

4-1 Light
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Hard
Standards-aligned
Tim Haberfield
FREE Resource
9 Slides • 8 Questions
1

2
Multiple Choice
What is the main focus of the lesson titled 'Light and Quantum Theory: Understanding Electromagnetic Radiation'?
Understanding light behavior
Exploring quantum mechanics
Studying electromagnetic waves
All of the above
3

4
Multiple Choice
What are the unanswered key questions regarding Rutherford's nuclear model?
Electron arrangement around nucleus
Why electrons don't collapse into nucleus
Chemical behavior differences among elements
All of the above
5

6
Multiple Choice
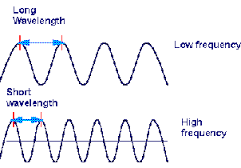
What are the properties of waves as described in the image?
Wavelength, Frequency, Amplitude
Speed, Direction, Frequency
Amplitude, Velocity, Wavelength
Frequency, Speed, Direction
7

8

9
Multiple Choice
10

11
Multiple Choice
What is the significance of Max Planck's discovery in 1900 regarding energy?
It introduced the concept of energy being continuous
It established that energy is quantized
It explained the behavior of heated objects
It laid the foundation for classical physics
12

13
Multiple Choice
What is the relationship between light frequency and electron energy in the photoelectric effect?
Higher frequency leads to lower electron energy
Higher frequency leads to higher electron energy
Light frequency has no effect on electron energy
Electron energy is constant regardless of light frequency
14

15
Multiple Choice
What did Einstein introduce in 1905 regarding light?
Wave properties
Particle properties
Both wave and particle properties
None of the above
16

17
Multiple Choice
Which electron transition between the energy
levels of hydrogen causes the emission of a photon of
visible light?
𝑛 = 6 to 𝑛 = 5
𝑛 = 5 to 𝑛 = 6
𝑛 = 5 to 𝑛 = 2
𝑛 = 2 to 𝑛 = 5

Show answer
Auto Play
Slide 1 / 17
SLIDE
Similar Resources on Wayground

15 questions
Gas Laws: Part 2
Presentation
•
10th Grade

13 questions
Trigonometry-Finding Missing Side
Presentation
•
10th Grade

13 questions
Advanced Mole Conversions
Presentation
•
10th Grade

11 questions
Density
Presentation
•
10th Grade

14 questions
Gas Laws - Boyle's and Charles'
Presentation
•
10th Grade

14 questions
Acid Naming & Formula Writing
Presentation
•
10th - 11th Grade

12 questions
Gas Laws
Presentation
•
10th Grade

15 questions
Arrhenius and Bronsted-Lowry Acids and Bases
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade