

Module 2 Review (Macro-Water-Enzymes)
Presentation
•
Biology
•
9th - 12th Grade
•
Medium
+9
Standards-aligned
Gasmine Jean-Baptiste
Used 1+ times
FREE Resource
6 Slides • 27 Questions
1
Chemistry of Biology
By Gasmine Jean-Baptiste
2
Macromolecules
Sheltered Biology

3
Multiple Choice

carbohydrate
lipid
protein
nucleic acid
4
Multiple Choice

Which macromolecule is shown?
lipid
protein
nucleic acid
carbohydrate
5
Multiple Choice

Which macromolecule is being represented in the image?
nucleic Acid
lipid
carbohydrate
protein
6
Multiple Select
Which elements can be found in proteins?
C
H
N
P
O
7
Multiple Choice
What makes the carbohydrate different from the lipid?
The shape (carbohydrate contain a ring whereas lipids are long chains)
The type of atoms
The size of the molecule
They don't have any difference
8
Multiple Choice
Which elements (atoms) are found in ALL the macromolecules?
Carbon, Hydrogen, Nitrogen
Carbon, Hydrogen, Oxygen, Phosphorus
Carbon and Hydrogen
Carbon, Hydrogen and Oxygen
9
Enzymes
Biological Catalysts

10
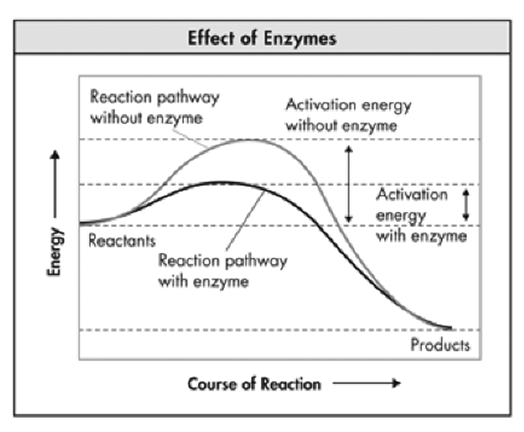
Enzymes speed up chemical reactions (Catalysts)
Activation energy is the energy needed to start a chemical reaction.
The less activation energy needed (smaller hill to climb) the faster the reaction will happen
Enzymes speed up reactions by lowering the activation energy

11
Multiple Choice
12
Multiple Choice

Enzymes are SPECIFIC. This means that an enzyme only works with molecules that are....
the right size and shape
broken down already
present in water
too big for the body to handle
13
Multiple Choice

14
Multiple Choice
15
Multiple Choice
16
Multiple Choice

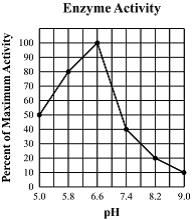
What can we conclude from the graph? At a a pH of 3..
Enzyme A works best
Enzyme B works best
Enzyme A works but low activity
Enzyme B works but low activity
17
Multiple Choice

18
Multiple Choice

This graph shows...
19
Multiple Choice
20
Denaturation = to lose shape
Structure (shape) = function
If enzymes lose their shape they no longer function correctly
Lower enzyme function = slower reaction rates
Factors that can affect shape & function of enzymes: Temperature, pH, salinity (ion concentration)

21
Multiple Choice

22

23
Multiple Choice
The oxygen atom in water is slightly ____.
negative
positive
24
Multiple Choice
The reason water sticks to your skin is ___.
adhesion
cohesion
25
Multiple Choice
The reason there are trillions of water molecules in a droplet is because of ___.
adhesion
cohesion
26
Multiple Choice
Water's high boiling point is due to ___.
hydrogen bonds
it's polarity
being nonpolar
27
Multiple Select
A high specific heat means water ____. (select all that are true)
takes a long time to change temperature.
takes a lot of heat to increase temperature.
holds a lot of heat.
is good at regulating Earth's temperature.
28
Multiple Choice
Because solid water is less dense than liquid water, ___ floats.
solid water (ice)
liquid water
29
Multiple Choice
What kind of molecule is water?
Ionic
Polar Covalent
Nonpolar Covalent
Metallic
30
Multiple Choice

You body sweats to help prevent you from overheating. How does water regulate temperature?
it lets heat pass through it
it absorbs a lot of heat, then decreases in temperature
it absorbs a lot of heat, but only increases a little in temperature
heat is afraid of water and avoids it
31
Multiple Choice

A water strider can skate along the top of a pond because:
covalent bonds result in water cohesion (surface tension)
hydrogen bonds result in water cohesion (surface tension)
water striders have adapted to take advantage of water cohesion
water striders are afraid of water and avoid it
32
Multiple Choice
33
Multiple Choice

Blood functions as a transport mechanism to take nutrients and oxygen to all cells in the body. What property of water allows for this to occur?
Expansion Upon Freezing
Universal Solvent
Cohesive Behavior
Ability to Moderate Temperature
Chemistry of Biology
By Gasmine Jean-Baptiste
Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

26 questions
Present Tense Verbs
Presentation
•
9th - 12th Grade

25 questions
Introduction to Electrons
Presentation
•
9th - 12th Grade

25 questions
Byzantine Empire
Presentation
•
9th - 12th Grade

25 questions
Simple Past Tense
Presentation
•
9th - 12th Grade

28 questions
Creature Quiz-Owls
Presentation
•
9th - 12th Grade

26 questions
Theory of Evolution
Presentation
•
9th - 12th Grade

26 questions
Nucleic Acids
Presentation
•
9th - 12th Grade

25 questions
3.1.2 RNA/Transcription
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Biology

15 questions
Biology EOC Review P2
Quiz
•
10th Grade

20 questions
Biology EOC Review 1
Quiz
•
10th Grade

33 questions
Sustainability Unit Review
Quiz
•
9th Grade

36 questions
Biology Regents Review
Quiz
•
9th - 10th Grade

50 questions
NC EOC Biology Review part 1
Quiz
•
9th - 12th Grade

144 questions
Bio 2nd Semester Final Review
Quiz
•
9th Grade

60 questions
Biology Regents Review
Quiz
•
9th Grade

40 questions
Biology EOC Review
Quiz
•
11th Grade