

AKS 6 Overview
Presentation
•
Science
•
9th - 12th Grade
•
Medium
+8
Standards-aligned
Marissa Brown
Used 5+ times
FREE Resource
17 Slides • 30 Questions
1
AKS 6 Overview - Solutions, Solubility, Acids/Bases
By Marissa Brown
2
What is Solubility?
Solubility is the mass of solute that can dissolve a fixed mass of solvent at a particular temperature

Remember: a saturated solution is one where no more solute can be dissolved at that particular temperature
3
What is the unit for solubility?
The solubility of a solute in water is usually measured as the mass of solute that will saturate a 100g of water that is, g per 100g water

4
Concentration Terminology
Concentration Terminology:
Dilute Solution (Unsaturated): Small amount of solute in a large amount of solvent.
Concentrated Solution (Unsaturated): Lots of solute in a small amount of solvent (all dissolves).

5
Size of the particles
Breaking a solute into smaller pieces increases its surface area and increases its rate of solution.
When a solute dissolves, the action takes place only at the surface of each particle. When the total surface area of the solute particles is increased, the solute dissolves more rapidly.
6
Concentration Terminology
Saturated Solution: The maximum amount of solute dissolved in a given amount of solvent at a given temperature. To make a saturated solution you add more solute than can be dissolved.

7
Multiple Choice

Numbering from the left, which is most dilute?
1
2
3
4
5
8
Stirring
With liquid and solid solutes, stirring brings fresh portions of the solvent in contact with the solute. Stirring, therefore, allows the solute to dissolve faster.
9
Multiple Choice
How do you know if a solution is saturated?
When you see some undissolved solute at the bottom that doesn't disappear if you stir it.
When it is darker colored.
When it is transparent.
When you can dissolve more solute if you stir it.
10

Electrolytes
Soluble ionic compounds are electrolytes, while most covalent compounds are non-electrolytes (except weak acids, covered in next unit).
Ionic(aq) = electrolyte
Anything else = nonelectrolyte
11
Multiple Choice
You are given a small beaker of solution at room temp. You add a bit of solute to the solution and it dissolves. The solution was:
saturated
unsaturated
concentrated
warm
12
Multiple Choice
The substance being dissolved is called the ________
solvent
solid
solute
solvate
13
Multiple Choice
When no more solute dissolves the solution is _________
supersaturated
unsaturated
saturated
supercalifragilisticexpialidocious
14
Multiple Choice
15
Multiple Choice
Identify the solvent
16
Multiple Choice
17
Multiple Choice

18
Multiple Choice
19
Factors affecting solubility
20
Increase the temperature
For liquids and solid solutes, increasing the temperature not only increases the amount of solute that will dissolve but also increases the rate at which the solute will dissolve. For gases, the reverse is true. An increase in temperature decreases both solubility and rate of solution.
21
Multiple Choice
22
Multiple Choice
A student is preparing solutions for a laboratory experiment by dissolving solid solutes in liquid solvents. Which action will increase the rate of solubility
lowering the temperature of the solvent
stirring the solute in the solution
increasing the pressure on the solution
increasing the particle size of the solute
23
Multiple Choice
Why does the rate of dissolving increase as the temperature increases?
The solute molecules move faster at higher temperatures
The solute particles become smaller at higher temperatures
The solvent molecules become slower at higher temperatures
What is the effect of agitation on the rate of dissolving?
24
Multiple Choice
What is the effect of agitation on the rate of dissolving?
It decreases the rate of dissolving
It has no effect on the rate of dissolving
It increases the rate of dissolving
It changes the color of the solute
25
Multiple Choice
How does agitation help in dissolving a solute more quickly?
It increases the temperature of the solvent
It increases the weight of the solute
It brings fresh solvent into contact with undissolved solid
It changes the shape of the solvent
26
Multiple Choice
Why does a solute with a larger surface area dissolve faster?
It has more solute surface area in contact with the solvent
It has fewer solute particles in contact with the solvent
It has a higher temperature compared to other solutes
It has a different color compared to other solutes
27

28

29

30

31

32
Multiple Choice

Most substances increase in solubility as temperature increases, why don't HCl, NH3, and SO2?
They are insoluble in water
They are all non-polar
They are all polar
They are all gases
33
Multiple Choice
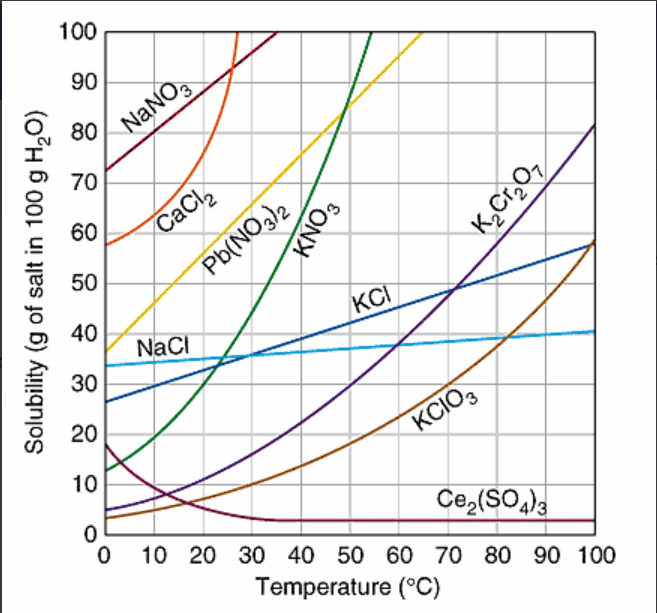
Which substance is MOST soluble at 0oC?
KI
NaNO3
NaCl
Ce2(SO4)3
34
Multiple Choice
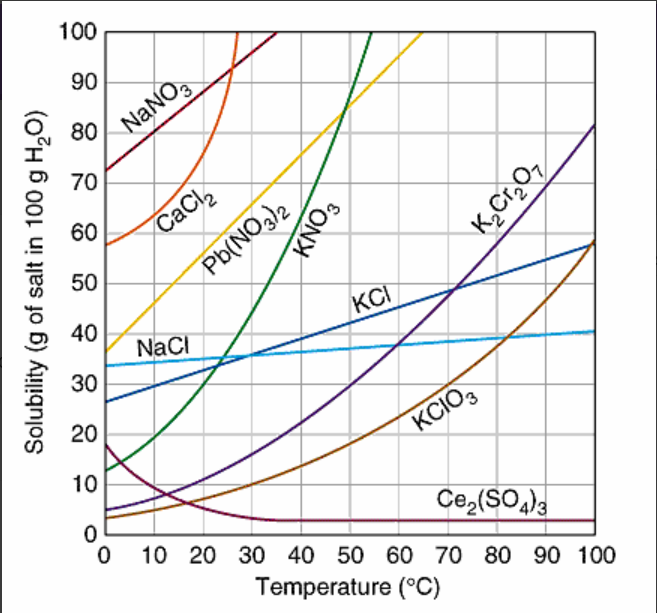
How many grams of K2Cr2O7, are soluble in 100 g of water at 95oC?
83 grams
75 grams
40 grams
12 grams
35
Multiple Choice
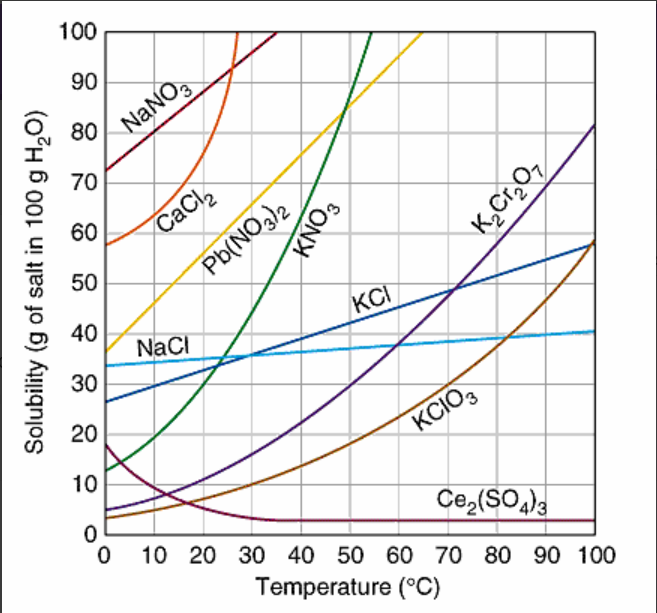
When 42 grams of potassium chloride, KCl, is dissolved in 100 grams of water at 50oC, the solution can be correctly described as:
supersaturated
saturated
unsaturated
36
Multiple Choice
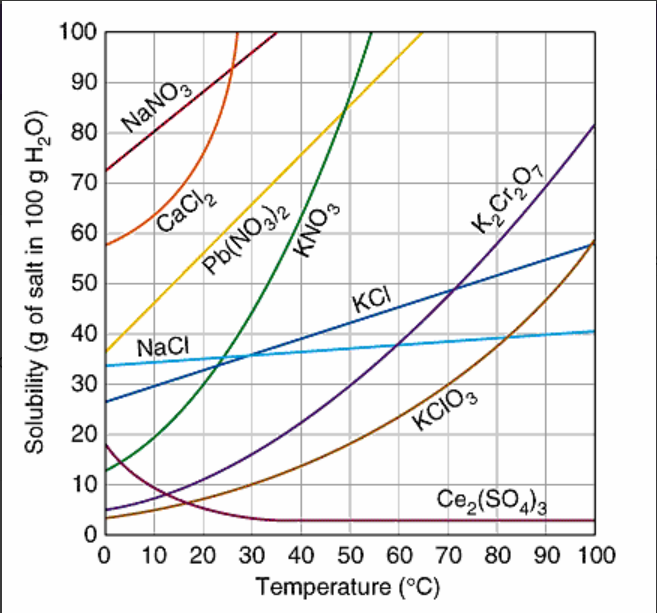
When 20 grams of potassium chlorate, KClO3, is dissolved in 100 g of water at 80oC, the solution can be correctly describe as:
supersaturated
saturated
unsaturated
37

38
Multiple Choice
In an aqueous solution, an acid
absorbs hydrogen ions (H+)
donates hydrogen ions (H+)
releases oxygen gas (O2)
39

40
Multiple Choice
NaOH is a base because,
41
Multiple Choice
Which statement about bases is incorrect?
Bases turn red litmus paper blue
Bases contain hydrogen ions
Bases taste bitter
Bases have a pH 7.1-14.0
42
Multiple Choice
Which of the following is a property of an acid?
Form hydroxide ions
Have a negative charge
Turn litmus paper blue
Have a pH of 0-6.9
43
Multiple Choice
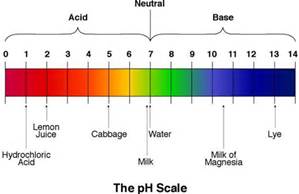
According to the pH range which substance is more acidic than cabbage juice?
lemon juice
lye
milk of magnesia
44
Multiple Choice
Litmus paper is made from water-soluble dyes which are extracted from lichens. This paper is used as an acid-base indicator. Which of these common household substances would turn blue litmus paper red?
Bleach
Lye
Ammonia
Vinegar
45
Multiple Choice
46
Multiple Choice

Observe the chart below. Which substance is likely an acid?
Substance A
Substance B
Substance C
Substance D
47
Multiple Choice
Milk of magnesia is a common ingredient in many heartburn and stomach medications. How does milk of magnesia act to calm and upset stomach?
Milk of magnesia is an acid that neutralizes stomach acid
Milk of magnesia is a base that neutralizes stomach acid
Milk of magnesia has a neutral nature which dilutes stomach acid.
Milk of magnesia is a base that neutralizes bases in the stomach.
AKS 6 Overview - Solutions, Solubility, Acids/Bases
By Marissa Brown
Show answer
Auto Play
Slide 1 / 47
SLIDE
Similar Resources on Wayground

43 questions
Force and Motion
Presentation
•
8th Grade - University

42 questions
Credit Score
Presentation
•
9th - 12th Grade

42 questions
Climate
Presentation
•
9th - 12th Grade

44 questions
Properties of Water
Presentation
•
9th - 12th Grade

41 questions
Interactions between organisms
Presentation
•
9th - 11th Grade

46 questions
Biomolecule terms - photosynthesis and respiration
Presentation
•
9th - 12th Grade

41 questions
Physical and Chemical Properties and Changes
Presentation
•
9th - 12th Grade

40 questions
6.3 Nuclear Energy
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

20 questions
Human Impact on the Environment Review #2
Quiz
•
9th Grade

10 questions
Exploring Natural Selection Concepts
Interactive video
•
6th - 10th Grade

20 questions
Balancing Chemical Equations
Quiz
•
9th Grade

10 questions
Exploring Air Masses and Weather Fronts
Interactive video
•
6th - 10th Grade