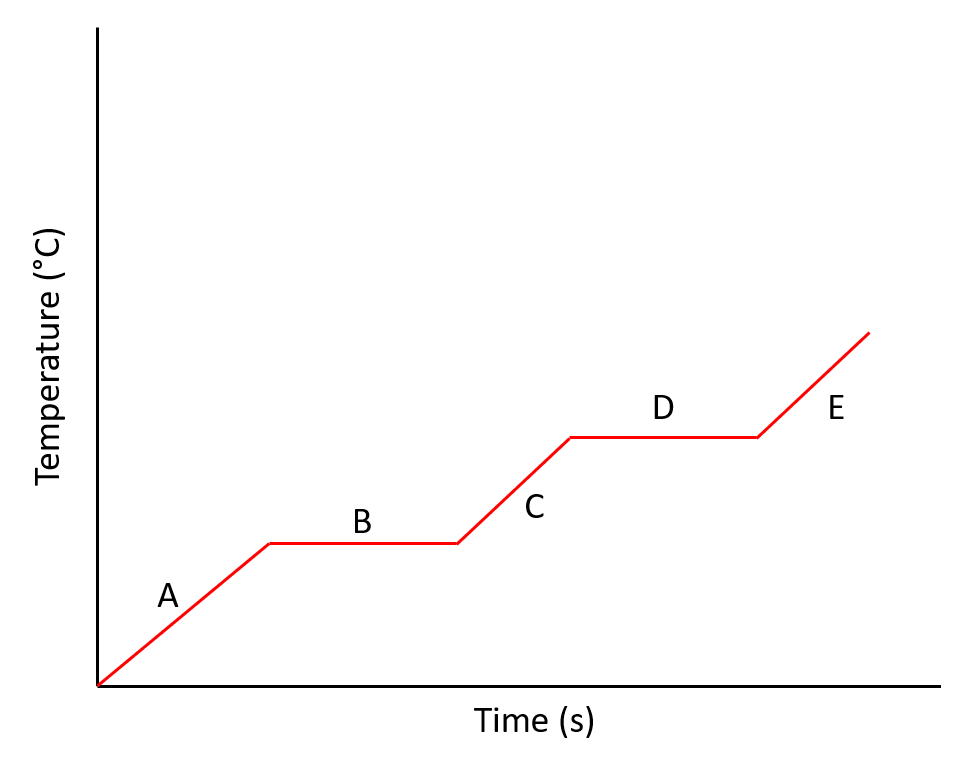
The diagram shows a heating curve for water. Which line represents the it in a liquid state (water)?
A
B
C
D
E
8
Multiple Choice

The diagram shows a heating curve for water. Which line represents ice melting?
A
B
C
D
E
9
Multiple Choice

Why are lines B and D flat?
The ice is melting
The ice is freezing
All of the thermal energy is used to break the bonds
They stopped the stopwatch
They stopped heating the ice.
10
Multiple Choice
Latent Heat is
the heat absorbed or released at a boiling process during a change of state
the heat absorbed or released at a constant temperature during a change of state
the heat absorbed or released at a constant temperature during a change of state of 1 kg object
the heat absorbed or released by an object to increase or decrease its temperature by 1 degree Celcius
11
12
Fill in the Blanks
Type answer...
13

14
Multiple Choice
The heat absorbed by melting a solid is known as
latent heat of fusion
latent heat of solid
latent heat of liquid
latent heat of vaporisation
15
Multiple Select
Select the state changes that latent heat of fusion applies to.
melting
evaporating
freezing
condensing
sublimation
16
Multiple Select
Select the state changes that latent heat of vaporisation applies to.
melting
evaporating
freezing
condensing
sublimation
17
Multiple Choice
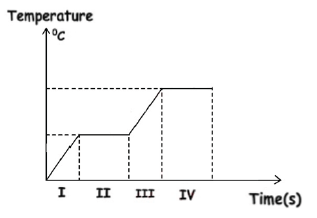
Consider the following temperature-time graph when a solid is heated. which part of the graph involves latent heat of vaporization?
Part I
Part II
Part III
Part IV
18

19

20
Multiple Select
Select the correct equations.
thermal energy = specific latent heat / mass
thermal energy = specific latent heat x mass
mass = specific latent heat / thermal energy
specific latent heat = thermal energy / mass
mass = specific latent heat x thermal energy
21
Multiple Choice
How much energy is required to melt down a 2000 g of mercury?(latent heat of fusion is 1.14 x 104 J/kg)
2.55 x 104 J
2.28 x 104 J
2,280 x 104 J
3.28 x 104 J
22
Multiple Choice
How much energy is required to change 5 kg of ice at 0 ° C into water at the same temperature? (latent heat of fusion is 3.36 X 106 J/kg )
16.1 X 106 J
16.8 X 106 J
15.1 X 106 cal
16.8 X 106 cal
Specific Latent Heat

Show answer
Auto Play
Slide 1 / 22
SLIDE
Similar Resources on Wayground

14 questions
Minerals
Presentation
•
9th - 12th Grade

20 questions
Soil Formation and Earth’s Structure
Presentation
•
9th - 10th Grade

18 questions
Selection Graphs
Presentation
•
8th - 10th Grade

16 questions
Sex-linked traits
Presentation
•
10th Grade

16 questions
Observations and Inferences
Presentation
•
9th - 12th Grade

15 questions
ES.4 Minerals
Presentation
•
9th - 12th Grade

15 questions
ATP
Presentation
•
9th - 10th Grade

19 questions
Sedimentary Rock Cycle Part 1
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Naming Ionic and Covalent Compounds
Quiz
•
9th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

21 questions
Biology EOC Review (ecology)
Quiz
•
9th - 12th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

30 questions
Environmental Science Final Exam REVIEW
Quiz
•
9th - 12th Grade