

Atomic Theory
Presentation
•
Science
•
6th Grade
•
Hard
Joseph Anderson
FREE Resource
6 Slides • 37 Questions
1
Big Idea 8: Properties of Matter
Atoms and States of Matter

2
Big Idea 8: Properties of Matter
All objects and substances in the world are made of matter. Matter has two fundamental properties: matter takes up space and matter has mass which gives it inertia.
Objects and substances can be classified by their physical and chemical properties. Mass is the amount of matter (or "stuff") in an object. Weight, on the other hand, is the measure of force of attraction (gravitational force) between an object and Earth.
The concepts of mass and weight are complicated and potentially confusing to elementary students. Hence, the more familiar term of "weight" is recommended for use to stand for both mass and weight in grades K-5. By grades 6-8, students are expected to understand the distinction between mass and weight, and use them appropriately.
3
Explore the scientific theory of atoms (also known as atomic theory) by recognizing that atoms are the smallest unit of an element and are composed of sub-atomic particles (electrons surrounding a nucleus containing protons and neutrons). (SC.8.P.8.7 - DoK 1)
A scientific theory is supported by evidence and is different from an opinion, belief, or hypothesis. Because theories are based on evidence, they may be modified as better technology is developed and new evidence is collected, but they are rarely discarded completely.
Scientific theories are different from laws because they explain why something occurs in the the natural world, rather than what is occurring. Because of this scientific theories never become laws and laws never become theories.
Every THING in our universe contains matter and is made of atoms and molecules. The only things that are not are not actual things, like energy and forces.
Atoms are made of sub-atomic particles. Protons and neutrons make up the nucleus, or center of the atom, while electrons orbit the nucleus.
Protons have a postive charge, while electrons have a negative charge and neutrons are neutral, or have no charge.
Protons and neutrons each have one atomic mass unit, while electrons are so small that their mass is considered insignificant.
Elements are all of the different types of atoms and are identified by their number of protons.
4
5
Multiple Choice

Neon (Ne) is an element, one of many different kinds of matter that occur on Earth. The diagram shows a model of one atom of the element neon. Which of the following statements best describes the atom shown in the diagram?
It is identical with atoms of some other elements.
It can be broken down into smaller parts by chemical means.
It is the smallest unit of neon that has all the properties of neon.
It is the largest unit of neon that can combine with other elements.
6
Multiple Choice

Neon (Ne) is an element, one of many different kinds of matter that occur on Earth. The diagram shows a model of one atom of the element neon. The atom of neon shown is made up of subatomic particles. Which of the following are shown in the diagram?
electrons
neutrons
protons
ions
7
Multiple Choice

In its natural state, neon has no definite shape or volume. Its atoms move fast and are far apart. Which term describes neon in its natural state?
gas
liquid
solid
solution
8
Multiple Choice
Which is the smallest unit of matter?
compound
mixture
atom
nucleus
9
Multiple Choice

The illustration is a model of an atom. Which two subatomic particles make up the nucleus of an atom?
proton and electron
neutron and proton
electron and neutron
ion and proton
10
Multiple Choice
Contributes basically no mass to an atom.
protons
neutrons
electrons
protons and neutrons
11
Multiple Choice
True or False: The majority of an atom is made up of empty space
True
False
12
Multiple Choice

People have always agreed that matter is made of atoms.
True
False
13
Multiple Choice
All matter is made of
energy
atoms
air
chemistry
14
Multiple Choice
Something that takes up space and has mass.
volume
matter
mass
density
15
Multiple Choice

Why is the overall charge of an atom neutral (0)?
They have the same number of protons and neutrons.
They have the same number of protons and electrons.
They have the same number of electrons and neutrons.
16
Multiple Choice
What particle uniquely identifies an element?
Number of Valence Electrons
Number of Protons
Number of Electrons
Atomic Mass
17
Multiple Choice
What subatomic particles would you find in the nucleus of an atom?
Protons only
Protons and Neutrons
Neutrons and Electrons
Protons and Electrons
18
Multiple Choice
What two particles determine the mass number?
Protons and electrons
Electrons and neutrons
Protons and neutrons
19
Multiple Choice
The positive particles of an atom are
electrons
positrons
neutrons
protons
20
Multiple Choice
How did each model of the atom help to develop the atomic theory?
Each model provided opinions that were added.
Each model showed different properties of the same structure.
Each model showed new particles that had been discovered.
Each model built upon the other to show new particles or properties of previously discovered particles.
21
Explore the scientific theory of atoms (also known as atomic theory) by using models to explain the motion of particles in solids, liquids, and gases. (SC.8.P.8.1 - DoK 2)
The kinetic energy, or motion, of the particles (atoms and molecules) in a substance determine its state of matter. Atoms are ALWAYS moving. The four states of matter are: solid, liquid, gas, and plasma.
Solids have particles that are packed closely together and vibrate in place. Anything in the form of a solid is technically "frozen".
When you add energy (heat) to a solid, it will eventually reach its melting point and change states to a liquid. Freezing point and melting point are technically the same thing, it just depends on whether energy is being added or taken away from the substance.
The particles in a liquid have enough energy that they are able to slide past/around each other. Liquids take the shape of the container they are held in.
When you add energy (heat) to a liquid, it will eventually reach its boiling point and change states to a gas. During a state change, temperature remains constant.
The particles in a gas are moving rapidly and fill the shape and space of their container.
Plasma has all of the same properties of a gas, except its particles have an electrical charge. The sun, lighting, and fluorescent light bulbs are all plasma.
22
23
Multiple Choice
Madison is heating two different liquids. For each liquid, she wants to compare the temperatures at which gas bubbles form, rise, and begin to escape. What physical property is Madison using to compare the liquids?
boiling point
conductivity
melting point
melting point
24
Multiple Choice

