

Chemical Reactions: Types.1
Presentation
•
Science
•
10th Grade
•
Practice Problem
•
Medium
Standards-aligned
Pip Smith
Used 1+ times
FREE Resource
19 Slides • 27 Questions
1

2

3
Multiple Choice
Which part of a chemical equation indicates the number of molecules involved in the reaction?
Coefficient
Subscript
Chemical Formula
Yield
4

5
6

write this down
7
Multiple Choice
Which of the following is NOT a type of chemical reaction listed previously?
Synthesis reaction
Decomposition reaction
Neutralization reaction
Combustion reaction
8

write this down
9
Multiple Select
Select all the correct statements about synthesis reactions.
They always produce a single product.
They involve two or more reactants.
They can only occur with elements, not compounds.
They are also known as combination reactions.
10
Fill in the Blanks
Type answer...
11
Open Ended
Explain the difference between a synthesis reaction and a decomposition reaction, using examples if possible.
12

13
Synthesis of NaCl
14
Multiple Choice
Which of the following best represents the synthesis of sodium chloride?
2Na(s) + Cl2(g) → 2NaCl(s)
NaCl(s) → Na(s) + Cl2(g)
2NaCl(s) + O2(g) → 2Na2O(s) + Cl2(g)
Na(s) + Cl2(g) → NaCl2(s)
15

write this down
16
Fill in the Blanks
17

write this down
18
Multiple Choice
Which of the following best describes a decomposition reaction?
A reaction in which two elements combine to form a compound.
A reaction in which a single compound breaks down into two or more elements or new compounds.
A reaction in which two compounds exchange ions.
A reaction in which an element displaces another from a compound.
19
20
Multiple Choice
Based on the experiment shown, what energy source is being used to initiate the decomposition reaction?
Electricity
Light
Heat
Sound
21

22
Open Ended
Explain the process of decomposition of copper carbonate as shown in the diagram. What are the products formed?
23

24
Multiple Choice
Which of the following are possible products when a metal carbonate undergoes decomposition?
Metal oxide and carbon dioxide
Metal and oxygen
Metal hydroxide and water
Metal oxide and water
25

write this down
26
Fill in the Blanks
Type answer...
27

write this down
28
Open Ended
Compare and contrast decomposition reactions and single displacement reactions. Provide an example of each.
29

30
Multiple Choice
Which metal is the most reactive according to the activity series shown?
Lithium
Potassium
Calcium
Aluminum
31
32

33
Multiple Choice
Which of the following statements about the thermite reaction is correct?
Aluminum displaces iron from iron(III) oxide because it is more reactive.
Iron displaces aluminum from aluminum oxide because it is more reactive.
Iron(III) oxide is reduced to iron by magnesium.
Aluminum is less reactive than iron.
34
Open Ended
Explain how the activity series of metals helps predict the outcome of the thermite reaction demonstrated in the experiment.
35

36
Fill in the Blanks
Type answer...
37
Open Ended
How confident do you feel about identifying the different parts of a chemical equation after this lesson?
38
Multiple Choice
What is the role of the coefficient (number that comes before the formula) in a chemical equation?
It shows the number of molecules or atoms involved in the reaction.
It indicates the type of chemical reaction.
It represents the yield of the reaction.
It identifies the subscript in the formula.
39
Multiple Choice
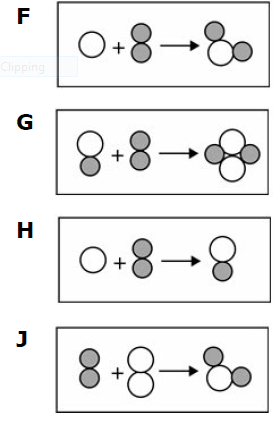
40
Multiple Choice
H2 + O → H2O
41
Multiple Choice
42
Multiple Choice
Based on the activity series, will this reaction occur?
Ni + HCl →
Yes
No
43
Multiple Choice
44
Multiple Choice
Without balancing the final equation, predict the products for the synthesis reaction: Li + Cl2 → ?
LiCl2
LiCl
Li2Cl2
Li2Cl
45
Multiple Choice
Without balancing the final equation, predict the products for the synthesis reaction: Li + Cl2 → ?
LiCl2
LiCl
Li2Cl2
Li2Cl
46
Multiple Choice
How many products are in a synthesis reaction?
there are never any products
only one product
there are seven products

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

43 questions
El imperfecto
Presentation
•
10th Grade

42 questions
Ionic Compound Writing
Presentation
•
10th Grade

41 questions
Untitled Lesson
Presentation
•
KG

42 questions
De_Chem_1.6_Volcanoes
Presentation
•
9th Grade

38 questions
Intro to Bonds
Presentation
•
9th - 11th Grade

37 questions
EOC Vocabulary Review English II
Presentation
•
10th Grade

39 questions
Colon and Semicolon use.
Presentation
•
10th Grade

40 questions
Concordância verbal
Presentation
•
KG
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

11 questions
EOC Domain 1: Cells
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
Exploring the Human Respiratory System
Interactive video
•
6th - 10th Grade

10 questions
Exploring Newton's Laws of Motion
Interactive video
•
6th - 10th Grade

10 questions
Exploring the Formation of the Solar System
Interactive video
•
6th - 10th Grade