Melinda drew an illustration of the three states of matter. The illustration shows what she drew. Melinda wanted to replace the numbers with labels identifying the correct state of matter. How should she change the numbers?
1 --> solid; 2 --> liquid; 3 --> gas
1 --> gas; 2 --> liquid; 3 --> solid
1 --> liquid; 2 --> solid; 3 --> gas
1 --> liquid; 2 --> gas; 3 --> solid
25
Multiple Choice
Atoms and molecules are constantly in motion. The more energy the atom or molecule contains, the faster they move about. In which state of matter do the atoms and molecules have the most energy?
in a gas
in a solid
in a liquid
in both a liquid and a solid
26
Multiple Choice
As temperature increases, what happens to particles?
They speed up
They slow down
27
Multiple Choice

If the tops were removed from the cylinders, the following would happen:
The solid would fly out of the container
The liquid would fly out of the container.
The gas would fly out of the container.
They ALL would fly out of the container.
28
Multiple Choice

In the above diagram, "State of Matter B" is a liquid. This means that it has:
definite shape and volume.
no definite shape or volume.
definite volume, but changing shape.
definite shape, but changing volume.
29
Multiple Choice

Which state of matter does the diagram best represent?
Solid
Liquid
Gas
30
Multiple Choice

What state of matter does the picture best describe?
Solid
Liquid
Gas
31
Multiple Choice

What state of matter is segment 5?
Solid
Liquid
Gas
Plasma
32
Multiple Choice
Fluid states are
solid and gas
liquid and gas
solid and liquid
solid, liquid and gas
33
Multiple Choice

From 30 degrees to 55 degrees, what state of matter is the substance?
Solid
Liquid
Gas
Plasma
34
Multiple Choice
Which is a property of matter that you CANNOT observe just by looking at an object?
temperature
shape
color
35
Multiple Choice
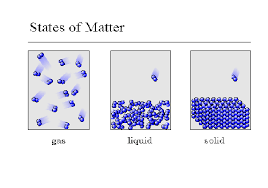
At higher temperatures
particles in an object have less energy
particles in an object move faster
a gas contracts
36
Multiple Choice
Define a liquid.
Indefinite shape, indefinite volume
Definite shape, definite volume
Found in stars
Definite volume, indefinite shape
37
Multiple Choice
The state of matter that has no definite size or shape is
Solid
Liquid
Gas
38
Multiple Choice
Particles (molecules) in a ______________________ have more energy than the other states of matter.
gas
solid
liquid
39
Multiple Choice
Particles of a liquid
are tightly packed together and stay in a fixed position.
have no viscosity.
decrease in volume with increasing temperature.
are free to move around one another but still touch.
40
Multiple Choice
What term describes a liquid changing to a solid?
Deposition
Sublimation
Evaporation
Freezing
41
Multiple Choice
How is a gas defined?
Indefinite shape, indefinite volume
Definite shape, definite volume
Found in stars
Definite volume, indefinite shape
42
Multiple Choice
43
Multiple Choice
True or False: During a state change, temperature does not change.
True
False
Big Idea 8: Properties of Matter
Atoms and States of Matter

Show answer
Auto Play
Slide 1 / 43
SLIDE
Similar Resources on Wayground

36 questions
Levels of Organization - Cells to Biome
Presentation
•
KG

39 questions
Introduction to Claims Evidence Resoning
Presentation
•
6th Grade

36 questions
Ecological Succession & Human Impact on Ground&Surface water
Presentation
•
7th Grade

33 questions
ENERGY TRANSFORMATION
Presentation
•
6th Grade

35 questions
#2 Thermal Energy Lesson
Presentation
•
6th Grade

38 questions
Temperature, Salinity, and Density
Presentation
•
6th Grade

37 questions
Potential and Kinetic Energy Guided Lesson
Presentation
•
6th Grade

41 questions
Heat Transfer Lesson & Quiz
Presentation
•
6th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Food webs and food chains
Quiz
•
4th - 6th Grade

21 questions
Tides Review 6.9B
Quiz
•
6th Grade

10 questions
Exploring the Rock Cycle
Interactive video
•
6th - 8th Grade

20 questions
Cells! Cell Theory and Characteristics of Eukaryotes/Prokaryotes
Quiz
•
6th Grade